The kidneys receive the largest percentage of the core organ cardiac output (25%).
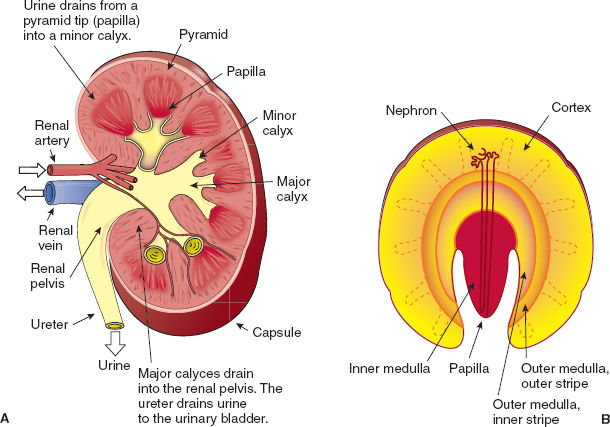
Figure 5-1 A: Gross anatomy of the kidney. B: Medullary inner and outer stripes. (From Preston RR, Wilson TE. Filtration and micturition. In: Harvey RA, ed. Physiology. Philadelphia: Lippincott Williams & Wilkins, 2013, with permission.)
The renal parenchyma is enclosed by a tough but thin fibrous membrane (except for the hilus) and is divided into two distinct regions: the cortex and the medulla (Fig. 5-1). The cortex is the outer portion of the kidney and contains alternating bands of cortical labyrinth (glomeruli and convoluted tubules) and parallel arrays of straight tubules (medullary rays). The medulla is the deeper part of the parenchyma and is divided into an outer region, which contains the thick ascending limb of the loop of Henle, and the inner region, which is marked by the absence of loops of Henle. The outer region of the medulla is itself divided into an outer and inner stripe, which are defined by the presence (outer) or absence (inner) of proximal tubules (Fig. 5-1). Tubules in the medulla are arranged into pyramids, which are oriented with the base toward the cortex and the tip (papilla) toward a minor calyx to where urine drains.
B. Physiology: Correlation of Structure and Function
The Nephron
The nephron is the structural and functional unit of the kidney and is responsible for urine formation. It plays a dominant role in water and electrolyte homeostasis, acid base balance, and blood pressure control. The nephron comprises a glomerulus and a tubule (Fig. 5-2). The glomerulus is a capillary tuft encased in a fibrous structure called Bowman’s capsule, together known as the renal corpuscle. This is where filtration of blood occurs. Absorption and secretion occur in the renal tubule (proximal convoluted tubule, loop of Henle, and distal convoluted tubule). Filtrate from each nephron drains into the collecting duct system and pass toward the renal calyces.
 VIDEO 5-1
VIDEO 5-1
Renal Regulation of Water
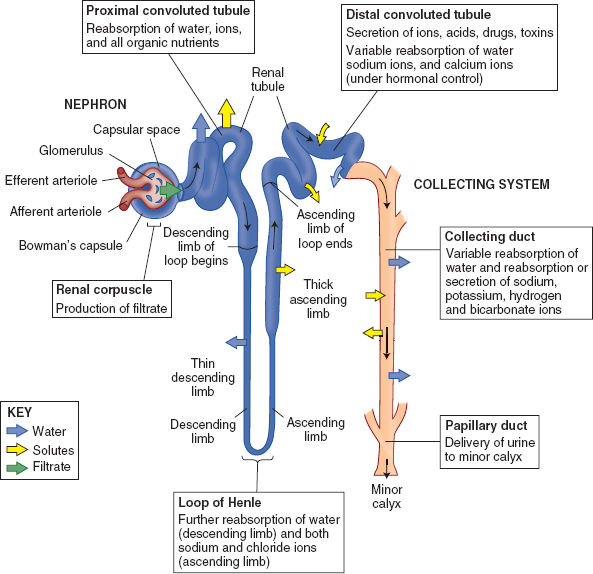
Figure 5-2 The nephron: Structure and function.
The Glomerulus
The glomeruli are supplied by an afferent arteriole and drained by an efferent arteriole. They can be divided into superficial glomeruli (80% to 85% of glomeruli; located near the renal capsule and associated with short loops of Henle) and juxtamedullary glomeruli (15% to 20% of glomeruli, which have long loops of Henle extending deep into the medulla) (Fig. 5-3). The capillaries have three layers (fenestrated endothelium on the blood side, visceral epithelium on the filtrate side, and a basement membrane in between), which create a filtration barrier and provide selective filtration of the blood.

