Chapter 37 The Difficult Airway in Obstetric Anesthesia
III. Anesthesia-Related Maternal Mortality
IV. Obstetric Anesthesia Closed Claims Studies
V. Why Is Advanced Airway Management Important?
VII. Incidence of Difficult or Failed Intubation in Obstetrics
VIII. Prediction of Difficult Airway Management
IX. Recommendations for Management of the Difficult Airway
X. Equipment (Difficult Airway Cart)
XI. Anticipated Difficult Intubation
XII. Airway Management Strategies
XIII. Unanticipated Difficult Tracheal Intubation
XIV. Communication and Documentation
I Introduction
Difficult laryngoscopy, failed intubation, and inability to ventilate or oxygenate after induction of general anesthesia (GA) for cesarean delivery (C/D) are major contributory factors leading to maternal morbidity and mortality. In Western countries, the recognition of adverse maternal and neonatal outcomes associated with difficult airway (DA) management has led to a dramatic decline in the use of GA for both elective and emergency C/D.1–3
The overall general anesthesiology practice in industrialized countries in the last 2 decades has undergone a dramatic change. Management of the DA has emerged as one of the most important patient safety issues. Guidelines and strategies for management of the DA have been published by the American Society of Anesthesiologists (ASA),4 the Difficult Airway Society (DAS) in the United Kingdom,5 and the Canadian,6 French7 and Italian8 national societies’ work groups on difficult airway management. These guidelines are applicable to the general surgical population. However, none of the guidelines address management of the DA in an obstetric situation, especially in the context of urgency in delivering the baby.
There are differences among these standards, guidelines, algorithms, recommendations, and protocols, but in practice and in medicolegal cases, the distinction between terminologies can be blurred (Table 37-1).9 Published literature shows that the guidelines from the ASA Task Force on Management of the Difficult Airway have been discussed in 18% of nonobstetric medicolegal cases and were useful both in defending care (defense, 8%) and in criticizing care (plaintiff, 3%).10 Expert witnesses also have used these guidelines in litigated obstetric DA management cases.
TABLE 37-1 Definition of Terms and Degree of Obligation
| Terms | Definitions | Degree of Obligation |
|---|---|---|
| Standards | Generally accepted principles for patient management; exceptions are rare, and failure to follow is often difficult to justify | Mandatory |
| Strategy | A well-planned series of steps for achieving a goal | Voluntary |
| Guidelines | Systematically developed statements to assist practitioners for specific clinical circumstances; incorporates the best scientific evidence with expert opinion | Voluntary |
| Practice policies | Describe present recommendations issued to influence practitioners in reaching decisions about interventions | Voluntary |
| Recommendations | Suitable and useful strategy; not as strict as standards or guidelines | Voluntary |
| Options | Different possibilities are available; neutral assessment | Voluntary |
| Protocols and algorithms | Stepwise procedures or decision trees to guide practitioners through diagnosis and treatment of various clinical problems | Voluntary |
From Henderson JJ, Popat MT, Latto IP, Pearce AC: Difficult Airway Society guidelines for management of the unanticipated difficult intubation. Anaesthesia 59:675–694, 2004.
II Definitions
There is no consensus on a standard definition of the DA in the literature. The ASA Task Force defined DA as a clinical situation in which a conventionally trained anesthesiologist experiences difficulty with face mask ventilation, difficulty with intubation, or both. The original ASA description of difficult intubation (DI) included a limit of 10 minutes or multiple attempts. The wisdom of this definition must be questioned in obstetrics especially, given the fact that GA is generally reserved for emergency C/D in which delivery of the baby is of the utmost urgency. The common practice in obstetric anesthesia is to use a single dose of succinylcholine when anesthetizing for C/D under GA. In the obstetric situation, difficult intubation is the inability of an experienced anesthesiologist to intubate within one dose of succinylcholine.11 A more apt definition of failed intubation in obstetric patients is inability to secure the airway with two attempts using a conventional laryngoscope or an alternative airway device to assist with tracheal intubation.
