III. Postoperative Pain Management
Effective control of surgical pain with minimal side effects during and beyond the PACU interval should be a high priority, even if large doses of analgesics are necessary (see Chapter 37). The correlation between the patient’s perception of pain and observed sympathetic nervous system (SNS) response varies widely due to cardiovascular, psychological, and cultural factors. Assessment of discomfort is difficult and imprecise. Use of a quantitative pain scale yields more reliable results. PACU staff should ensure that the nature and intensity of the patient’s pain are appropriate so as to avoid masking an evolving complication. For example, tachycardia or hypertension can reflect pain, but they can also be caused by hypoxemia, cerebral hypoperfusion, or hypovolemia. Administration of analgesia may accentuate these phenomena. Fear and anxiety accentuate pain, so titration of an intravenous sedative such as midazolam may be a useful adjunct to analgesics.

Opioids are relatively ineffective sedatives, and benzodiazepines are ineffective analgesics. It is important to choose the correct class of drug when treating postoperative pain and/or anxiety.
IV. Discharge Criteria
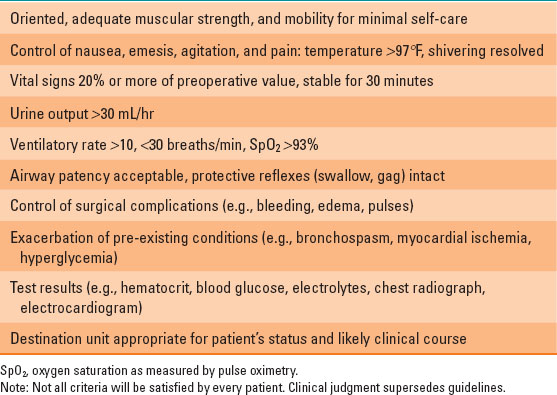
Prior to PACU discharge, patients should be assessed using consistent criteria that ensure that the patient will have sufficient reserve to tolerate minor deterioration after discharge (Table 39-2). Simple scoring systems exist, such as the Aldrete scale, which quantifies physical status and vital sign thresholds, but they lack sensitivity and specificity for identifying subtle problems. Oxygen saturation as measured by pulse oximetry (SpO2) should be satisfactory while the patient breathes room air prior to discharge. Inpatients and selected ambulatory patients can be discharged before they void if urination is carefully tracked after discharge.

Some PACU discharge criteria, such as ability to void or recovery from a peripheral nerve block, can be bypassed following a few procedures in selected patients, but the patients must be given explicit instructions and capable of following them.
V. Postoperative Hypotension
There are multiple causes for hypotension in the PACU. Hypotension causes hypoperfusion of vital organs, anaerobic metabolism, and lactic acidemia. Symptoms referable to the brain or heart (e.g., disorientation, nausea, unconsciousness, angina) indicate that the patient’s ability to compensate for hypotension is exhausted. Morbidity is higher in patients with chronic hypertension, arteriosclerotic disease, increased intracranial pressure (ICP), and renal insufficiency.
Table 39-2 Guidelines for Postanesthesia Care Unit Discharge
Low intravascular volume (absolute hypovolemia) caused by inadequate replacement of fluid deficits and blood loss decreases cardiac output. SNS-mediated tachycardia, vasoconstriction, and venoconstriction compensate for a 15% to 20% deficit before hypotension occurs. In hypothermic patients, rewarming often unmasks hypovolemia. A “normal” intravascular volume can be inadequate to maintain blood pressure (relative hypovolemia) if increased venous capacity (histamine release, sympathectomy), thoracic vein compression (positive pressure ventilation, tension pneumothorax), or pericardial tamponade impair venous return. Using urine output to assess intravascular volume can be misleading. Impaired renal tubular concentrating ability or glycosuria can maintain output despite hypovolemia.
