CHAPTER 56

THE IMMUNOCOMPROMISED PATIENT
Immunocompromised patients present a special challenge to the acute care surgeon. Although transplant recipients, human immunodeficiency virus (HIV)-infected individuals, and chemotherapy patients are the most poignant examples, much more common are the immunocompromised states imposed by advanced age, malnutrition, chronic disease, and critical illness. As modern medicine continues to improve outcomes for such patients, they will continue to be an increasingly important part of the population that may eventually require emergency surgery. Therefore, knowledge of the various immunocompromised states and their management is of considerable importance to the acute care surgeon.
THE IMMUNE SYSTEM
As our knowledge of the immune system has grown, it has become clear that it is an extremely complex system with multiple dimensions that we are only beginning to understand. A detailed description of the immune system and our current state of knowledge is beyond the scope of this chapter. However, some basic premises remain true even if the full knowledge of the mechanisms behind them is still forthcoming. The immune system can be divided into two broad categories: innate and adaptive immunity. The innate response is the initial inflammatory response to infection or injury and is designed to proceed in a nonspecific manner. The adaptive immune system is tailored to the inciting pathogen and results in immunologic memory. While it is useful to categorize the immune system in this way, it should be pointed out that the two systems are closely integrated and each enhances the efficacy of the other. For example, the complement system as well as the host of inflammatory cytokines (tumor necrosis factor-alpha (TNFα), interferon-gamma (INFγ), interleukin (IL)-1, IL-2, etc.) play roles in both systems and illustrate their interdependence. The cells important for innate immunity are neutrophils, macrophages, natural killer (NK) cells, and dendritic cells. Mast cells, basophils, and eosinophils play a role in innate immunity as well, although their actions are classically demonstrated by type I hypersensitivity reactions. Neutrophils and macrophages phagocytose pathogens and/or cells infected with pathogens, while NK cells attack host cells that have been infected. TNFa and IL-1, primarily produced by macrophages, are the major inflammatory cytokines, and are implicated in many of the manifestations of the systemic inflammatory response syndrome.
Dendritic cells, positioned in tissues that are exposed to the external environment, link the innate immune response to the adaptive immune response by participating as antigen presenting cells (APCs). Via the major histocompatibility complex (MHC) class II, APCs present exogenous antigens to CD4+ (helper) T lymphocytes, which subsequently secrete cytokines such as INFγ, IL-2, and IL-4, which increases the activity of macrophages, CD8+ (cytotoxic) T lymphocytes and B lymphocytes, respectively. Cytotoxic T lymphocytes recognize and target cells presenting endogenous antigen (such as viral proteins) via MHC class I present on all nucleated cells. B lymphocytes recognize antigens bound to cell surface antibodies and subsequently differentiate into plasma cells that secrete antigen-specific antibodies. Perhaps the most important distinction to be made between the innate and adaptive immune response is that the adaptive immune response results in immunologic memory. Circulating memory B lymphocytes that secrete antigen-specific antibodies as well as circulating antigen-specific helper and cytotoxic T lymphocytes manifest this memory. The presence of antigen-specific immune cells allows for a much more rapid and robust response to an antigen if it is encountered again.
PROFOUND IMMUNOCOMPROMISE VERSUS RELATIVE IMMUNOCOMPROMISE
Immunocompromised patients can generally be separated into two broad categories: those with profound and those with relative immunocompromise. The most prominent example of profound immunocompromise is post solid-organ transplant immunosuppression. However, bone marrow transplantation, immunosuppressive therapy for autoimmune disorders, cancer chemotherapy resulting in neutropenia, and HIV infection/acquired immunodeficiency syndrome (AIDS) are also common etiologies of profound immunocompromise. Furthermore, an increasing number of genetic disorders, referred to as primary immunodeficiency disorders have been and continue to be elucidated (Table 56.1). While the individual conditions in this category may be rare, the aggregate should not be considered so.1 Profoundly immunocompromised patients have a markedly increased susceptibility to infection as well as a considerable inability to combat infection once it is established. On the other hand, relative immunocompromise has many etiologies and is more difficult to quantify. Chronic illness such as diabetes mellitus, cancer, alcoholism, and drug abuse are associated with impaired innate and adaptive immunity. Advanced age and malnutrition, often synchronous with chronic disease, are also associated with impaired immune function and make up a growing portion of the general population. Additionally, intensive care units across the nation are filled with critically ill patients with immune dysfunction related to their acute illness. Similar to those with profound immunocompromise, these patients exhibit increased susceptibility to infection and demonstrate increased postoperative morbidity and mortality.