III Anesthesia-Related Maternal Mortality
Although the total number of maternal deaths has been decreasing steadily in the last few decades both in the United States and United Kingdom (Table 37-2),12,13 anesthesia-related complications rank seventh among the leading causes of maternal death, in both countries (Fig. 37-1).14,15 Even in the developing countries, anesthesia is emerging as an additional risk for maternal mortality and remains largely under-reported.16,17 Failures in airway management are a primary cause of anesthesia-related maternal deaths in the underdeveloped countries.18
TABLE 37-2 Anesthesia-Related Maternal Deaths in the United States and United Kingdom, 1979-2001
| Year of Death | United States* | United Kingdom† |
|---|---|---|
| 1979-1981 | 4.3 | 8.7 |
| 1982-1984 | 3.3 | 7.2 |
| 1985-1987 | 2.3 | 1.9 |
| 1988-1990 | 1.7 | 1.7 |
| 1991-1993 | 1.4 | 3.5 |
| 1994-1996 | 1.1 | 0.5 |
| 1997-1999 | 1.2 | 1.4 |
| 2000-2002 | 1.0 | 3.0 |
* Maternal deaths per million live births.
† Maternal deaths per million maternities (live births, stillbirths, pregnancy terminations, ectopic pregnancies, and abortions).
(From Hawkins JL, Chang J, Palmer SK, et al: Anesthesia-related maternal mortality in the United States: 1979-2002. Obstet Gynecol 117:71, 2011.)
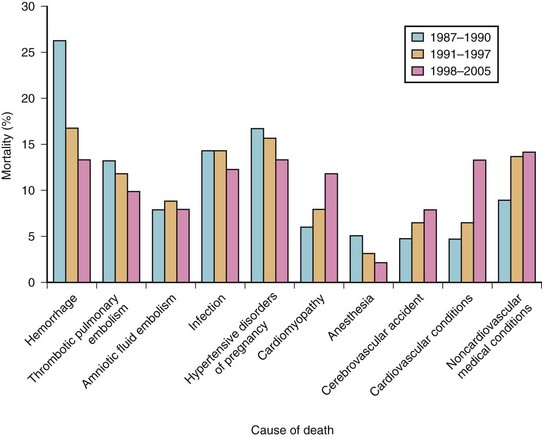
Figure 37-1 Maternal mortality in the United States.
(From Berg CJ, Callaghan WM, Syverson C, Henderson Z: Pregnancy-related mortality in the United States, 1998 to 2005. Obstet Gynecol 116:1302–1309, 2010.)
The first national study of anesthesia-related maternal mortality in the United States between 1979 and 1990 was published in 1997.19 Most of these deaths (82%) took place during C/D. Death rates for GA during C/D increased from 20 per million in 1979-1984 to 32.3 per million in 1985-1990. Conversely, the death rate for regional anesthesia (RA) during the same periods declined from 8.6 to 1.9 per million, respectively. The relative risk for GA increased to 16.7 from 1985 to 1990. The case fatality risk ratio for GA was. 2.3 times that of RA from 1979 to 1984 and increased to 16.7 times from 1985 to 1990.19 The majority of maternal deaths were related to difficult or failed intubation, pulmonary aspiration, or respiratory complications.
A follow-up study examined 12 years of anesthesia-related maternal deaths between 1991 and 2002 and compared them with data from 1979 to 1990 to estimate trends over time and to compare the risks of GA and RA during cesarean delivery.13 Results showed that 86 pregnancy-related deaths were associated with complications of anesthesia, accounting for 1.6% of the total. Case-fatality rates for GA declined from 16.8 per million in 1991-1996 to 6.5 per million in 1997-2002 (Table 37-3).
TABLE 37-3 Case-Fatality Rates and Rate Ratios of Anesthesia-Related Deaths during Cesarean Delivery by Type of Anesthesia, United States, 1979-2002
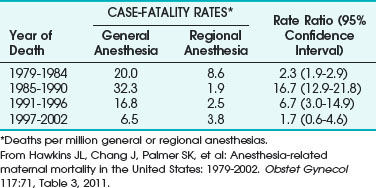
Complications related to anesthesia still occur, despite the decrease of almost 60% in anesthesia-related maternal mortality between 1979-1990 and 1991-2002. Although deaths from GA during C/D declined, about two thirds of the anesthesia-related deaths were caused by intubation failure or induction problems.13 The decline in GA-related case fatalities was explained by improvements in management of the DA and failed intubation and by increased expertise of anesthesiologists with the laryngeal mask airway (LMA) and other airway devices.13
A review of maternal mortality in Michigan from1985 to 2003 identified eight anesthesia-related deaths.20 Seven of these deaths were due to anesthesia-related factors. Interestingly, there were no deaths during induction of GA. The deaths were caused by airway obstruction or hypoventilation during emergence, lapses in monitoring, or lack of supervision in the postoperative period. Other risk factors included African-American race and obesity. This study highlighted the importance of airway-related problems during emergence, particularly in obese patients and in the African-American population, as well as the importance of vigilance in monitoring and management in the postoperative period for prevention of airway-related complications.
