Honorio T. Benzon
Pain is defined by the International Association for the Study of Pain (IASP) as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage” (1). Acute pain is a normal physiologic response to injury, disease, or surgery and is usually temporally self-limited. Though unpleasant, pain is protective and serves the purpose of avoiding, stopping, or minimizing tissue damage and should be seen as a symptom of an underlying disease. Chronic pain is usually defined as pain lasting more than 3 months. It can be due to ongoing disease or tissue injury or can persist after resolution of or in the absence of injury. Chronic pain is associated with neuroplastic changes in the central and peripheral nervous systems that may manifest as hypersensitivity, windup, and allodynia. When these changes occur, the pain itself can be called a disease state.

When chronic pain is associated with neuroplastic changes in the central and peripheral nervous systems that may manifest as hypersensitivity, windup, and allodynia, the pain itself becomes a disease state.
Nociceptive pain results from transmission of a noxious stimulus through an intact nervous system. Nociceptive pain can be worsened by inflammation, which causes hyperalgesia, the phenomenon of normally painful stimuli being perceived as more painful than usual. Nociceptive pain can be somatic or visceral. Somatic pain originates within the skin, superficial tissue, and musculoskeletal system and is typically easy to localize and described as sharp. Visceral pain is typically vague, diffuse, achy, and may refer to surrounding areas.
In contrast to nociceptive pain, neuropathic pain results from a lesion in the central or peripheral nervous system. It is often described as electric or lancinating in character. If this is found in the distribution of a known nerve, it is termed neuralgia. Neuropathic pain is often associated with altered sensations. Paresthesias are abnormal, spontaneous, or evoked sensations. Dysesthesia are unpleasant abnormal sensations. Allodynia is the perception of pain from a normally nonpainful stimulus (such as light touch). Hyperesthesia is increased sensitivity to stimulation, and hypoesthesia is decreased sensation of stimulus.
I. Anatomy, Physiology, and Neurochemistry of Pain
A. Pain Processing
The physiology of pain processing functionally comprises four steps: transduction, transmission, modulation, and perception. These processes are clinically relevant as each provides targets for pain treatment and prevention (Fig. 37-1). Transduction is the generation of an action potential from a noxious chemical, mechanical, or thermal stimulus. Transmission is the propagation of the signal through the afferent pathway from the nociceptor to the sensory cortex. Modulation is the positive or negative modification of the pain signal along the afferent pathway, while perception is the integration of the pain signal into consciousness.
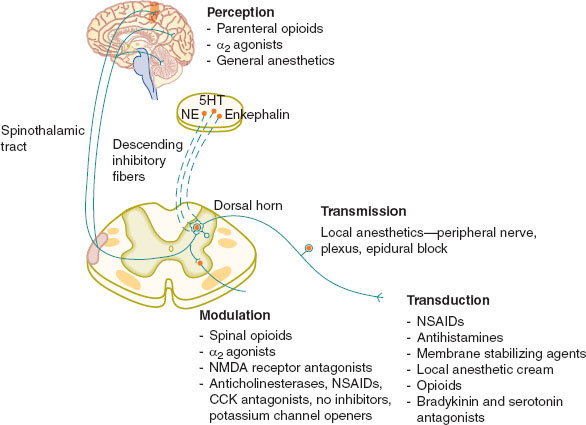
FIGURE 37-1 The four elements of pain processing: transduction, transmission, modulation, and perception. (From Macres SM, Moore PG, Fishman SM. Acute pain management. In: Barash PG, Cullen BF, Stoelting RK, et al., eds. Clinical Anesthesia. 7th ed. Philadelphia: Lippincott Williams & Wilkins; 2013:1611–1642, with permission.)