Hypotension caused by ventricular dysfunction often indicates acute myocardial ischemia precipitated by either tachycardia or inadequate diastolic pressure. Hypoxemia or anemia will exacerbate ischemia. Chest pain might be masked by analgesia or confused with surgical pain or gastric distention. Ischemia is sometimes silent in patients with diabetic neuropathy. The ST segment and T-wave morphology on electrocardiogram should be evaluated, as well as pulmonary artery pressures and echocardiography if available. Some patients with nonischemic cardiomyopathy require high left ventricular end diastolic pressure and elevated SNS activity to maintain blood pressure. Right ventricle dysfunction caused by pulmonary thromboembolism often presents with hypotension.
Heart rate below 40 to 45 beats per minute (sinus or nodal bradycardia, complete heart block) decreases both cardiac output and blood pressure. A tachydysrhythmia above 140 to 150 beats per minute (bpm) can compromise ventricular filling, especially if atrial kick is absent. Changes in heart rate place patients with aortic or mitral stenosis at particular risk of hypotension. Decreased systemic vascular resistance from regional anesthesia, α-adrenergic blockade, warming, or sepsis may also generate hypotension. Rarely, hypotension reflects acute steroid deficiency.
Indications to treat hypotension include symptoms of vital organ hypoperfusion or a 20% to 30% reduction of blood pressure (BP) below preoperative levels. Tighter control of BP is indicated in high-risk patients. Always ensure that hypotension is real before treating. A BP cuff that is too large or a transducer that is improperly calibrated may yield false values. Before administering potent drugs to treat BP, consider using simple maneuvers (e.g., repositioning, reducing airway pressure) when appropriate. A 500-mL crystalloid infusion often improves hypotension in the PACU because hypovolemia is by far the most common etiology. Plasma expanders or blood products may be required in some circumstances. Administration of an α-adrenergic pressor (e.g., phenylephrine) may provide temporary support of BP until sufficient volume is infused. Treat myocardial ischemia with control of precipitating factors, support of diastolic pressure, β-adrenergic blocking agents, and nitrates. Bradycardia usually responds to atropine, glycopyrrolate, or ephedrine. Refractory bradycardia should be managed with epinephrine or with cardiac pacing.
VI. Postoperative Hypertension
Moderate hypertension in response to noxious stimuli (pain, surgical stress) is common in the PACU. High BP can increase blood loss, ventricular wall tension, and intraocular or ICP. Indications to treat hypertension include a BP 20% to 30% above preoperative baseline, an unusual risk of morbidity (e.g., increased ICP, mitral regurgitation, open eye injury), or evidence of complications (e.g., bleeding, headache, visual changes, ischemia). As in the case of hypotension, a BP cuff that is too small or an improperly calibrated transducer can yield erroneously high BP values. The treatment of hypertension in the PACU should be aimed at addressing causes of increased SNS activity. Common causes include pain, anxiety, or a full bladder. If hypertension persists, intravenous antihypertensive medications such as labetalol, esmolol, hydralazine, or nicardipine can achieve temporary control. Potent vasodilators such as nitroprusside or nitroglycerin should be reserved for refractory or profound hypertension.

Non-invasive measurement of BP requires a properly fitting cuff. A cuff that is too wide will yield falsely low values, and one that is too narrow will yield falsely high values.
VII. Inadequate Ventilation
Unfortunately, hypoventilation is common in the PACU. Frequent causes include residual effects of inhaled anesthetics or opioids, residual neuromuscular relaxation, pain, respiratory secretions, airway edema, hypothermia, and physical limitations to breathing associated with the surgical procedure (e.g., increased abdominal pressure).
Elevated exhaled carbon dioxide (CO2) or arterial partial pressure of CO2 (PaCO2) usually reflects hypoventilation (see “Acid–Base Disorders” below). Ventilation is likely to be inadequate if (a) hypercarbia occurs coincident with tachypnea, anxiety, dyspnea, or increased SNS activity; (b) if the arterial pH falls below 7.30; or (c) if there is a progressive decrease in arterial pH.
Hypoventilation can also result from airway obstruction, increased airway resistance, or decreased lung compliance. High resistance to gas flow increases the work of breathing and CO2 production. If inspiratory muscles cannot maintain sufficient ventilation, respiratory acidemia and hypoxemia occur. Increased upper airway resistance or obstruction can occur in the pharynx (posterior tongue displacement, edema, soft tissue collapse, secretions), the larynx (laryngospasm, edema), or the large airways (extrinsic compression, tracheal stenosis). Initial assessment and treatment of these problems should include arousal of the patient, lateral head positioning, chin lift, mandibular advancement, airway suctioning, or placement of an oropharyngeal or nasopharyngeal airway. Soft tissue edema may respond to nebulized racemic epinephrine. During emergence, pharyngeal or vocal cord stimulation can generate laryngospasm, especially in patients who smoke, have had recent upper respiratory infections, or have undergone upper airway surgery. Laryngospasm can usually be effectively treated with mandibular advancement and positive pressure ventilation with oxygen and a tight-fitting mask. Rarely, a small dose of succinylcholine (0.1 mg/kg) may be required. Pathologic upper airway obstruction, such as an impingement from an expanding hematoma, might require emergency decompression of the hematoma, tracheal intubation, or cricothyroidotomy. Pharyngeal or tracheal stimulation can trigger reflex bronchospasm in patients with reactive airways, as can histamine release. Flow might be so impeded that no wheezing is appreciated. To treat, administer albuterol via an inhaler. Some patients may require an anticholinergic medication such as ipratropium. If bronchospasm is life-threatening, administer an intravenous epinephrine infusion.
Low pulmonary compliance causes respiratory muscle fatigue, hypoventilation, and respiratory acidemia. Collapsed airspaces are difficult to re-expand. Excess lung water increases the lung’s inertia and elevates surface tension. Extrathoracic factors such as adipose tissue, tight dressings, intragastric gas, or high intra-abdominal pressure impair thoracic expansion. Placement of the patient in a semisitting position may be useful to improve compliance. Note that signs of increased airway resistance mimic those of decreased compliance. Spontaneously breathing patients exhibit labored ventilation, while mechanically ventilated patients exhibit high peak inspiratory pressures.
VIII. Neuromuscular Problems
Residual paralysis compromises ventilation, airway patency, and airway protection. Partial paralysis is also dangerous because a somnolent patient with mild stridor and shallow ventilation might be overlooked, promoting insidious hypoventilation or aspiration. Overdosing patients with nondepolarizing relaxants or incomplete reversal after surgery can lead to residual weakness in the PACU. Neuromuscular abnormalities (myasthenia gravis) or medications (antibiotics, furosemide, phenytoin) can prolong the action of relaxants. In addition to the use of a nerve stimulator, recovery from muscle relaxants in the PACU can be assessed by observing sustained supine head elevation, a vital capacity of 10 to 12 mL/kg, or an inspiratory pressure more negative than –25 cm H2O. Occasionally, painful chest expansion, thoracic restriction, low compliance, or hyperventilation will generate dyspnea, labored breathing, or rapid, shallow breathing that can mimic ventilatory insufficiency due to residual paralysis.
IX. Postoperative Hypoxemia
Arterial partial pressure of oxygen (PaO2) is the best indicator of pulmonary oxygen transfer. Pulse oximetry, although easy to measure, yields less information on the alveolar–arterial gradient. A PaO2 above 80 mm Hg (93% saturation) with an acceptable hemoglobin level ensures adequate oxygen content, but the lowest acceptable PaO2 varies among individuals. Elevating PaO2 above 110 mm Hg (100% saturation) offers little benefit because the additional oxygen dissolved in plasma is negligible. Elevating PaO2 to above normal levels can also mask dangerous hypoventilation. That is, PaCO2 may be quite high yet the patient will appear to be ventilating well because of a high arterial oxygen saturation. During mechanical ventilation, a PaO2 above 80 mm Hg with 0.4 fraction of inspired oxygen and 5 cm H2O continuous positive airway pressure usually predicts sustained oxygenation after extubation. Adequate PaO2 does not guarantee that cardiac output, arterial pressure, or distribution of blood flow will maintain oxygen delivery. Lactic acidemia best reflects inadequate tissue oxygenation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