In the United Kingdom, despite the decline in the total number of maternal deaths, anesthesia-related causes consistently accounted for approximately 10% of the total direct deaths. During the period 1982-1984, anesthesia was the third leading cause of maternal death, resulting in 19 of 243 deaths, of which 15 were due to airway-related difficulties.21 The confidential enquiry spanning 1994-1996 showed that anesthesia was responsible for only 1 of 268 maternal deaths. In the Confidential Enquiry into Maternal and Child Health (CEMACH) 2000-2002 study, there were six direct deaths, all related to GA.15 Maternal deaths from complications of GA included a risk of 1 maternal death in 20,000. These cardiopulmonary arrests and deaths were related to difficult or failed intubation, difficult pulmonary ventilation resulting in failure to oxygenate, pulmonary aspiration, or acute respiratory distress syndrome (ARDS). In all of these cases, the anesthesia care was considered substandard.15
IV Obstetric Anesthesia Closed Claims Studies
The 2009 closed claims study published in the United States revealed that obstetric anesthesia claims for injuries from 1990 to 2003 had declined compared with claims for injuries before 1990.1 In the period 1990-2003, the proportion of maternal death or brain damage and that of newborn death or brain damage decreased. Respiratory causes of injuries also decreased, from 24% to 4% in claims from 1990 or later. Claims related to inadequate oxygenation or ventilation and those related to aspiration also decreased.
The number of claims related to difficult tracheal intubation after 1990, compared with pre-1990 claims, was unchanged.1 However, the overall improvement in closed claims statistics and the decline in anesthesia-related maternal mortality in more recent years were probably attributable to implementation of a minimum standard of care requiring the use of respiratory system monitors (pulse oximetry and capnography) during anesthesia in modern practice, enhanced awareness of the risk of pulmonary aspiration of gastric contents in the obstetric patient, decreased use of GA in obstetric practice, and use of advanced airway management techniques.
V Why Is Advanced Airway Management Important?
There have been tremendous advances in airway management in recent years, including an explosion in the use of airway devices as adjuncts to airway management. There has been a vast increase in the body of knowledge and in publications worldwide in this field. Improvements in advanced airway management have led to a documented decline in the incidence of airway-related perioperative morbidity in the surgical population.22 Similarly, in the last 2 decades, anesthesiologists’ focus on improving management of the DA and of failed intubation, experience with the LMA, application of the ASA difficult airway algorithm (DAA), and advanced airway management strategies have helped decrease GA-related case fatalities in obstetric cases.13
Because the medical-legal liability associated with airway-related adverse maternal outcomes is high,1 it is essential that all those practicing obstetric anesthesia as part of airway management should do the following:
1. Assess risk factors that predispose to airway-related complications
2. Be cognizant of predictors of the DA
3. Have appropriate algorithms and airway devices and equipment immediately available in the labor and delivery suite and in the operating room to manage the DA
4. Have an airway rescue plan, within the framework of the ASA DAA, for managing the DA
5. Be cognizant of the twin goals of urgently delivering the baby and preventing pulmonary aspiration, which can be hard to balance
VI Risk Assessment
A Airway
Airway changes occur during pregnancy, labor, and delivery.23 Further, the incidence of Mallampati (MP) class III and class IV scores increases during labor compared with the prelabor period, and these changes are not reversed by 48 hours after delivery. Therefore, it is absolutely necessary to examine the airway of a parturient in labor before administering anesthesia for a cesarean delivery.24
An increase in the ground substance of the airway connective tissue, caused by elevated levels of estrogen during pregnancy, an increase in total body water, and an increase in interstitial fluid and blood volume, results in hypervascularity and edema of oropharynx, nasopharynx, and respiratory tract. Excessive weight gain during pregnancy, preeclampsia, iatrogenic fluid overload, excessive bearing-down efforts during labor, and increase in venous pressure all lead to increased upper airway mucosal edema. Additional upper airway changes include tongue engorgement during pregnancy, which leads to decreased mobility of the floor of the mouth and changes in the MP score.25 Several published reports have described development of airway edema during labor and delivery, in preeclampsia, and after massive fluid and blood transfusion resuscitation following postpartum hemorrhage.26–30 In some of these reports, the associated difficulties in tracheal intubation were secondary to changes in the MP score.