 VIDEO 37-1
VIDEO 37-1
Pain Processing
B. Transduction
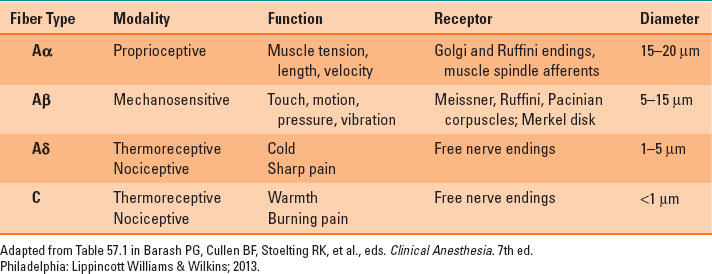
Nociceptors are located in the skin, mucosa, muscle, fascia, joint capsules, dura, viscera, and adventitia of blood vessels. Most Aδ and C nociceptors are polymodal (i.e., their terminals express transducer channels that are sensitive to multiple stimuli). When they are activated by pressure, chemical, or thermal stimuli, the channels activate voltage-sensitive sodium and calcium channels, starting an action potential. Nociceptors can be activated by bradykinin, serotonin, and protons and sensitized by prostaglandins, leukotrienes, and cytokines. Glutamate, substance P, and nerve growth factor can also promote transduction of a pain signal (Table 37-1).
Table 37-1 Classification of Neural Fibers
C. Transmission
Pain transmission occurs via a three-neuron afferent pathway, beginning in the periphery (Fig. 37-2). First-order neuronal cell bodies are located in the dorsal root ganglia with fibers projecting to peripheral tissue where the receptors are located. Fibers enter the spinal cord and travel up or down through the posterolateral tract before entering the dorsal horn to synapse on second-order neurons. Second-order neuron cell bodies are located in the dorsal horn and are either nociceptive specific or wide dynamic range. Axons transmitting somatic nociception decussate and ascend via the contralateral spinothalamic tract, while axons transmitting visceral nociception ascend via the ipsilateral dorsal column medial lemniscus. Both synapse on the third-order neurons in the thalamus, the axons of which terminate in the sensory cortex. In the face, the primary afferent neuron has its cell body in the trigeminal ganglion and synapses on the second-order neuron in the medulla in the spinal trigeminal nucleus. From here the signal is transmitted to the thalamus, as are pain signals from the rest of the body.
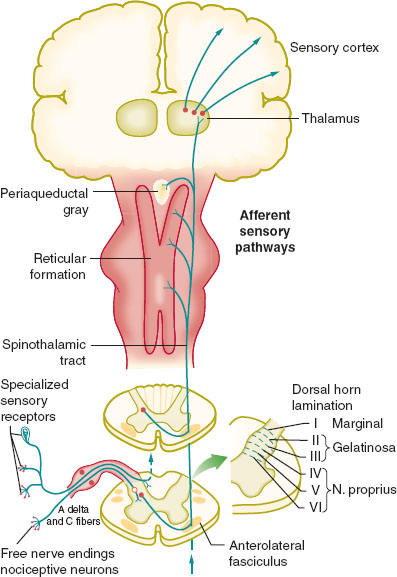
FIGURE 37-2 The afferent nociceptive pathway. (From Macres SM, Moore PG, Fishman SM. Acute pain management. In: Barash PG, Cullen BF, Stoelting RK, et al., eds. Clinical Anesthesia. 7th ed. Philadelphia: Lippincott Williams & Wilkins; 2013:1611–1642, with permission.)
 VIDEO 37-2
VIDEO 37-2
Nociceptive Pathways
D. Modulation
Modulation of the pain response occurs at many levels and can be positive or negative (Fig. 37-3). Activity between first- and second-order neurons is decreased by feedback from interneurons and descending inhibition from the periaqueductal gray matter, rostral ventromedial medulla, and the dorsolateral pontine tegmentum. Augmentation of pain may occur as part of the transition from acute to chronic pain. Repetitive activation of wide dynamic range neurons by C fibers causes windup. Axonal sprouting causes crosstalk between different fibers, causing nonnoxious stimuli to become painful. In addition, neuromas and axonal sprouting may be associated with upregulation of sodium and downregulation of potassium channels, which cause destabilized cell membranes to become more prone to forming an action potential. Normal Aβ fibers do not produce substance P, but in the presence of tumor necrosis factor-α from injury, Aβ fibers can secrete it. This transition is termed phenotypic switch.
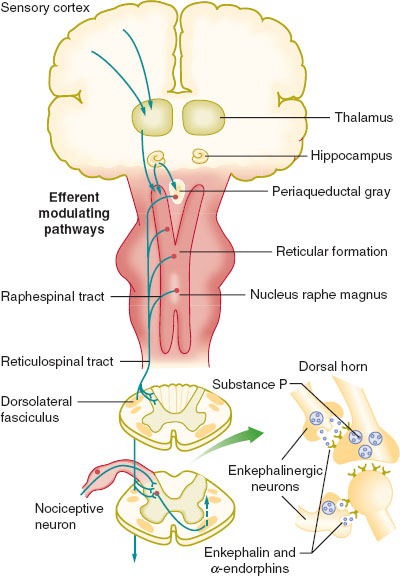
FIGURE 37-3 The efferent pathway for modulation of nociception. (From Macres SM, Moore PG, Fishman SM. Acute pain management. In: Barash PG, Cullen BF, Stoelting RK, et al., eds. Clinical Anesthesia. 7th ed. Philadelphia: Lippincott Williams & Wilkins; 2013:1611–1642, with permission.)
E. Perception
Perception of pain is mediated by multiple structures. The primary and secondary somatosensory cortices are involved with sensory discrimination of pain. The frontal cortex and insula may facilitate learning and memory of pain. The anterior cingulate gyrus is related to emotional significance of pain, while the lentiform nucleus and cerebellum are involved in self-protective reflexes related to pain.
II. Assessment of Pain
Pain is a highly subjective experience and affects many aspects of life. As such, the evaluation of the patient in pain relies primarily on patient-reported information and should include assessment of multiple domains. Because pain is dynamic, it should be reassessed regularly and adjustments to therapy made as appropriate. The location of the pain and where it radiates, if at all, are important. In the postoperative setting, it is still necessary to ask where the pain is located rather than presume the pain is incisional. Patients may have additional pain related to pre-existing conditions, positioning or retraction during surgery, and immobility. The onset and temporal pattern as well as exacerbating and ameliorating factors are established. The quality of the pain can help indicate its origin and possible treatment options—sharp incisional pain may respond well to nonsteroidal anti-inflammatory drugs (NSAIDs), opioids, and nerve blocks, but shooting neuropathic pain may respond to an N-methyl-D-aspartate (NMDA) antagonist, clonidine, or antiepileptic agent.
The intensity of pain should be assessed in multiple contexts. The baseline level of pain is the pain that exists at all times, while breakthrough pain escalates beyond the background. Multiple tools exist to aid in the assessment of pain intensity, all of which are arbitrary, subjective, and have a high degree of variability between patients. Despite these flaws, they are useful to determine trends in pain control. The numerical rating scale is most commonly used, which asks patients to rate their pain from 0, no pain, to 10, worst pain imaginable. For young children or patients with cognitive impairments, the facial grimace scale allows for a more descriptive approach (Fig. 37-4).
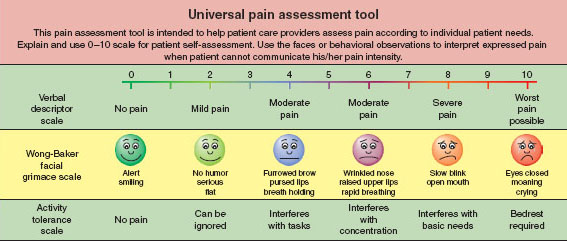
FIGURE 37-4 Universal pain assessment tool. Different scales can be used depending on the patient’s age and other medical conditions. (From Macres SM, Moore PG, Fishman SM. Acute pain management. In: Barash PG, Cullen BF, Stoelting RK, et al., eds. Clinical Anesthesia. 7th ed. Philadelphia: Lippincott Williams & Wilkins; 2013:1611–1642, with permission.)
 VIDEO 37-3
VIDEO 37-3
Universal Pain Assessment Tool
Because pain impacts many activities, it is important to investigate the patient’s functional status including the ability to eat, sleep, ambulate, work, and perform one’s activities of daily living. Any side effects from pain therapy should be thoroughly discussed as well. Physical examination is focused on possible etiologies of pain and should include a thorough neurologic and musculoskeletal examination.
III. Pharmacologic Management of Pain
A. Opioids
Opioids are useful for acute pain and cancer-related pain and can be a component of a chronic pain regimen. In acute pain, short-acting agents are typically used alone. In chronic pain, 80% of the daily dose is given in a basal long-acting medication with the remainder given as a short acting opioid as needed for breakthrough. When assessing patients for opioid management, it is important to inquire about the level of analgesia provided, whether the patient’s functional status is improved, whether there are side effects of therapy, and whether the patient displays aberrant behaviors (i.e., early refills, lost pills).
Opioids bind to the μ, κ, and δ receptors to cause analgesia and side effects, such as pruritus, nausea, constipation, and respiratory depression. Various opioids have different potencies, bioavailabilities, and dosages (Table 37-2). For ease of comparison and conversion from one medication to another, they are all compared to the prototypical opioid, morphine. When converting from one opioid to another, it is important to reduce the expected dosage by 25% to 50% to account for incomplete cross-tolerance to the new agent. When treating patients with opioids, precision of vocabulary is important. Tolerance is the phenomenon of decreased effect of a given amount of medication. It usually occurs after prolonged administration of the drug. Dependence is the physiologic condition of withdrawal symptoms when an opioid is discontinued. Addiction is a disease marked by altered behavior to seek the desired substance despite negative consequences. Pseudo-addiction is aberrant drug-seeking behavior due to undertreatment of pain.

It is important to know the differences between drug tolerance, dependence, addiction, and pseudo-addiction.
Morphine is metabolized by the liver to morphine-6-glucuronide (M6G) and morphine-3-glucuronide (M3G), which are renally excreted. M6G is analgesic due to μ-binding activity and is responsible for respiratory depression, sedation, and nausea. M3G has no μ effect and is associated with hyperalgesia, seizures, and tolerance. Morphine’s half-life is 2 hours, but its duration of action is 4 to 5 hours due to its slow elimination from the brain compartment.
Hydromorphone is five times more potent than morphine and is associated with fewer side effects. It is metabolized in the liver to dihydromorphine and dihydroisomorphine, which are active, and hydromorphone-3-glucuronide, which does not cause analgesia but is similar in side effects to M3G. Its onset is 15 minutes when administered intravenously and its duration of action is similar to that of morphine.
Table 37-2 Opioid Pharmacokinetics and Equianalgesic Dosing
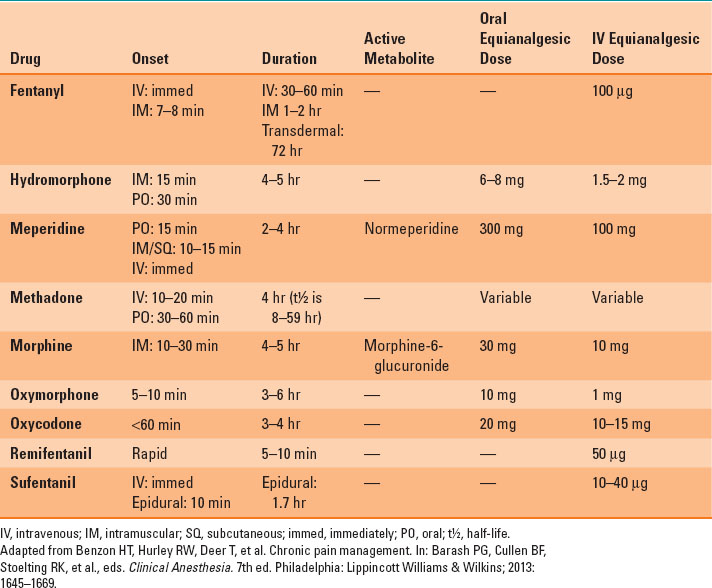
Fentanyl is 80 times more potent than morphine and is associated with less histamine release and pruritus. It is more lipophilic than morphine. It is metabolized by the liver and is appropriate for patients with renal failure. It is available as a transdermal patch. Due to gradual absorption of the drug, the patch requires 6 to 8 hours to reach maximum plasma concentrations. The patch provides steady-state analgesia without periods of side effects related to high serum concentrations and periods of pain due to low serum concentrations. After removal of the patch, significant serum levels remain, hence, intramuscular opioid should not be given immediately.
Sufentanil is 1,000 times as potent as morphine and is typically used in intraoperative infusions or neuraxially. It has a slightly shorter elimination half-life than fentanyl. Alfentanil is 10 times the potency of morphine and has its peak effect within 2 minutes. It has a very short duration of action, <10 minutes, and is ideal for brief periods of intraoperative stimulation. Remifentanil is approximately 100 times the potency of morphine. Like alfentanil, it is also rapid acting. It is eliminated by plasma cholinesterases, so its terminal half-life is 10 to 20 minutes. Its termination of analgesia is so rapid that it may result in rebound hyperalgesia.
Methadone is a unique opioid because it enhances analgesia by antagonizing the NMDA receptor and inhibiting serotonin reuptake in addition to its μ effect. It is metabolized in the liver by cytochrome P450 and has many drug interactions. It has a variably long elimination half-life between 8 and 80 hours, requiring slow titration to avoid accidental overdose. After a single dose, it provides analgesia for 3 to 6 hours, but with prolonged around-the-clock dosing, the duration of analgesia can be 8 to 12 hours. Methadone can cause prolonged QT and torsades de pointes, requiring periodic electrocardiograms.
Meperidine is a short-acting opioid that is metabolized in the liver to normeperidine, which can be neurotoxic and result in seizures, especially in the setting of renal failure or prolonged dosing. It is indicated for short-term use only. Its most common usage is in low doses to treat postoperative rigors.
Oxycodone is activated by conversion to oxymorphone. Both drugs are associated with less pruritus than morphine. Tramadol, hydrocodone, and codeine are considered weak opioids. Tramadol is a μ agonist with monoaminergic properties. It carries low rates of constipation, respiratory failure, and abuse. Codeine is a prodrug that is metabolized to morphine by cytochrome P450 2D6. Reductions in enzymatic activity, as seen in children and certain ethnic groups (whites and Asians), cause decreased analgesia and increased respiratory depression.
B. Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
NSAIDs work by inhibiting cyclooxygenase (COX) enzymes, exerting anti-inflammatory, antipyretic, and analgesic effects. COX-1 is present in healthy tissue and serves gastroprotective and hemostatic functions. COX-2 is induced in injury and produces prostaglandins that sensitize peripheral nociceptors to pain and promote hyperalgesia. NSAIDs are effective at reducing postoperative pain and opioid consumption and are commonly used in both acute and chronic pain. Side effects include platelet dysfunction, nephrotoxicity, and gastric ulcers. Acetaminophen is a centrally acting COX inhibitor with minimal peripheral action. It causes analgesia and antipyrexia, but has no anti-inflammatory effect.

Acetaminophen is a centrally acting COX inhibitor with minimal peripheral action that causes analgesia and antipyrexia but has no anti-inflammatory effect.
C. Anticonvulsants
Chronic nerve damage is associated with spontaneous ectopic firing of neurons and changes in sodium and calcium channel expression. Anticonvulsants reduce ectopic signals by blocking sodium or calcium channels. Gabapentin and pregabalin both block α2-δ subunit of calcium channels. Both have been shown to be helpful in multiple neuropathic pain syndromes, including postherpetic neuralgia (PHN), diabetic painful neuropathy (DPN), trigeminal neuralgia, human immunodeficiency virus (HIV) neuropathy, spinal cord injury pain, phantom limb pain, and poststroke pain. In acute pain, preoperative gabapentin has been shown to decrease narcotic requirements, improve pain control, and reduce opioid-related side effects. Neither gabapentin nor pregabalin have significant drug–drug interactions. Side effects of both drugs include dizziness, fatigue, peripheral edema, and cognitive slowing.

Chronic nerve damage is associated with spontaneous ectopic firing of neurons and anticonvulsants reduce ectopic signals by blocking sodium or calcium channels. Thus, anticonvulsants may be useful in treating neuropathic pain.
D. Antidepressants
Tricyclic antidepressants (TCAs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) exert an independent analgesic effect distinct from their mood stabilizing properties. TCAs affect many pathways, including inhibition of the reuptake of serotonin and adenosine, interaction with α receptors, opioid receptor binding, and blockade of sodium channels, calcium channels, and NMDA receptors. They are effective at treating neuropathic pain, especially postherpetic neuralgia (PHN) and diabetic peripheral neuropathy (DPN), but frequent side effects limit their use. These include sedation, xerostomia, urinary retention, and blurred vision and tend to be more pronounced in the elderly. Nortriptyline and desipramine are better tolerated than amitriptyline.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