TABLE 56.1
WELL-CHARACTERIZED PRIMARY IMMUNODEFICIENCY DISORDERS
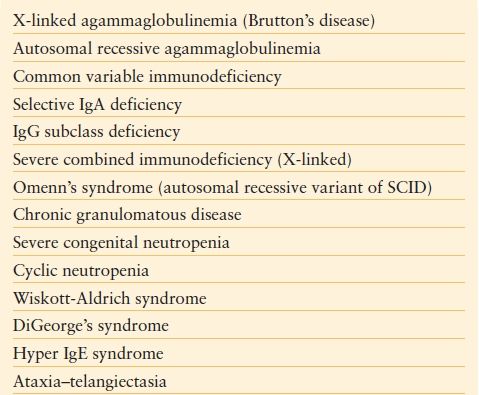
Adapted from Turvey SE, Bonilla FA, Junker AK. Primary immunodeficiency diseases: a practical guide for clinicians. Postgrad Med J. 2009;85: 660–666.
ORGAN TRANSPLANTATION
Over 20,000 organ transplants are performed annually in the United States (http://optn.transplant.hrsa.gov). The kidney is by far the most commonly transplanted organ followed by liver, heart, and lungs. A large part of the modern success of organ transplantation has come from the development of more effective immunosuppressive agents. As organ transplantation continues to improve and becomes an increasingly viable option for a myriad of disease processes, the population of transplant recipients is likely to increase. It is well established that the rate-limiting factor in transplantation is donor availability. This observation underscores the reality that the acute care surgeon should expect to encounter transplant recipients in his or her practice and be prepared to manage them appropriately. A basic working knowledge of immunosuppressive regimens is crucial to providing proper management of transplant patients. The field of immunosuppression has undergone many changes over recent decades driven by a much better understanding of the immune system. This knowledge has allowed for the development of targeted therapies with increased efficacy. Today, most patients will receive a combination of agents to prevent rejection. While traditional corticosteroid therapy remains an integral part of immunosuppression, modern therapy includes a multiplicity of agents including antimetabolites, calcineurin inhibitors, and lymphocyte-activation inhibitors. Other agents frequently used for induction and to combat rejection include antilymphocyte antibodies and IL-2 receptor antagonists. A summary of these agents with mechanisms of action and side effects is provided in Tables 56.2 and Tables 56.3. Corticosteroids are the most commonly encountered class of immunosuppressive agents, although arguably they are more frequently used outside of transplantation. Corticosteroids inhibit cytokine release and impair neutrophil chemotaxis, rendering patients susceptible to infection. In addition, fibroblast function, collagen production, and angiogenesis are impaired, which negatively impact wound healing. Animal data clearly demonstrate impaired wound healing with corticosteroid therapy and limited human studies have correlated increasing doses of corticosteroids with impaired wound healing and increased postoperative infectious complications.2,3 Unfortunately, patients receiving chronic corticosteroid therapy do not have the luxury of halting therapy as this places them at risk for adrenal insufficiency due to a depressed hypothalamic–pituitary–adrenocortical (HPA) axis. For this reason, supplemental corticosteroid therapy is commonly administered during periods of acute stress.
TABLE 56.2
IMMUNOSUPPRESSIVE AGENTS AND MECHANISM OF ACTION
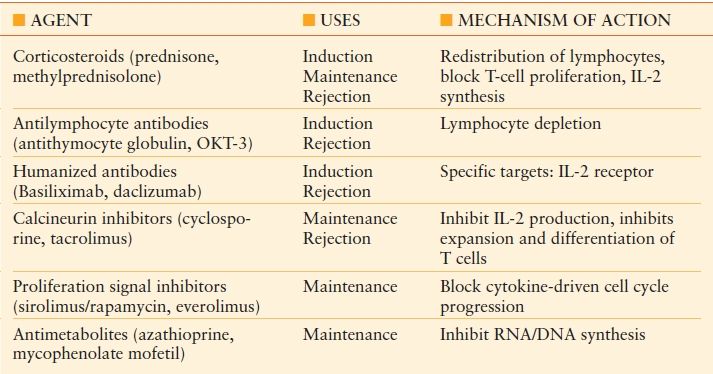
TABLE 56.3
SIDE EFFECTS OF COMMON IMMUNOSUPPRESSIVE AGENTS
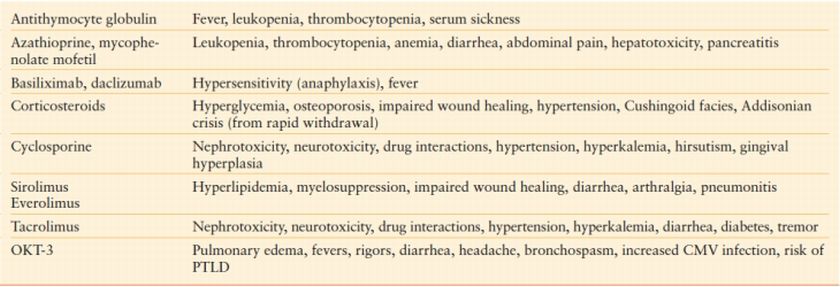
In effect, all immunosuppressive therapy places patients at risk for infectious complications. Up to 63% of solid-organ transplant recipients experience an infectious complication within the first year of transplant.4 The risk of infection is highest during the first 6–12 months, corresponding to the period of most intensive immunosuppression. The risk of infection also increases with treatment of rejection. The most frequent infections seen early and late after transplantation are presented in Table 56.4. Infectious complications in the first month after transplantation are frequently caused by the same organisms common to immunocompetent hosts. Thereafter, opportunistic infections predominate, correlating with the highest degree of immunosuppression. Included among the most common problems are viral infections such as cytomegalovirus (CMV) and opportunistic fungal infections such as Candida and Aspergillus species.5 A key component to treatment of infection in transplant patients is decreasing and/or ceasing immunosuppression, allowing the host immune system a chance to respond and increasing the prospects of clearing the infection. In the balance, of course, remains the risk of developing rejection in the transplanted organ. Consequently, close collaboration with the transplant team is required to appropriately manage immunosuppressive therapy and to prevent and/or detect signs of rejection.
TABLE 56.4
RISK OF INFECTION AFTER TRANSPLANT WITH RESPECT TO TIME AFTER TRANSPLANT
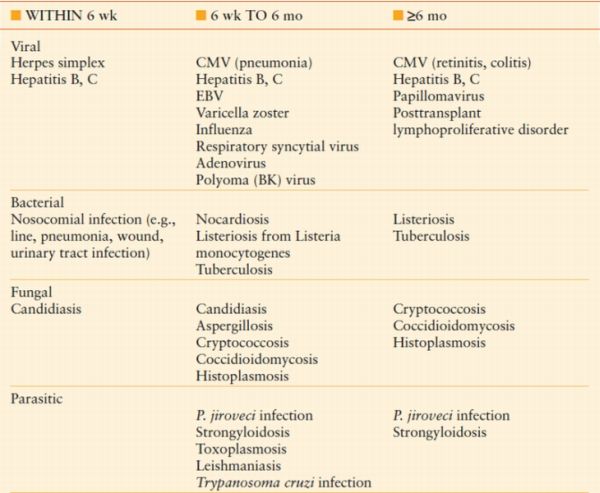
Modified from Snydman DR. Epid 2001;33 (suppl 1):S5–S8. emiology of infections after solid-or gan transplantation. Clin Infect Dis. 2001;33 (suppl 1):S5–S8.
AUTOIMMUNE DISORDERS
A multitude of chronic inflammatory and autoimmune disorders require anti-inflammatory, immunomodulating, and occasionally, immunosuppressive therapy for adequate symptom and disease control. Well-known examples include rheumatoid arthritis, systemic lupus erythematosus, and inflammatory bowel disease. Pharmacologic treatment ranges from nonsteroidal anti-inflammatory drugs (NSAIDs) and cyclooxygenase-2 (COX-2) inhibitors to corticosteroids, disease modifying antirheumatic drugs (DMARDs), and biologic response modifiers such as TNFα inhibitors (see Table 56.5).58,59 By nature of their varying mechanisms of action, these drugs contribute to problems with surgical wound healing and increase rates of postoperative infectious complications. However, cessation of therapy for surgical procedures often leads to disease flares and worsening symptoms, which can decrease functional status and retard postoperative recovery. This conundrum poses a challenge to physicians and patients alike when it comes to surgical decision making.
Unfortunately, surgical outcome data in patients with chronic inflammatory and autoimmune disorders receiving various pharmacotherapies is limited. Animal studies have clearly demonstrated that NSAIDs negatively impact wound healing. However, selective COX-2 inhibitors have not been shown to negatively impact wound healing in animal models.3 There is a paucity of human data for either class of drugs and current recommendations are based on extrapolations from the animal data. Although concerns exist regarding applicability of this data, it is generally recommended that NSAIDs be withheld for 3–4 days prior to surgery involving soft tissue and tendons. This recommendation has little applicability to the acute care surgeon, however.
There is also modest data regarding surgical outcomes and some of the more potent agents such as methotrexate, azathioprine, and other DMARDs as well as TNFα receptor inhibitors such as infliximab. Two small studies in the orthopedic literature, one retrospective review, and one prospective study, demonstrated that continuing methotrexate in rheumatoid arthritis patients in the perioperative setting is associated with increased infectious and wound healing complications. However, several other small studies, both retrospective and prospective, failed to demonstrate the same results, finding no significant difference in complication rates in patients who stopped methotrexate prior to surgery compared to those who continued methotrexate treatment perioperatively.3 Similarly, there is conflicting data regarding outcomes in patients with inflammatory bowel disease being treated with biologic agents who are undergoing surgery. Several small studies have demonstrated equivalent postoperative complication rates in patients being treated with infliximab and other immunosuppressive agents such as azathioprine and 6-mercaptopurine for Crohn’s disease and chronic ulcerative colitis compared to those who either had their medications discontinued before surgery or were not being treated with these agents.6–9 However, multiple additional studies have noted an increased postoperative infectious complication rate in patients receiving infliximab preoperatively for treatment of Crohn’s disease and ulcerative colitis.10–13 One particular study noted that the risk of postoperative infectious complications, most notably sepsis, was mitigated by the presence of a diverting stoma in patients undergoing ileocolonic resection for Crohn’s disease.10 As a whole, these studies are small, underpowered, and possess confounding variables such as concomitant use of corticosteroids and other immunosuppressive medications. Though the authors generally attempt to compensate with multivariate analyses, the small study sizes of current reports do raise concerns about adequately detecting differences.
TABLE 56.5
DMARDS AND BIOLOGIC RESPONSE MODIFIERS USED IN TREATMENT OF AUTOIMMUNE AND OTHER INFLAMMATORY DISORDERS
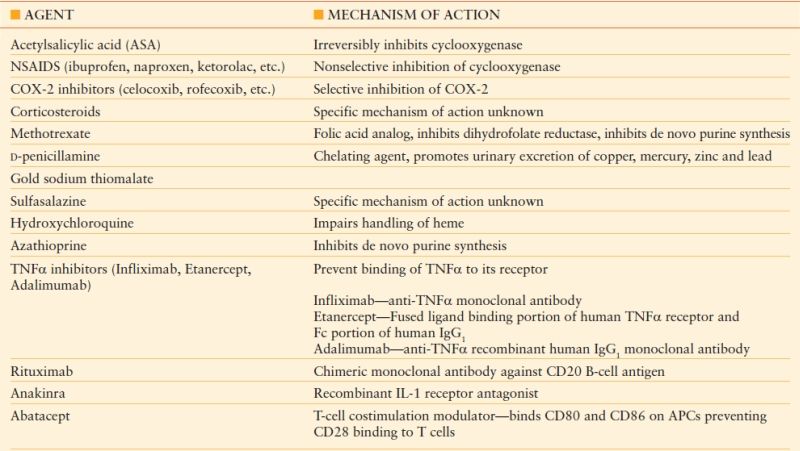
Data from Brunton LL, Lazo JS, Parker KL, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 11th ed. New York: McGraw Hill; 2006; Katzung BG, Masters SB, AJ Trevor, eds. Basic and Clinical Pharmacology. 11th ed. New York: McGraw Hill; 2009.
Despite its deficiencies, it is this data that is extrapolated to provide recommendations for other clinical scenarios. Obviously, the luxury of discontinuing immunomodulating therapy preoperatively is not typically available to acute care surgeons. The problem that most surgeons will face will revolve around the potential increased risk of postoperative infectious and wound healing complications as well as perioperative management of the primary disease state for which anti-inflammatory and immunomodulating therapy was intended. Clear guidelines are lacking given the current state of knowledge. In elective surgery, the recommended approach is to thoroughly assess risk factors and individually weigh risk versus benefit.3,14 In some semielective instances that are encountered in acute care surgery, this approach is reasonable. In those patients in whom therapy is continued during the perioperative period, caution and vigilance is advised to detect postoperative complications early and initiate appropriate treatment in a timely fashion. When the risks clearly outweigh the benefits (e.g., intra-abdominal catastrophe), immunomodulatory therapy should be withheld until infectious concerns have subsided.
NEUTROPENIA
The most significant problem that acute care surgeons encounter in patients receiving chemotherapy is neutropenia. Defined as an absolute neutrophil count (ANC) of <1,500 cells per mL, neutropenia significantly inhibits the ability to combat bacterial infection. Moderate (ANC 500–1,000 cells per mL) and severe (ANC < 500 cells per mL) neutropenia impart the greatest risk compared to milder degrees of neutropenia. While chemotherapy is a common etiology, radiotherapy, leukemias, myelodysplastic disorders, and severe viral infections can also cause neutropenia. Neutropenic patients may experience fever and can develop life-threatening sepsis from transient bacteremia that would otherwise be clinically silent in an immunocompetent host. Common sources include the skin and oral cavity. The vast majority of infectious problems can be resolved with antimicrobial agents and cessation of the inciting agent until neutrophil counts increase. Often, granulocyte colony stimulating factor (G-CSF) has been administered to help stimulate a rise in neutrophil counts. Recently, the American Society of Clinical Oncology Update committee expanded recommendations for the use of colony stimulating factors (CSFs) to include prevention of febrile neutropenia in high-risk patients and to allow a modest to moderate increase in dose density and/or intensity of chemotherapy regimens.15 While this decision-making process will likely fall to oncologists and hematologists, surgeons should be aware of the potential for use of CSFs in the perioperative management of neutropenic patients. Typically, surgeons become involved in the management of these patients when a gastrointestinal source is suspected for neutropenic fever or when patients complain of abdominal pain. The differential diagnosis can range from common entities such as calculous cholecystitis, peptic ulcer disease, appendicitis, diverticulitis, and bowel obstruction to less common conditions such as neutropenic enterocolitis, spontaneous intestinal perforation, and acalculous cholecystitis.
HUMAN IMMUNODEFICIENCY VIRUS/ACQUIRED IMMUNODEFICIENCY SYNDROME
HIV is a blood borne retrovirus that targets the CD4 lymphocytes of the host. It uses reverse transcriptase to translate its viral RNA into DNA in the host cell, which thereafter enables the cell to manufacture millions of viral particles. These particles are released systemically after lysis of the host cell and are free to target other CD4 lymphocytes. The end result is massive depletion of CD4 lymphocytes and subsequent host immunosuppression, referred to as AIDS. Modern antiretroviral drug therapy has improved outcomes and prolonged survival in patients with HIV infection; however, a significant portion will go on to develop AIDS, the most advanced stage of HIV infection.16 Clinically, this corresponds with a state of profound immunosuppression and is defined by the development of an AIDS-defining illness or when CD4 counts drop to <200 cells per mm3 (Tables 56.6 and Tables 56.7).16
According to Center for Disease Control estimates, more than 1 million people in the United States are infected with HIV, one-fifth of which are unaware of their infection. Each year in the United States, an estimated 56,300 new HIV infections occur and approximately 36,000 people are newly diagnosed with AIDS. In 2007, an estimated 455,636 people were living with AIDS in the United States (http://www.cdc.gov). While the numbers are striking, they pale in comparison to the global crisis affecting millions of people.
A recent review highlighted the current state of surgical care in HIV/AIDS patients.17 The advent of antiretroviral drug therapy has improved the overall outcomes for these patients and has concomitantly decreased the number of operations for AIDS-related surgical illnesses. However, these patients will continue to require surgical intervention for a number of intra-abdominal problems. The authors highlight that although AIDS-related conditions (CMV gastroenteritis, typhlitis, GI lymphoma and Kaposi sarcoma, mycobacterial infection, etc.) should remain in the differential diagnosis in HIV patients undergoing evaluation for abdominal pain, illnesses common in non-HIV patients and unrelated to the immunocompromised state (appendicitis, diverticulitis, peptic ulcer disease, etc.) should also be considered and are often the most likely etiology of the patient’s complaints. As with any immunocompromised patient, the physical and laboratory findings may be blunted in AIDS patients. Typical findings such as peritonitis and leukocytosis may be diminished or absent. Occasionally, CD4 counts are absent or outdated and may not be available in the acute situation to impact clinical decision making. In this instance, total lymphocyte count (TLC) has been shown to be a reasonable predictor of absolute CD4+ count (specificity 97% for CD4+ count < 200 per µL when TLC <1.0 × 109 per L) and may be useful in the acute setting to gauge the degree of immunosuppression.18 This is typically used as prognostic information as low CD4+ counts have been correlated with increased postoperative morbidity and mortality.19,20 However, recent reviews have not found a correlation with low CD4+ counts and adverse postoperative outcomes although an increase in postoperative pneumonia and overall 12-month mortality was noted in a cohort of HIV+ patients, irrespective of CD4+ count.21,22 In the trauma literature, it has been recently demonstrated that no major difference in mortality exists for patients with HIV, although this patient population had a higher risk of infectious complications and a longer hospital length of stay.23
TABLE 56.6
AIDS-DEFINING CONDITIONS

aFrom Centers for Disease Control. 1993 Revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Morb Mortal Wkly Rep. 1992;41:1–19.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