Because of the increased vascularity and engorgement of the mucosa, the parturient is at increased risk for epistaxis after manipulation of the nasopharynx with nasotracheal intubation and for swelling of the airway and is vulnerable to increased trauma with repeated attempts at intubation.31 Avoidance of manipulation of the nasopharynx, use of smaller-sized tracheal tubes, and strict adherence to no more than two attempts at tracheal intubation32 are vital measures to avoid airway-related trauma, complications, and catastrophes.
D Gastrointestinal Changes
Other risk factors include pregnancy-related hormonal, anatomic, and physiologic gastrointestinal changes that place the parturient at risk for gastric regurgitation and pulmonary aspiration during GA. The gravid uterus shifts the stomach cephalad and changes the angle of the gastroesophageal junction, resulting in incompetence of the gastroesophageal pinchcock mechanism. The lower esophageal tone is decreased, leading to increased gastric reflux. There is a progesterone-mediated smooth muscle relaxant effect on the gastrointestinal mucosa. Furthermore, gastric emptying is prolonged during labor. Therefore, the parturient is at risk for gastric regurgitation, active vomiting, and pulmonary aspiration during GA, in case of a DA, or during failed intubation. Aspiration-related deaths during pregnancy occur from complications associated with induction problems such as DI, esophageal intubation, and inadequate attempts at ventilation.12
E Obesity
Weight gain during pregnancy results from the increasing size of the uterus and fetus, increased blood and interstitial fluid volumes, and deposition of new fat. There is a correlation between weight gain and an increase in the MP score.30 Pregnancy also results in a significant increase in breast size and engorgement. In the supine position, the enlarged breasts can encroach into the neck area, impeding effective application of cricoid pressure and leading to difficulty with laryngoscope blade insertion.
Obesity has reached epidemic proportions in the United States, and the incidence of obesity in pregnancy has doubled in the last 10 years. A body mass index (BMI) greater than 25 kg/m2 is considered overweight, and a BMI greater than 30 kg/m2 is considered obese.33 In the nonobstetric population, a BMI greater than 26 kg/m2 results in a threefold increased incidence of DMV and a 10-fold increased incidence of difficult tracheal intubation.34,35 Both prepregnancy obesity and excessive weight gain during pregnancy are associated with comorbidities such as hypertension or preeclampsia with intrauterine growth retardation, diabetes and macrosomia, and dysfunctional labor, thus increasing the incidence of operative cesarean delivery. The incidence of postpartum hemorrhage is higher in these patients, leading to increased likelihood of a GA intervention.
Because weight gain and obesity are associated with an increase in the MP score, the incidence of a partially obliterated oropharyngeal space in an obese parturient is doubled compared with that in nonpregnant patients. The aforementioned changes, the breast engorgement, and anthropometric differences between patients create a risk for difficult laryngoscopy, DI, and DMV.30,36,37 DI is encountered more frequently in morbidly obese parturients weighing more than 130 kg.38 Mask ventilation tends to be difficult because of low chest wall compliance and increased intra-abdominal pressure. In obesity, the respiratory-related changes of pregnancy are even more significant, with a marked decrease in FRC such that the closing capacity exceeds the FRC during tidal breathing, leading to a decrease in arterial oxygen tension and predisposing the parturient to a much higher risk of hypoxemia during a DI or DMV encounter. Obesity compounds all the risk factors associated with normal pregnancy, including DA, difficult laryngoscopy, DI, DMV, and aspiration-related complications.
VII Incidence of Difficult OR Failed Intubation IN Obstetrics
In 1998, the incidence of difficult laryngoscopy or tracheal intubation in the nonobstetric population was 0.1% to 13%.39 In the obstetric population, the incidence of difficult tracheal intubation was found to be 1/25040 to 1/280,41 1/300,42 and 1/750.36
A review of GA for cesarean deliveries from 1990 to 1995 showed the incidence of difficult tracheal intubation to range from a high of 16.3% to a low of 1.3%.2 There was a sentinel CICV incidence of 1 in 536 cases; in this patient, multiple attempts at intubation, unsuccessful mask ventilation, failed Combitube placement, and unsuccessful cricothyroidotomy resulted in cardiopulmonary arrest followed by surgical tracheostomy. Resuscitation was accomplished, but the mother remained in a coma until death, and the baby suffered significant neurologic injury.