The macula densa cells are positioned to sense changes in the tubular fluids and interact with the effector cells of the juxtaglomerular apparatus, creating changes in blood flow and glomerular filtration rate (tubuloglomerular feedback).
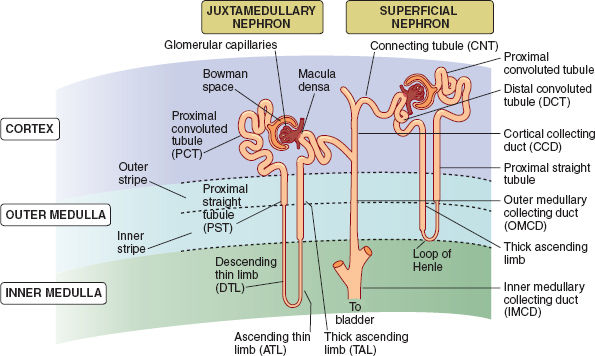
Figure 5-3 Nephron types and the collecting duct system. (From Preston RR, Wilson TE: Filtration and micturition. In: Harvey RA, ed. Physiology. Philadelphia: Lippincott Williams & Wilkins, 2013, with permission.)
The Juxtaglomerular Apparatus
The juxtaglomerular apparatus comprises the afferent and efferent arterioles, extraglomerular mesangial cells, and the macula densa (Fig. 5-4). The macula densa is a region of the specialized, distal, thick ascending limb of the loop of Henle of the parent nephron. It is formed from low columnar cells that have their apical membranes exposed to the tubular fluid and the basilar aspect in contact with cells of the mesangium and the afferent arteriole. Gap junctions exist between the mesangial cells, serving as a functional link between the macula densa, glomerular arterioles, and mesangium.
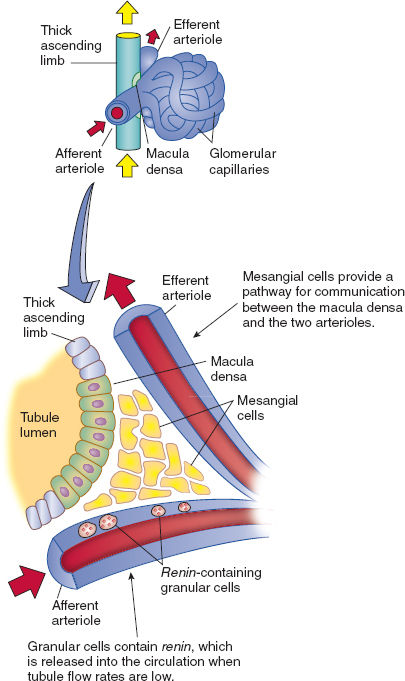
Figure 5-4 Juxtaglomerular apparatus. (From Preston RR, Wilson TE. Filtration and micturition. In: Harvey RA, ed. Physiology. Philadelphia: Lippincott Williams & Wilkins, 2013, with permission.)
Proximal Tubule and Loop of Henle
The cellular structure of the proximal tubule is highly specialized, reflecting the high energy demands required for a range of complex transport functions (Table 5-1). Virtually no active transport occurs in the descending part of the loop of Henle (Fig. 5-4). Urine concentration occurs in this part of the loop through passive urea transporters and simple water channels. The thick ascending limb is where sodium and potassium adenosine triphosphatase (Na, K-ATPase)–driven active transport resorbs sodium and chloride. The macula densa, is the modified part of the thick ascending limb of the loop of Henle which forms part of the juxtaglomerular apparatus (1).
Table 5-1 Relations of Cellular Structure and Function in the Proximal Tubule
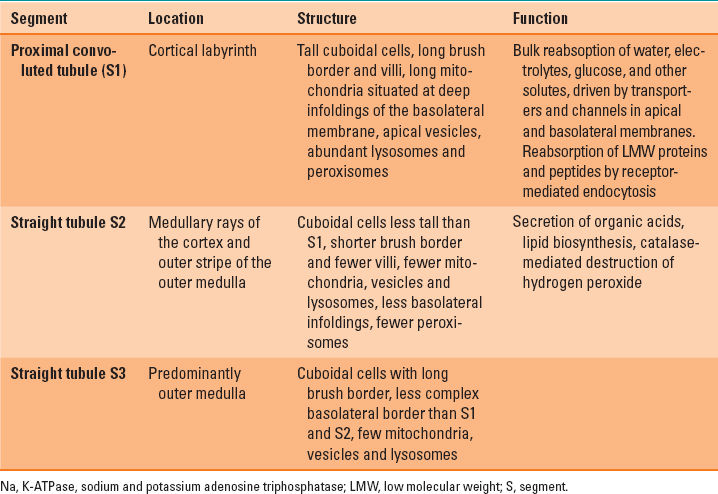
The Distal Tubule
The distal tubule is composed of the distal convoluted tubule, the connecting segment, and the initial collecting tubule (Fig. 5-3). The cells of the distal convoluted tubule actively transport sodium and have calcium ATPase, which is important in the reabsorption of divalent cations. The connecting segments lie within the cortical labyrinth, where several join together to form the collecting duct. Cells of the connecting segment are similarly involved in sodium and cation transport but, unlike the distal convoluted tubule cells, they have water channels.