In a review of GA for cesarean deliveries from 2000 to 2005, the incidence of CICV was 1 in 98. The single case of CICV occurred after failed tracheal intubation, unsuccessful LMA placement, and hypoxemia; successful cricothyroidotomy resulted in a good outcome for both mother and baby.43
VIII Prediction of Difficult Airway Management
The ASA DAA recommended an airway-related history to detect medical, surgical, and anesthetic factors that might indicate the presence of a DA. The guidelines also recommended an airway physical examination using assessment of multiple airway features before initiation of anesthetic care and airway management in all patients.4 The ASA closed claims analysis (2005) showed that 8% of patients did not have a preoperative history or airway physical examination.10
A retrospective audit was performed of all obstetric GAs, a total of 3430 cases over an 8-year period.44 None of the patients had a failed or esophageal intubation. There were 23 DIs, an incidence of 1:156. Anticipated difficult tracheal intubation occurred in 9 patients, 3 of whom underwent awake fiberoptic intubation; in the remaining 6 patients, who were morbidly obese, the DI was managed by senior trainees or consultants. Unanticipated difficulties occurred in 14 patients (61%). The preoperative assessment was found to be inadequate in these cases, being either not recorded (6 cases) or poorly documented (8 cases).
A Preoperative Assessment
1 History and Evaluation
According to the ASA Task Force, an airway history and a focused review of medical records must be conducted when feasible.4 Obviously, airway evaluation and prediction of DA in an obstetric patient starts with a thorough, focused airway-related history and evaluation of specific airway-related examination findings.45 A thorough history addresses any difficulty with previous GAs, obstructive sleep apnea (OSA) or snoring, head and neck abnormalities, and diseases that might impair the airway and result in difficult tracheal intubation. A history of DA management should be considered a strong predictor of problems unless the history was related to a specific reversible disease process such as a dental abscess. The history may be available from verbal recollections by the patient, previous anesthetic records, hospital notes, a letter describing DA management, or a Medic-Alert bracelet. The introduction of anesthesia information management systems and mandatory electronic medical records should be extremely useful in the future as “airway alerts” are built in to notify future practitioners of the specific details encountered with DA management in a particular patient.
2 Predictors of Difficult Airway
Yentis described the problems with many studies examining the prediction of DA.46 It is appropriate here to delineate the terms used to describe the accuracy and predictive power of these tests. A test to predict DI should have high sensitivity, so that it will identify most of the patients in whom intubation will be truly difficult. It should also have a high positive predictive value (PPV), so that only a few patients with airways actually easy to intubate will be subjected to the protocol for DA management. The test should also have a high specificity, so that it will identify most patients in whom tracheal intubation will be truly easy (Table 37-4).46 Excellent interobserver reliability is essential for any test to have high specificity, sensitivity, and PPV in predicting a DA.
TABLE 37-4 Statistical Terminology and Calculation*
| Term | Calculation |
|---|---|
| True positive (TP) | Difficult intubation that had been predicted to be difficult |
| False positive (FP) | Easy intubation that had been predicted to be difficult |
| True negative (TN) | Easy intubation that had been predicted to be easy |
| False negative (FN) | Difficult intubation that had been predicted to be easy |
| Sensitivity | Percentage of correctly predicted difficult intubations as a proportion of all intubations that were truly difficult—that is, TP/(TP+FN) |
| Specificity | Percentage of correctly predicted easy intubations as a proportion of all intubations that were truly easy—that is, TN/(TN+FP) |
| Positive predictive value | Percentage of correctly predicted difficult intubations as a proportion of all predicted difficult intubations—that is, TP/(TP+FP) |
| Negative predicted value | Percentage of correctly predicted easy intubations as a proportion of all predicted easy intubations—that is, TN/(TN+FN) |
* The sensitivity, specificity, and positive predictive value of each test are calculated using the Cormack-Lehane score as the constant variable.
Adapted from Yentis SM: Predicting difficult intubation: Worthwhile exercise or pointless ritual? Anaesthesia 57:105-109, 2002.
B Quantitative Evaluation of Difficult Intubation
The LEMON mnemonic represents one such assessment that is simple and quick, can be performed on any emergency patient, and has proved to have high PPV.47 The LEMON mnemonic represents the following five elements for preanesthetic assessment (Box 37-1):
L:Look externally—The initial impression of potential airway difficulty is based on assessment for any obvious anatomic distortions or external features that may make intubation difficult, such as facial and periorbital edema in a preeclamptic patient.
E:Evaluate the 3-3-2 Rule—Measuring the geometry of the airway can predict the anesthesia practitioner’s ability to align the oral, pharyngeal, and tracheal axes.
M:Mallampati score—The degree to which the tongue obstructs the visualization of the posterior pharynx has some correlation with the ability to visualize the glottis. The MP score can be estimated by having the patient, in a sitting position, open the mouth fully and protrude the tongue as far as possible without phonation. The relationship of the base of the tongue to the oropharyngeal structures—uvula and tonsillar pillars and fauces—is assessed as follows:
O:Obstruction—Pathologic conditions such as edema, glottic tumor, lingular tonsil, hyperplasia, and trauma can cause obstruction and can make laryngoscopy and ventilation difficult.
N:Neck mobility—This is a vital requirement for successful intubation. The sniffing position is the optimal, classic position of the head and neck for facilitating intubation. The extension of the atlanto-occipital (A-O) joint on the cervical spine so as to be able to align the three axes (oral, pharyngeal, and laryngeal) during laryngoscopy enhances the ease of laryngoscopy and tracheal intubation. Normal A-O joint extension of the head over the neck is 35 degrees. It can be assessed easily by getting the patient to place the chin down on the chest and tilt the head backward as far as possible. A reduction in A-O joint extension of 12 degrees (33%) or more correlates with intubation difficulty; complete joint immobility significantly compromises the laryngeal view. Limited A-O joint extension is present in certain pathologic states such as spondylosis, rheumatoid arthritis, and cervical spine stenosis, resulting in symptoms of nerve compression with cervical extension. Complete A-O joint immobility (e.g., hard-collar neck immobilization) can compromise the view of the glottis during laryngoscopy.
Box 37-1
LEMON Airway Assessment Method
L—Look externally for anatomic features that may make intubation difficult
O—Obstruction: Examine for partial or complete upper airway obstruction
From Reed MJ, Dunn MJ, McKeown DW: Can an airway assessment score predict difficulty at intubation in the emergency department? Emerg Med J 22:99–102, 2005.
C Additional Tests
Difficult laryngoscopy was defined by the ASA Task Force as the inability to visualize any part of the vocal cords despite multiple attempts at conventional laryngoscopy.4 The original Cormack and Lehane classification of laryngoscopic views was used to describe the visibility of the glottis during laryngoscopy with a conventional laryngoscope and to predict the ease of intubation.48 The view at laryngoscopy was divided into four grades on the basis of a study in obstetric patients.49 The entire glottis is visible in a grade 1 view, whereas in a grade 2 view only the posterior portion of the glottis is visible; in a grade 3 view, only the epiglottis is seen, and in grade 4 view not even the epiglottis is seen. Grades 3 and 4 are considered to indicate DI.
1 Thyromental Distance
The thyromental distance (TMD) is defined as the distance from the chin (mentum) to the top of the notch of the thyroid cartilage with the head fully extended; it must be measured with a ruler for accuracy. The TMD gives an estimate of the mandibular space and helps in determining how readily the laryngeal axis will fall in line with the pharyngeal axis when the A-O joint is extended50:
• A TMD measurement of 6.5 cm or greater with no other abnormalities indicates the likelihood of easy intubation.
• A TMD measurement of 6.0 to 6.5 cm indicates that alignment of the pharyngeal and laryngeal axes will be challenging and that difficulty with laryngoscopy may result. However, intubation is possible with the use of adjuncts such as an Eschmann introducer or an optical stylet.
• A TMD measurement of less than 6 cm indicates difficult laryngoscopy; specifically, intubation may be impossible.
2 Sternomental Distance
The sternomental distance (SMD) is measured from the sternum to the tip of the mandible with the head fully extended and the mouth closed. The normal measurement is 13.5 cm. The SMD and the corresponding laryngoscopic view were documented in 523 parturients undergoing elective or emergency C/D under GA.51 An SMD of 13.5 cm or less had a sensitivity of 66.7%, a specificity of 71%, and a PPV of only 7.6%. Eighteen patients (3.5%) had a Cormack-Lehane grade 3 or 4 laryngoscopic view and were classified as having potentially difficult tracheal intubations. The SMD on its own as a sole indicator of DI was not useful, and the suggestion was to incorporate it with other tests in the preoperative airway examination.