C. Glomerular Filtration Rate
The glomerular filtration rate is the volume of filtrate formed by both kidneys per minute. This is approximately 125 mL/min in an average patient with normal renal function. Fluid and solutes are forced under pressure from the glomerulus (afferent arteriole) into the capsular space (enclosed by Bowman’s capsule) of the renal corpuscle. A filtration membrane allows the passage of fluid and small solutes into the capsular space based on their physical size and charge. Filtration occurs by bulk flow driven by the hydrostatic pressure of the blood; small molecules pass rapidly through the filtration membrane while larger molecules are retained within the arteriole. The relatively large diameter of the afferent arterioles and small diameter of the efferent arterioles result in a high capillary pressure (~60 mm Hg). This driving pressure is opposed by the back pressure of the capsular hydrostatic pressure (~15 mm Hg). Although the concentration of the small solutes is the same across the filtration membrane, large proteins are retained and the osmotic pressure of the blood increases as the fluid moves out of the glomerulus. This results in an overall net filtration pressure of approximately 17 mm Hg. There is a direct relation between the net filtration pressure and the glomerular filtration rate. If either the hydrostatic or osmotic pressure of the glomerular capillaries or the hydrostatic pressure of the capsular space changes, the glomerular filtration rate will also change.

Glomerular filtration is the primary determinant of urine composition.
D. Autoregulation of Renal Blood Flow and Glomerular Filtration
Autoregulation of renal blood flow and glomerular filtration rate are intimately related. In normal humans, renal blood flow is maintained nearly constant over the range of 70 to 120 mm Hg mean systemic arterial pressure (Fig. 5-5). There are three main mechanisms whereby renal blood flow and thus glomerular filtration rate are regulated: myogenic response, tubuloglomerular feedback, and sympathetic nervous system stimulation.

Both renal blood flow and glomerular filtration rate are autoregulated within very narrow limits.
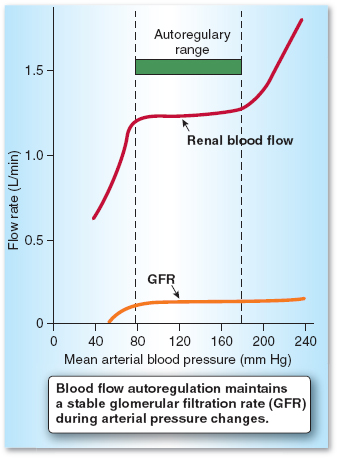
Figure 5-5 Autoregulation of renal blood flow. (From Preston RR, Wilson TE. Filtration and micturition. In: Harvey RA, ed. Physiology. Philadelphia: Lippincott Williams & Wilkins, 2013, with permission.)
Myogenic Response
The myogenic response is the intrinsic property of vascular smooth muscle, whereby arterioles constrict in response to increased transmural pressure and allow a relatively constant blood flow and glomerular filtration rate (2).
The Tubuloglomerular Feedback
The tubuloglomerular feedback mechanism occurs via the macula densa. By virtue of its proximity to the afferent arteriole (Fig. 5-4), it is perfectly positioned to create a feedback loop controlling blood flow through the glomerulus. If the myogenic response does not fully regulate blood flow through the glomerulus, the increased capillary pressure increases the glomerular filtration rate, inhibiting sodium reabsorption in the proximal tubule. The increased delivery of sodium chloride in the tubular fluid reaching the macula densa in the distal tubule results in vasoconstriction of the afferent arteriole (tubuloglomerular feedback). Increased sodium transport in the distal tubule is not the only mechanism triggering tubuloglomerular feedback. Tubular fluid flow, independent of sodium concentration, is sensed by primary cilia located on the apical (luminal) aspect of the macula densa cells.
Sympathetic Nervous System
During periods of stress, such as hypotension or hemorrhage, sympathetic stimulation overrides autoregulation. Increased sympathetic discharge causes intense constriction of all of the renal vessels, reducing the glomerular filtration rate and subsequent fluid and electrolyte losses. Increased renal sympathetic activity decreases sodium and water excretion by 1) increasing tubular water and sodium reabsorption throughout the nephron, 2) decreasing renal blood flow and glomerular filtration rate by vasoconstriction of the arterioles, and 3) increasing activity of the renin angiotensin system by releasing renin from the juxtaglomerular granular cells (3).

The renal sympathetic nerves innervate the arterioles, the tubules, and the juxtaglomerular apparatus. This regulates blood flow, glomerular filtration, sodium and water reabsorption, and activity of the renin angiotensin system.
E. Tubular Reabsorption of Sodium and Water
Sodium moves freely from the glomerulus across the filtration membrane into Bowman’s capsule and has the same concentration in the tubular fluid as it has in the plasma. Approximately two-thirds of the filtrate reaching the proximal tubule is reabsorbed and is regulated both acutely and chronically by blood pressure, extracellular fluid volume, renin angiotensin system, sympathetic nervous system, and an intrarenal dopamine natriuretic system (4). The bulk of sodium chloride, bicarbonate, phosphate, glucose, water, and other substrates is reabsorbed into the tubular cells as they move passively down their concentration gradients (Fig. 5-6). Sodium potassium ATPase in the basolateral membrane then actively pumps sodium out of the cell into the interstitial fluid, maintaining a low intracellular concentration. This maintains the driving force for sodium entry from the tubular fluid at the luminal side. The colloid oncotic pressure in the capillaries accompanying the proximal tubules is high because large molecules are retained by the filtration membrane of Bowman’s capsule. Sodium and water are reabsorbed from the interstitial fluid into the capillaries by bulk flow mediated by both hydrostatic and osmotic forces. The fraction of sodium reabsorbed in the proximal tubule varies according to prevailing conditions (Table 5-2). Water reabsorption occurs passively in the proximal tubule by osmosis and is coupled with sodium transport through the cells. Water also passes through the tight junctions between cells, which allows for diffusion of water and small ions.
Table 5-2 Factors Affecting Sodium Reabsorption by the Renal Tubule
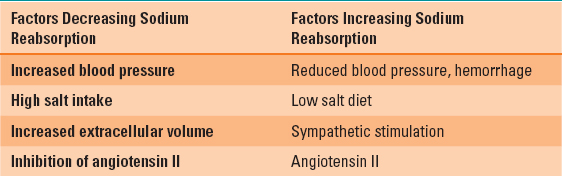
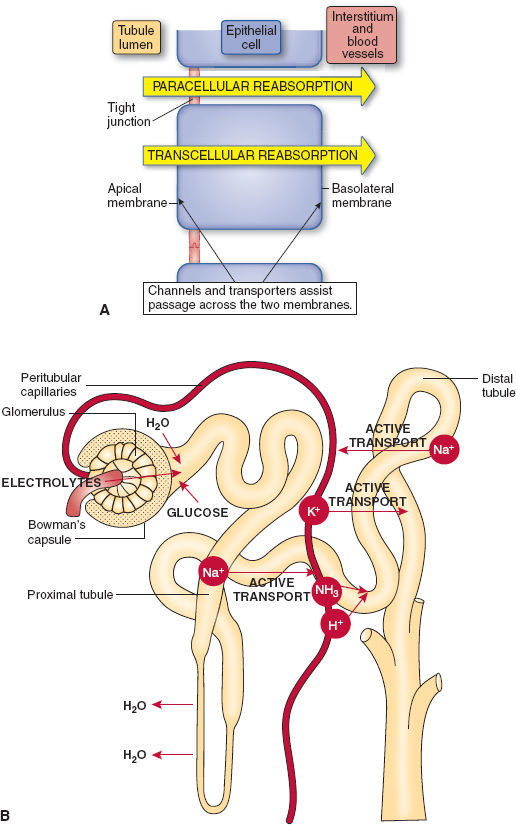
Figure 5-6 A: Reabsorption from tubular lumen. (From Preston RR, Wilson TE. Filtration and micturition. In: Harvey RA, ed. Physiology. Philadelphia: Lippincott Williams & Wilkins, 2013, with permission.) B: Transport of substances across glomerulus. (From Straight A’s in Anatomy and Physiology. Ambler, PA: Lippincott Williams & Wilkins; 2007:313, with permission.)
The loop of Henle has three distinct regions: the thin descending segment, the thin ascending segment, and the thick ascending segment. The thin segments have thin membranes with no brush borders and few mitochondria (Fig. 5-3) because of low metabolic requirements. There is little active
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