3 Jaw Protrusion
Jaw protrusion (also termed prognathism or subluxation) is assessed by the mandibular protrusion test, which demonstrates the extent to which the lower incisors can be slid in front of the upper ones52:
• Class A: The lower incisors can be protruded anterior to the upper incisors.
• Class B: The lower incisors can be brought edge-to-edge with the upper incisors.
• Class C: The lower incisors cannot reach the top incisors.
4 Studies Assessing Predictors of Difficult Airway in Obstetrics
a Mallampati Classification
In obstetric patients, the MP score has been used as a single parameter to illustrate the dramatic airway changes that occur in pregnancy and to highlight the importance of preoperative assessment of the airway. Pilkington and coworkers (1995) evaluated the MP class at 12 weeks’ and 38 weeks’ gestation30; photographs taken at the two time periods demonstrated the increase in MP class in the same patient as gestation advanced. The MP score correlated with the increase in body weight, implying that oropharyngeal edema was responsible for the increase in the MP score.
b Multivariate Predictors
Rocke and colleagues (1992) were the first to use multivariate predictors to predict difficult tracheal intubation.36 They evaluated the MP classification as modified by Samsoon and Young, referring to it as the Modified Mallampati Test (MMT), along with other predictors in 3440 patients undergoing elective or emergency C/D under GA. Data were collected on 1606 patients, representing 46.7% of the obstetric surgical patients. Of the patients studied 1500 underwent general anesthesia. Other risk parameters for DA that they assessed included short neck, which equates with decreased A-O joint extension; receding mandible or decreased TMD (<3 finger breadths); and protruding maxillary incisors indicating significant overbite, which would equate to the current class C jaw protrusion or class III upper lip bite test.53
Rocke’s group made subjective assessments of the ease or difficulty of tracheal intubation according to the following scale (Table 37-5)36:
• Grade 1: Easy—intubation at first attempt with no difficulty
• Grade 2: Some difficulty—insertion of tracheal tube not achieved at first attempt; no difficulty but successful intubation after adjustment of laryngoscope blade and/or adjustment of head position, not requiring additional equipment, removal and reinsertion of the laryngoscope, or senior assistance
• Grade 3: Very difficult—requiring removal of the laryngoscope, further oxygenation by mask ventilation, and subsequent intubation with or without the use of airway adjuncts (e.g., Eschmann introducer, alternative laryngoscope blade) or intubation by a senior colleague
• Grade 4: Failed intubation—several attempts at intubation or unrecognized esophageal intubation by resident, followed by subsequent tracheal intubation by senior anesthesiologist
TABLE 37-5 Association between Oropharyngeal Structures Visualized Preoperatively and Subsequent Difficulty at Tracheal Intubation*
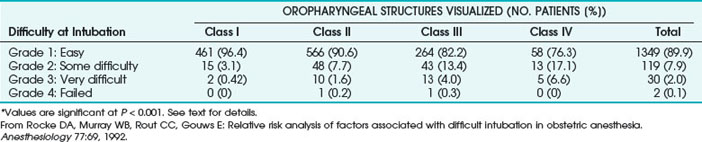
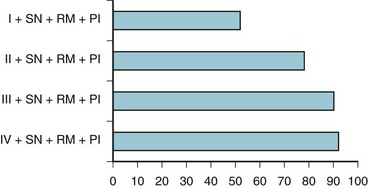
Based on these various parameters, the relative risk of experiencing difficult tracheal intubation (compared with an uncomplicated MMT class I airway) was determined as follows: 3.23 for MMT class II, 7.58 for MMT class III, 11.3 for MMT class IV, 5.01 for short neck, 9.71 for receding mandible, and 8.0 for protruding incisors. The investigators analyzed the univariate individual risk factor (i.e., MMT class) and combinations of the various risk factors and showed that a patient with an MMT III or IV classification plus protruding incisors, a short neck, and a receding mandible would have a probability of difficult laryngoscopy greater than 90% (Fig. 37-2). This study highlighted the importance of preoperative airway assessment to determine the best anesthetic intervention and the importance of prospectively preparing for airway interventions in the true obstetric emergency C/D under GA.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree