Minimally invasive surgery reduces surgical stress and postoperative pain as well as reduces overall morbidity, thus resulting in rapid recovery, earlier ambulation, shorter hospital stay, and rapid return to daily living activities.
 Robotic surgery offers technical advantages over laparoscopic surgery and thus should further expand the field of minimally invasive surgery.
Robotic surgery offers technical advantages over laparoscopic surgery and thus should further expand the field of minimally invasive surgery.
 Pneumoperitoneum creation and patient positioning during laparoscopic and robotic surgeries can cause significant physiologic changes, which may result in significant cardiorespiratory compromise, particularly in patients with significant cardiopulmonary dysfunctions.
Pneumoperitoneum creation and patient positioning during laparoscopic and robotic surgeries can cause significant physiologic changes, which may result in significant cardiorespiratory compromise, particularly in patients with significant cardiopulmonary dysfunctions.
 The cardiovascular changes during pneumoperitoneum include an increase in systemic vascular resistance and mean arterial pressure, cardiac arrhythmia (bradycardia and tachycardia), while changes in cardiac filling volumes and cardiac index vary depending on the patients’ status.
The cardiovascular changes during pneumoperitoneum include an increase in systemic vascular resistance and mean arterial pressure, cardiac arrhythmia (bradycardia and tachycardia), while changes in cardiac filling volumes and cardiac index vary depending on the patients’ status.
 The hemodynamic changes in the morbidly obese are similar to those observed in the nonobese.
The hemodynamic changes in the morbidly obese are similar to those observed in the nonobese.
 Pneumoperitoneum can reduce splanchnic and renal blood flows, while increasing intracranial pressures.
Pneumoperitoneum can reduce splanchnic and renal blood flows, while increasing intracranial pressures.
 Cephalad displacement of the diaphragm caused by increased intra-abdominal pressure and patient positioning, which can lead to reduction in functional residual capacity and total lung compliance, results in basal atelectasis and increased airway pressures.
Cephalad displacement of the diaphragm caused by increased intra-abdominal pressure and patient positioning, which can lead to reduction in functional residual capacity and total lung compliance, results in basal atelectasis and increased airway pressures.
 Carbon dioxide insufflation for creation of pneumoperitoneum increases carbon dioxide absorption, but the changes in arterial carbon dioxide concentrations remain clinically insignificant in healthy patients.
Carbon dioxide insufflation for creation of pneumoperitoneum increases carbon dioxide absorption, but the changes in arterial carbon dioxide concentrations remain clinically insignificant in healthy patients.
 Balanced general anesthesia with tracheal intubation and mechanical ventilation with acceptance of higher end-tidal carbon dioxide levels remains the best practice for minimally invasive surgical procedures.
Balanced general anesthesia with tracheal intubation and mechanical ventilation with acceptance of higher end-tidal carbon dioxide levels remains the best practice for minimally invasive surgical procedures.
 Aggressive multimodal analgesic as well as antiemetic prophylaxis and treatment are required to reduce these common postoperative adverse effects.
Aggressive multimodal analgesic as well as antiemetic prophylaxis and treatment are required to reduce these common postoperative adverse effects.
 Should intraoperative cardiopulmonary impairment occur, it is important to confirm that the intra-abdominal pressure is <15 mm Hg as well as rule out traumatic vascular injuries, carbon dioxide embolism, subcutaneous emphysema, capnothorax, and capnomediastinum.
Should intraoperative cardiopulmonary impairment occur, it is important to confirm that the intra-abdominal pressure is <15 mm Hg as well as rule out traumatic vascular injuries, carbon dioxide embolism, subcutaneous emphysema, capnothorax, and capnomediastinum.
 If subcutaneous emphysema extends to the chest and neck, the carbon dioxide can track to the thorax and mediastinum, thereby resulting in capnothorax or capnomediastinum.
If subcutaneous emphysema extends to the chest and neck, the carbon dioxide can track to the thorax and mediastinum, thereby resulting in capnothorax or capnomediastinum.
 Significant subcutaneous emphysema may lead to development of late hypercarbia (i.e., in the recovery room).
Significant subcutaneous emphysema may lead to development of late hypercarbia (i.e., in the recovery room).
 Hyperventilation and positive end-expiratory pressure should allow early resolution of capnothorax and carbon dioxide embolism.
Hyperventilation and positive end-expiratory pressure should allow early resolution of capnothorax and carbon dioxide embolism.
 If there is no improvement in the vital signs with routine management, it is imperative to release the pneumoperitoneum and place the patient in a supine (or Trendelenburg) position.
If there is no improvement in the vital signs with routine management, it is imperative to release the pneumoperitoneum and place the patient in a supine (or Trendelenburg) position.
Multimedia
 Laparoscopic
Laparoscopic
INTRODUCTION
 The development of “minimally invasive surgery” or “minimal access surgery” has revolutionized the field of surgery. The growth of laparoscopic surgical procedures is due to the use of smaller incisions that reduce surgical stress and postoperative pain as well as reduce overall morbidity, thus resulting in rapid recovery, earlier ambulation, shorter hospital stay, and rapid return to daily living activities (Table 43-1). These benefits are achieved without compromising surgical outcomes.1–6 In addition, a minimally invasive approach has allowed several procedures to be performed on an outpatient basis, where previously a hospital stay was necessary.7–11 Furthermore, it has allowed the implementation of rapid rehabilitation recovery programs that have been shown to further improve perioperative outcome and reduce hospital stay.12 Overall, there is potential for significant cost savings.
The development of “minimally invasive surgery” or “minimal access surgery” has revolutionized the field of surgery. The growth of laparoscopic surgical procedures is due to the use of smaller incisions that reduce surgical stress and postoperative pain as well as reduce overall morbidity, thus resulting in rapid recovery, earlier ambulation, shorter hospital stay, and rapid return to daily living activities (Table 43-1). These benefits are achieved without compromising surgical outcomes.1–6 In addition, a minimally invasive approach has allowed several procedures to be performed on an outpatient basis, where previously a hospital stay was necessary.7–11 Furthermore, it has allowed the implementation of rapid rehabilitation recovery programs that have been shown to further improve perioperative outcome and reduce hospital stay.12 Overall, there is potential for significant cost savings.
TABLE 43-1. ADVANTAGES OF MINIMALLY INVASIVE SURGERY

 With growing surgical expertise and continuing improvements in technology, more extensive laparoscopic procedures are being performed in a wide range of patients, including morbidly obese, older, and sicker patients with significant comorbidities as well as pregnant and pediatric patients.1 However, conventional laparoscopy has several technical limitations, such as reduced range of motion and instrument dexterity as well as a two-dimensional view of the operative field.6 Technologic advances in computer power and robotic engineering have allowed us to address some of these limitations. Robotic surgery improves depth perception through a high-definition, magnified, and three-dimensional view of the operative field and provides intuitive instrument control that mimics natural hand and wrist movements and eliminates surgeon tremor. Overall, robotic surgery enhances surgeons’ skills and thus has the potential to transform technically challenging open or laparoscopic procedures, particularly those requiring improved visualization and/or complex reconstruction necessitating extensive suturing, into technically feasible minimally invasive procedures. Robotic surgery can be applied to virtually every surgical subspecialty (e.g., general, colorectal, head and neck, gynecologic, thoracic, and cardiac surgeries). The advantages of robotic surgery are expected to broaden the application of the minimally invasive surgical paradigm.
With growing surgical expertise and continuing improvements in technology, more extensive laparoscopic procedures are being performed in a wide range of patients, including morbidly obese, older, and sicker patients with significant comorbidities as well as pregnant and pediatric patients.1 However, conventional laparoscopy has several technical limitations, such as reduced range of motion and instrument dexterity as well as a two-dimensional view of the operative field.6 Technologic advances in computer power and robotic engineering have allowed us to address some of these limitations. Robotic surgery improves depth perception through a high-definition, magnified, and three-dimensional view of the operative field and provides intuitive instrument control that mimics natural hand and wrist movements and eliminates surgeon tremor. Overall, robotic surgery enhances surgeons’ skills and thus has the potential to transform technically challenging open or laparoscopic procedures, particularly those requiring improved visualization and/or complex reconstruction necessitating extensive suturing, into technically feasible minimally invasive procedures. Robotic surgery can be applied to virtually every surgical subspecialty (e.g., general, colorectal, head and neck, gynecologic, thoracic, and cardiac surgeries). The advantages of robotic surgery are expected to broaden the application of the minimally invasive surgical paradigm.
 Despite the potential advantages of laparoscopic and robotic surgeries, they are associated with significant physiologic changes as well as new complications (some potentially life-threatening) that are usually not seen with the traditional open approach1 (Table 43-2). In addition, robotic procedures can have significantly prolonged operative times and require patients to be placed in extreme positions. Because the robotic system is large, it limits the access to the patient and invades the anesthesia working space. Overall, this presents significant challenges in anesthetic management. Therefore, a thorough understanding of potential physiologic changes and perioperative complications associated with laparoscopic and robotic surgeries is necessary to provide optimal patient care and improve perioperative morbidity and mortality.
Despite the potential advantages of laparoscopic and robotic surgeries, they are associated with significant physiologic changes as well as new complications (some potentially life-threatening) that are usually not seen with the traditional open approach1 (Table 43-2). In addition, robotic procedures can have significantly prolonged operative times and require patients to be placed in extreme positions. Because the robotic system is large, it limits the access to the patient and invades the anesthesia working space. Overall, this presents significant challenges in anesthetic management. Therefore, a thorough understanding of potential physiologic changes and perioperative complications associated with laparoscopic and robotic surgeries is necessary to provide optimal patient care and improve perioperative morbidity and mortality.
TABLE 43-2. POTENTIAL COMPLICATIONS DURING LAPAROSCOPY

This chapter discusses the anesthetic management of adult patients undergoing laparoscopic and abdominal robotic surgeries. However, endoscopic and robotic cardiac and thoracic surgeries (e.g., video-assisted thoracic surgery [VATS]) that are increasingly performed are discussed elsewhere (Chapter 37).
SURGICAL TECHNIQUES
 Laparoscopic procedure entails intraperitoneal insufflation of carbon dioxide (CO2) to create pneumoperitoneum that allows surgical exposure and manipulation. Carbon dioxide is used because it is noncombustible and more soluble in blood, which increases the safety margin and decreases the consequences of gas embolism. Unlike nitrous oxide (N2O), CO2 does not support combustion and, therefore, can be used safely with diathermy. Compared to helium, the high blood solubility of CO2 and its capability for pulmonary excretion reduce the risk of adverse outcomes in the event of gas embolism. An abdominal wall lift system (i.e., a gasless laparoscopy) has been developed to achieve surgical space while avoiding the cardiopulmonary effects of CO2 pneumoperitoneum.13 Although this approach appears to be safe, it has not been accepted in routine clinical practice because it increases operating times and surgical costs without improving clinical outcomes.13
Laparoscopic procedure entails intraperitoneal insufflation of carbon dioxide (CO2) to create pneumoperitoneum that allows surgical exposure and manipulation. Carbon dioxide is used because it is noncombustible and more soluble in blood, which increases the safety margin and decreases the consequences of gas embolism. Unlike nitrous oxide (N2O), CO2 does not support combustion and, therefore, can be used safely with diathermy. Compared to helium, the high blood solubility of CO2 and its capability for pulmonary excretion reduce the risk of adverse outcomes in the event of gas embolism. An abdominal wall lift system (i.e., a gasless laparoscopy) has been developed to achieve surgical space while avoiding the cardiopulmonary effects of CO2 pneumoperitoneum.13 Although this approach appears to be safe, it has not been accepted in routine clinical practice because it increases operating times and surgical costs without improving clinical outcomes.13
The initial access necessary for CO2 insufflation could be achieved either through a blind insertion of a Veress needle that consists of a blunt-tipped, spring-loaded inner stylet and sharp outer needle through a small subumbilical incision or a trocar inserted under direct vision. The open insertion of the trocar using a minilaparotomy approach guarantees safe creation of pneumoperitoneum and avoids the dangers of blind insertion.
Upon confirmation of appropriate placement, a variable flow electronic insufflator that automatically terminates gas flow at a preset intra-abdominal pressure (IAP) is used to achieve pneumoperitoneum. It is standard of care to maintain the IAP below 15 mm Hg, because higher pressures can have significant physiologic consequences and can increase the incidence of intraoperative complications. An access port is then inserted in place of the needle to maintain insufflation during the procedure. A video laparoscope, inserted through the port, allows visualization of the operative field. Additional access ports are inserted through a number of small skin incisions, which allow the introduction of surgical dissection and suction instruments. The secondary ports are placed under direct vision, preferably with the help of transillumination of the abdominal wall to identify superficial abdominal wall vessels.
The Da Vinci Surgical System (Intuitive Surgical Inc., Sunnyvale, California) is the most common robotic platform used currently. It consists of three components: a control console where the surgeon sits and operates the robotic arms and camera (Fig. 43-1), an equipment tower that includes an optical system, and the patient side cart that includes robotic arms.3,4,6 Similar to a laparoscopic procedure, robotic surgery involves development of pneumoperitoneum and placement of a video camera (a high-definition three-dimensional vision system) and ports. This is followed by placement of the robotic arms, a crucial and tedious part of the procedure (Fig. 43-2). An assistant is at the patient side for suctioning, retraction, and passage of suture or sponges.
FIGURE 43-1. A control console where the surgeon is stationed and operates the robotic arms and camera. © 2012 Intuitive Surgical, Inc.
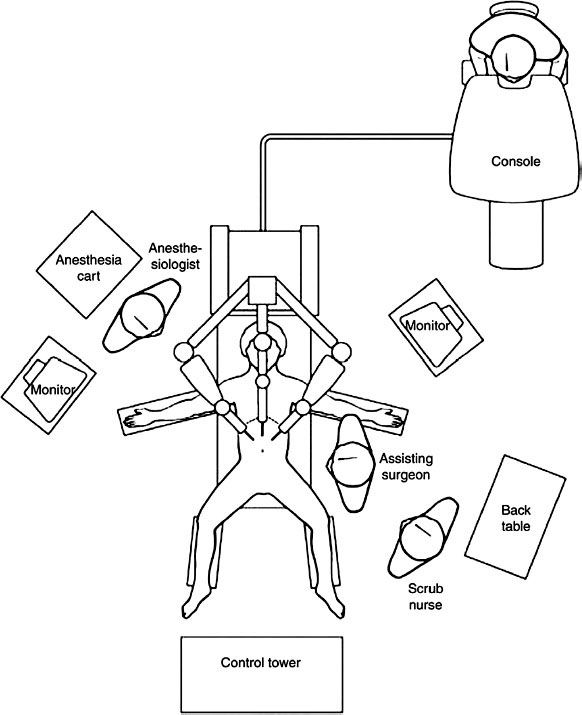
FIGURE 43-2. Layout of the operating room during robotic surgery. Reproduced from:.
Patient position during minimally invasive surgery varies significantly based on the surgical procedure. Patients undergoing upper abdominal procedures require a reverse Trendelenburg (head-up) position, while those undergoing lower abdominal procedures require Trendelenburg (head-down) position. The head-up or head-down position can be steep. In addition, the operating tables may be rotated laterally (right or left lateral) to further facilitate surgical exposure. Also, patients undergoing pelvic surgery (e.g., radial prostatectomy and hysterectomy) may be placed in a lithotomy position. Patients undergoing urologic surgery, particularly renal procedures, may be placed in lateral or semilateral positions combined with a flexion (i.e., jackknife) position. The considerations related to patient positioning are discussed in Chapter 28.
PHYSIOLOGIC EFFECTS
The physiologic consequences of laparoscopy can be complex and depend on the interactions between the patient’s pre-existing cardiopulmonary status and surgical factors such as the magnitude of IAP, degree of CO2 absorption, alteration of patient position, and the type of surgical procedure.2,14,15 In addition, the anesthetic technique may influence the physiologic changes; however, these effects may be minimal with modern anesthetic techniques. Of note, physiologic changes are well tolerated by most healthy patients; however, they could have adverse consequences in patients with limited cardiopulmonary reserve.
Cardiovascular Effects
 The changes in the cardiovascular function during laparoscopy are due to the mechanical and neuroendocrine effects of pneumoperitoneum and the effects of absorbed CO2 and patient positioning as well as patient factors such as cardiopulmonary status and intravascular volume (Table 43-3). The induction of pneumoperitoneum in the supine position (rather than head-down position) and limiting the IAP to 12 to 15 mm Hg minimize the alterations in cardiovascular function during laparoscopy.2,14
The changes in the cardiovascular function during laparoscopy are due to the mechanical and neuroendocrine effects of pneumoperitoneum and the effects of absorbed CO2 and patient positioning as well as patient factors such as cardiopulmonary status and intravascular volume (Table 43-3). The induction of pneumoperitoneum in the supine position (rather than head-down position) and limiting the IAP to 12 to 15 mm Hg minimize the alterations in cardiovascular function during laparoscopy.2,14
TABLE 43-3. HEMODYNAMIC EFFECTS OF MINIMALLY INVASIVE SURGERY

The cardiovascular changes of laparoscopy include an increase in systemic vascular resistance (SVR) and mean arterial pressure (MAP), which is caused by increased sympathetic output from CO2 absorption and a neuroendocrine response to pneumoperitoneum.2,14,15 Pneumoperitoneum-related increased IAP results in activation of the sympathetic system with catecholamine release and the renin–angiotensin system with vasopressin release.16 The profile of vasopressin release parallels the time course of changes in SVR. In addition, compression of the arterial vasculature from increased IAP may also lead to an increase in SVR. These neuroendocrine and mechanical responses supersede the hypercapnia-induced arteriolar dilation and decrease SVR. The increase in SVR may increase the myocardial wall tension and, thus, may increase the myocardial oxygen demand. However, myocardial ischemia, as suggested by electrocardiogram-ST–segment changes, is not observed.17
The changes in cardiac filling pressures and volumes during laparoscopy appear to be complex. Increased cardiac filling pressures may reflect increased intrathoracic pressures caused by pneumoperitoneum and increased sympathetic output due to hypercapnia from CO2 absorption and surgical stress. However, cardiac filling pressures may not always reflect cardiac filling volumes. Increased IAP may compress venous capacitance vessels, causing a decrease in preload (cardiac filling volume), particularly in hypovolemic patients. In contrast, compression of the abdominal organs (e.g., liver and spleen) caused by increased IAP may increase intravascular volume, which may increase cardiac filling, particularly if the patient is placed in a head-down position.18 Overall, the cardiac filling pressures and volumes increase, but minimally.
In healthy patients, the changes in cardiac index (CI) appear to be phasic with initial reduction after induction of pneumoperitoneum and subsequent recovery within 10 to 15 minutes. Overall, the changes in CI in healthy patients are minimal. However, in patients with severe cardiac dysfunctions, there may be a significant reduction in CI and significant hemodynamic deterioration.19 Although reduction in CI parallels the time course of increase in SVR, the cause–effect relationship between SVR and CI is unclear. In addition, significant hypercapnia and associated respiratory acidosis may decrease myocardial contractility and lower the arrhythmia threshold. Hypercarbia can cause pulmonary vasoconstriction, which may be deleterious in patients with pulmonary hypertension or right ventricular dysfunction.
 In the morbidly obese, the hemodynamic changes are similar to those observed in the nonobese.20,21 Although the reasons for this observation are not clear, it is hypothesized that lack of differences in hemodynamics may be related to intrinsically higher IAP in the obese (10 mm Hg vs. 5 mm Hg in the nonobese).
In the morbidly obese, the hemodynamic changes are similar to those observed in the nonobese.20,21 Although the reasons for this observation are not clear, it is hypothesized that lack of differences in hemodynamics may be related to intrinsically higher IAP in the obese (10 mm Hg vs. 5 mm Hg in the nonobese).
In the elderly, with significant coexisting cardiopulmonary disease, pneumoperitoneum and head-down position cause several hemodynamic changes.22 Induction of pneumoperitoneum significantly increased SVR accompanied with a significant reduction in CI and ejection fraction (EF). However, the left ventricular workload remained unchanged. Upon placement in the head-down position, the cardiac preload, as determined by the left ventricular end-diastolic area, increased and CI and EF improved.22 Oxygenation and ventilation remained unchanged, and no patients exhibited electrocardiogram signs of myocardial ischemia. Release of pneumoperitoneum resulted in a significant decrease in SVR and increased CI and left ventricular systolic work index.
The type of surgical procedure may also influence the degree of hemodynamic derangement. Surgical disruption of the esophageal hiatus during laparoscopic fundoplication may increase mediastinal and pleural pressures, resulting in a significant reduction in CI.23,24 Patients undergoing endoscopic radical prostatectomy in the Trendelenburg position did not experience hemodynamic changes, despite prolonged duration (average 4 hours) of pneumoperitoneum.25
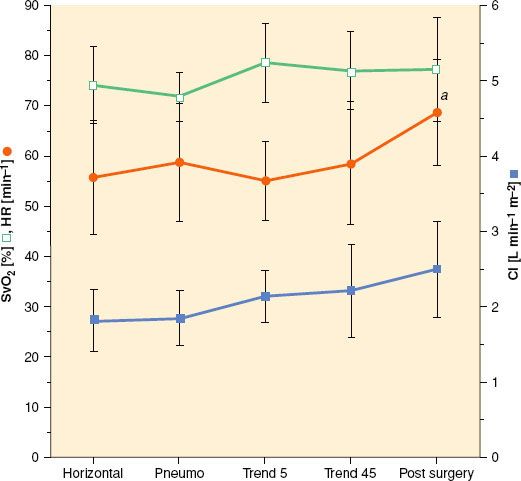
The hemodynamic changes that occur during abdominal robotic surgery appear to be similar to those observed during laparoscopic surgery. Most of the studies evaluating such changes are performed in patients undergoing prostatectomy with steep head-down position. Peritoneal insufflation and steep (40 degrees) head-down position during robotic-assisted prostatectomy increase SVR and MAP, while other hemodynamic variables remain in acceptable limits26,27 (Figs. 43-3 and 43-4). A recent study found that although cardiac filling pressures were increased, the cardiac performance (stroke volume, cardiac output, and mixed venous oxygen saturation as well as echocardiographic cardiac dimensions) was maintained during robotic-assisted prostatectomy with patients in 45-degree head-down tilt and pneumoperitoneum with IAP 12 mm Hg28 (Fig. 43-5). Overall, robotic surgery appears to be well tolerated in a healthy population. However, the physiologic changes in the elderly or in patients with impaired cardiopulmonary reserve undergoing robotic prostatectomy remain unknown.
FIGURE 43-3. Hemodynamic changes (heart rate (HR), mean arterial pressures (MAPs), central venous pressures (CVPs), cerebral perfusion pressure (CPP), arterial oxygen saturation measured by pulse oximetry (SpO2), and regional cerebral tissue oxygen saturation (SctO2) in patients undergoing laparoscopic robotic prostatectomy in steep head-down position. Thin lines indicate values in individual patients and thick lines indicate mean values (From: Kalmar AF, Foubert L, Hendrickx JFA, et al. Influence of steep Trendelenburg position and CO2 pneumoperitoneum on cardiovascular, cerebrovascular, and respiratory homeostasis during robotic prostatectomy. Br J Anaesth. 2012;104:433–439).
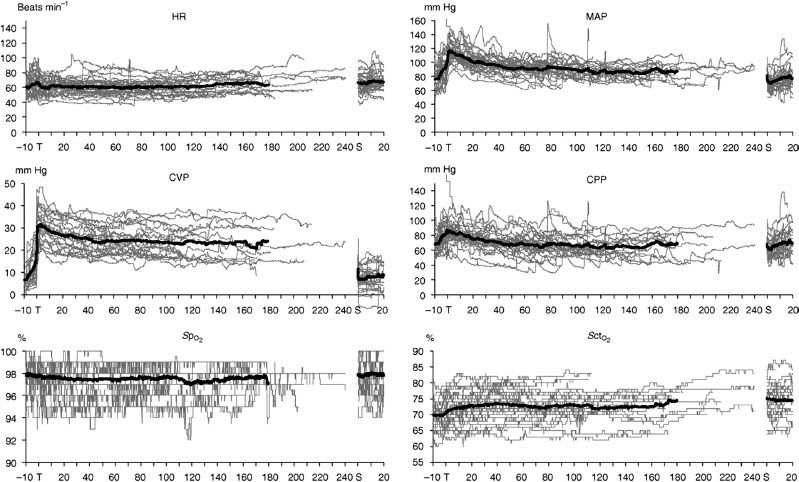
FIGURE 43-4. Changes in end-tidal CO2 values (PECO2), ventilatory plateau pressure (Pplat), tidal volume, and pulmonary compliance in patients undergoing laparoscopic robotic prostatectomy in steep head-down position. Thin lines indicate values in individual patients and thick lines indicate mean values (From: Kalmar AF, Foubert L, Hendrickx JFA, et al. Influence of steep Trendelenburg position and CO2 pneumoperitoneum on cardiovascular, cerebrovascular, and respiratory homeostasis during robotic prostatectomy. Br J Anaesth. 2012;104:433–439).
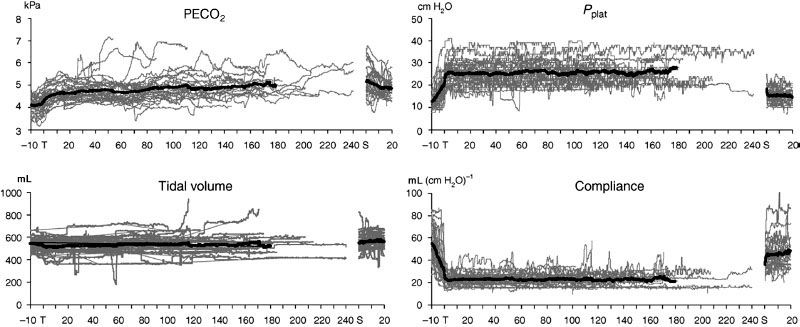
FIGURE 43-5. Changes (mean and standard deviations) in mixed venous oxygen saturation (SvO2), heart rate (HR), and cardiac index (CI).
Regional Perfusion (Splanchnic, Renal, Cerebral, Intraocular)
Increased IAP, systemic CO2 absorption, and changes in patient position, along with hemodynamic changes (e.g., SVR and CI), influence splanchnic, renal, and cerebral blood flow during minimal access surgery (Table 43-4). However, the clinical consequences of these changes depend largely on the patient’s pre-existing status.
TABLE 43-4. REGIONAL CIRCULATORY CHANGES DURING LAPAROSCOPY
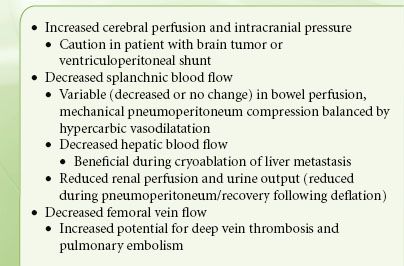
 The direct mechanical and neuroendocrine effects of pneumoperitoneum can decrease splanchnic circulation, causing reduced total hepatic blood flow and bowel circulation. However, these effects may be counterbalanced by the direct splanchnic vasodilatation caused by hypercapnia. Notwithstanding occasional reports of mesenteric ischemia following laparoscopy, the effects of pneumoperitoneum on the splanchnic circulation are not clinically significant.
The direct mechanical and neuroendocrine effects of pneumoperitoneum can decrease splanchnic circulation, causing reduced total hepatic blood flow and bowel circulation. However, these effects may be counterbalanced by the direct splanchnic vasodilatation caused by hypercapnia. Notwithstanding occasional reports of mesenteric ischemia following laparoscopy, the effects of pneumoperitoneum on the splanchnic circulation are not clinically significant.
The mechanical compressive and neuroendocrine effects of pneumoperitoneum may account for reduction in renal blood flow, glomerular filtration, and urine output (Table 43-5).29,30 However, the urine output generally normalizes following pneumoperitoneum deflation with no consequent renal dysfunction. Nevertheless, there may be clinical implications in critically ill patients and those with renal dysfunction undergoing extensive laparoscopic procedures requiring prolonged pneumoperitoneum.
TABLE 43-5. RENAL FUNCTION DURING LAPAROSCOPY

An increase in PaCO2 during steep Trendelenburg positioning can increase cerebral blood flow and intracranial pressure with implications for patients with intracranial mass lesions. Therefore, maintenance of normocarbia is essential for preservation of cerebrovascular homeostasis.31 However, cerebral oxygenation and cerebral perfusion remain within safe limits during combined pneumoperitoneum and Trendelenburg position.27,32,33
Choroidal vasodilatation and an increase in intraocular pressure may occur during CO2 pneumoperitoneum and steep head-down position.33 Intraocular pressure increased significantly during robotic-assisted radical prostatectomy with steep head-down position.33 Multivariate analysis suggested that the predictors of IOP include duration of surgery and end-tidal CO2 (ETCO2).
Respiratory and Gas Exchange Effects
 Changes in pulmonary function during laparoscopy include reduction in lung volume and pulmonary compliance secondary to cephalad displacement of the diaphragm caused by increased IAP and patient positioning14,15 (Table 43-6). Reduction in functional residual capacity (FRC) and total lung compliance results in basal atelectasis and increased airway pressure. In addition, the increase in minute ventilation required to avoid hypercarbia caused by systemic CO2 absorption further increases peak airway pressures. Although these changes are well tolerated by healthy patients, significant pulmonary dysfunction may occur in patients with pre-existing pulmonary disease (see the section on complications).
Changes in pulmonary function during laparoscopy include reduction in lung volume and pulmonary compliance secondary to cephalad displacement of the diaphragm caused by increased IAP and patient positioning14,15 (Table 43-6). Reduction in functional residual capacity (FRC) and total lung compliance results in basal atelectasis and increased airway pressure. In addition, the increase in minute ventilation required to avoid hypercarbia caused by systemic CO2 absorption further increases peak airway pressures. Although these changes are well tolerated by healthy patients, significant pulmonary dysfunction may occur in patients with pre-existing pulmonary disease (see the section on complications).
TABLE 43-6. PULMONARY CHANGES DURING LAPAROSCOPY

The CO2 insufflated into the peritoneal cavity is absorbed and causes hypercarbia. The absorption of gas from the peritoneal cavity depends on its diffusivity, the absorption area, and vascularity of insufflation site. Carbon dioxide absorption is greater during extraperitoneal (e.g., pelvic, hernia repair, and adrenorenal surgeries) insufflation than during intraperitoneal insufflation (e.g., cholecystectomy).34 The CO2 absorption reaches a plateau within 10 to 15 minutes after initiation of intraperitoneal insufflation and thus is not influenced by the duration of surgery.35 However, it continues to increase progressively throughout extraperitoneal CO2 insufflation.
 Although laparoscopic surgery is associated with increased CO2 absorption, the changes in arterial CO2 (PaCO2) concentrations remain clinically insignificant in healthy patients. However, in patients with severe pulmonary disease and limited elimination of CO2, the resulting rise in PaCO2 may be significant despite aggressive hyperventilation. In addition, in this patient population, the ETCO2 levels may underestimate arterial CO2 concentrations (PaCO2). Interestingly, the absorption and excretion of CO2 in morbidly obese patients appear to be similar to that of nonobese patients.20 However, in obese patients placed in the head-down position, arterial oxygenation and the alveolar–arterial oxygen gradient are impaired.21
Although laparoscopic surgery is associated with increased CO2 absorption, the changes in arterial CO2 (PaCO2) concentrations remain clinically insignificant in healthy patients. However, in patients with severe pulmonary disease and limited elimination of CO2, the resulting rise in PaCO2 may be significant despite aggressive hyperventilation. In addition, in this patient population, the ETCO2 levels may underestimate arterial CO2 concentrations (PaCO2). Interestingly, the absorption and excretion of CO2 in morbidly obese patients appear to be similar to that of nonobese patients.20 However, in obese patients placed in the head-down position, arterial oxygenation and the alveolar–arterial oxygen gradient are impaired.21
A recent study found that pH decreased during laparotomy open procedures and laparoscopic procedures with CO2 pneumoperitoneum. However, reduced pH during the pneumoperitoneum was due to an increase in PaCO2 and promptly returned to a normal value after the desufflation of the abdomen. In contrast, reduction in pH after laparotomy was from metabolic factors and persisted for approximately an hour postoperatively.36
A recent animal study found that the improved arterial oxygenation and gas exchange after induction of pneumoperitoneum was due to improved ventilation–perfusion matching caused by redistribution of perfusion away from the collapsed lung regions. This was probably caused by enhanced hypoxic pulmonary vasoconstriction possibly mediated via increased arterial CO2.37
During robotic-assisted hysterectomy and prostatectomy performed under steep (40 degrees) head-down position, the changes in dead-space ventilation and venous admixture appear to be small.38 Another study in patients undergoing robotic prostatectomy also found minimal changes in respiratory parameters.27 However, the arterial end-tidal CO2 gradient increased after 120 minutes. Therefore, ETCO2 values may underestimate arterial CO2 levels, and maintaining ETCO2 between 25 and 35 mm Hg will result in PaCO2 levels of 35 to 45 mm Hg. Similarly, the institution of pneumoperitoneum (IAP of 12 mm Hg) and 45-degree head-down positioning resulted in decreased lung compliance by 40%.28 The ventilation–perfusion distribution did not differ significantly from baseline measurements, and oxygenation actually improved, probably due to optimization of intraoperative ventilation.28
ANESTHETIC MANAGEMENT
 An optimal anesthetic technique would provide excellent intraoperative conditions while ensuring rapid recovery and low incidence of adverse effects as well as allowing early return to daily living activities.39,40 Local and regional anesthesia (spinal and epidural) can be used for shorter laparoscopic procedures, such as diagnostic laparoscopy, which requires lower IAP and minimal head-down tilt.40,41 Nevertheless, patient discomfort associated with creation of pneumoperitoneum and extreme position changes during the procedure can be significant. In addition, neuraxial anesthesia can cause significant sympathetic denervation, which may be associated with adverse ventilatory and circulatory responses, complicating perioperative management. Therefore, balanced general anesthesia with tracheal intubation and mechanical ventilation with acceptance of higher end-tidal carbon dioxide levels remains the best practice for minimally invasive surgical procedures.
An optimal anesthetic technique would provide excellent intraoperative conditions while ensuring rapid recovery and low incidence of adverse effects as well as allowing early return to daily living activities.39,40 Local and regional anesthesia (spinal and epidural) can be used for shorter laparoscopic procedures, such as diagnostic laparoscopy, which requires lower IAP and minimal head-down tilt.40,41 Nevertheless, patient discomfort associated with creation of pneumoperitoneum and extreme position changes during the procedure can be significant. In addition, neuraxial anesthesia can cause significant sympathetic denervation, which may be associated with adverse ventilatory and circulatory responses, complicating perioperative management. Therefore, balanced general anesthesia with tracheal intubation and mechanical ventilation with acceptance of higher end-tidal carbon dioxide levels remains the best practice for minimally invasive surgical procedures.
Induction of Anesthesia and Airway Management
Because of its unique recovery profile, propofol is considered the sedative–hypnotic drug of choice for induction of anesthesia. Propofol also offers an advantage over other intravenous anesthetics because of its antiemetic properties and associated euphoria on emergence. Tracheal intubation and controlled mechanical ventilation comprise the accepted anesthetic technique to reduce the increase in PaCO2 and avoid ventilatory compromise from pneumoperitoneum and position changes. Although the laryngeal mask airway (LMA) has been used during short pelvic laparoscopic procedures, this evidence cannot be extrapolated to procedures requiring high IAP, steep head-down position, and upper abdominal laparoscopy as well as in patients at increased risk of regurgitation.42,43
Maintenance of Anesthesia
Maintenance of anesthesia with the newer inhaled anesthetics (i.e., desflurane or sevoflurane) remains the mainstay of modern anesthesia practice, probably because of the ease of titratability.39,44 In addition, inhaled anesthetics exert some neuromuscular blocking effect. Furthermore, inhalation anesthesia may provide faster emergence as compared to total intravenous anesthesia (TIVA) with propofol. However, propofol-based TIVA is associated with a lower risk of postoperative nausea and vomiting (PONV), but its cost and apparent complexity (i.e., need for infusion and difficulty in titration) deter some practitioners.44,45 Of note, except for patients with very high risk of PONV, the incidence of PONV with TIVA appears to be similar to that with inhalation anesthesia combined with prophylactic antiemetics.44
Nitrous Oxide
N2O is widely used as part of a balanced anesthetic technique because of its amnestic and analgesic properties, as well as its ability to reduce the requirements of inhaled and intravenous anesthetic drugs and facilitate recovery.46,47 Elimination of N2O at the end of anesthesia produces a clinically significant acceleration in the reduction of inhaled anesthetic concentrations, further contributing to the speed of emergence.48 However, its use during laparoscopic procedures has been controversial as a result of concerns regarding its ability to diffuse into bowel lumen, causing distension and impaired surgical access as well as increased PONV.49 However, surgical conditions during laparoscopic procedures have been shown to be identical, regardless of whether N2O was used.50 In particular, surgeons were unable to distinguish between patients who had received N2O compared to air.50
Although N2O can contribute to PONV, the overall impact appears to be modest.51 The use of propofol for induction of anesthesia and antiemetic prophylaxis, which is the current standard of care, further negates the emetic effects of N2O.51 A meta-analysis of randomized controlled trials found that the emetic effect of N2O is not significant, and omitting N2O may increase the risk of awareness.52 Thus, there is no convincing reason to routinely avoid N2O during laparoscopic procedures. However, omitting N2O from the anesthesia regimen may be an option in patients at very high risk of PONV or when there are surgical difficulties.
In a laboratory experimental study, N2O was shown to diffuse into a CO2 pneumoperitoneum, and the levels were high enough to support combustion within 2 hours.53 As laparoscopic surgical techniques become more complex and with the increasing use of laser and electrocautery, the chance of bowel perforation and release of highly volatile bowel gases (methane and hydrogen) into the peritoneal cavity is highly likely. However, the clinical significance of these issues remains unclear. In practice, leakage of intraperitoneal gas and replacement with fresh CO2 are likely to wash out N2O and reduce the risk of explosion.54
Intraoperative Opioids
Opioids remain an important component of a balanced general anesthetic technique and are typically titrated to achieve hemodynamic stability. However, treatment of pneumoperitoneum-induced hypertension with opioids may lead to relative opioid overdose after abdominal deflation. Therefore, it may be prudent to use an ultrashort-acting opioid (i.e., remifentanil) or sympatholytic drugs (e.g., esmolol and nicardipine) to treat pneumoperitoneum-induced hypertension. Remifentanil is a novel opioid with a short duration of action independent of the duration of infusion. Numerous studies have reported a reliable and rapid emergence from anesthesia with the use of remifentanil. Because of its short duration, transitional analgesics must be administered before emergence from anesthesia.55
Overall, opioids should be used sparingly because of concerns of opioid-related adverse effects such as dizziness, drowsiness, nausea, and vomiting.55 Another concern has been raised regarding opioid-induced spasm of the sphincter of Oddi,56 leading to misinterpretation of intraoperative cholangiographic findings during laparoscopic cholecystectomy. Many opioids, including fentanyl, have been implicated, and there are conflicting reports regarding the relative effect of individual opioids.56 If necessary, the opioid-induced spasm of the sphincter of Oddi may be antagonized by glucagon.
Muscle Relaxants and Reversal of Residual Neuromuscular Blockade
Muscle paralysis reduces the IAP needed for the same degree of abdominal distension. However, several studies have not been able to show a correlation between the degree of muscle paralysis and the surgeon’s ability.57,58 Therefore, deep muscle paralysis may not be necessary during laparoscopic procedures. However, patient movement during robotic surgery can result in displacement of the robotic arms, which has a potential for patient harm.59
Recent evidence suggests that even a minor degree of residual neuromuscular blockade can increase the incidence of postoperative respiratory complications in high-risk patients such as the morbidly obese and elderly.60–62 Unfortunately, residual neuromuscular blockade is difficult to diagnose clinically or by using train-of-four (TOF) response monitoring.63 Recently, acceleromyography that provides a TOF ratio has been shown to reduce the incidence of residual blockade and associated unpleasant symptoms of muscle weakness in the recovery room and thus improve the overall quality of recovery.63 Reversal with neostigmine is sometimes avoided due to concerns of higher incidence of PONV. However, the incidence of PONV and the need for antiemetics do not increase with the appropriate use of neostigmine combined with glycopyrrolate.64,65 Therefore, reversal drugs should be used without hesitation. The dose of neostigmine should be determined by the degree of blockade at the time of reversal.61,66
Mechanical Ventilation
The changes in pulmonary function (e.g., reduction in lung volume, increase in peak inspiratory pressure, and decrease in pulmonary compliance) during laparoscopy may require intraoperative modification in mechanical ventilation. Typically the minute ventilation needs to be increased by 20% to 30%, which could be achieved by increasing the respiratory rate while maintaining a constant tidal volume. Lung protective ventilation strategies include the use of pressure-controlled ventilation with low tidal volumes (6 to 8 mL/kg ideal body weight) and positive end-expiratory pressure (PEEP) of 5 to 10 cm water (H2O).67 Use of PEEP has been shown to improve arterial oxygenation during prolonged pneumoperitoneum.68 In addition, recruitment maneuvers are beneficial and should be applied, particularly before and after a laparoscopic procedure.69 Unfortunately, the effects of recruitment maneuvers are short lasting and often limited by hemodynamic instability. It is important to avoid hyperventilation (and hypocapnia), as this may result in metabolic alkalosis and lead to postoperative hypoventilation. The acceptance of higher ETCO2 levels may limit peak airway pressures (<50 cm H2O) and potential barotrauma. Mild hypercapnia (i.e., ETCO2 of 40 mm Hg) can improve tissue oxygenation through improved tissue perfusion, resulting from increased cardiac output and vasodilatation as well as increased oxygen off-loading from the shift of the oxyhemoglobin dissociation curve to the right.67,70,71
Monitoring
In addition to standard intraoperative monitoring, minimally invasive hemodynamic monitoring (e.g., arterial waveform analysis) may be appropriate in patients with significant cardiopulmonary disease to monitor the cardiovascular response to pneumoperitoneum and position changes in order to institute therapy. The arterial waveform analysis provides a relatively noninvasive means of estimating ventricular contractility and stroke volume on a beat-to-beat basis.72,73 In addition, these monitors could guide fluid therapy by using dynamic indicators such as stroke volume variation.
Although ETCO2 provides a reasonable approximation of PaCO2 in healthy patients, this may not be true in patients with pre-existing cardiopulmonary disease. Therefore, monitoring of arterial blood gases may be prudent in patients with significant pulmonary disease or those with persistent refractory intraoperative hypercapnia. Also, compliance and pressure–volume loop monitoring may be helpful in diagnosing complications resulting in increased airway pressure, such as endobronchial intubation, bronchospasm, and pneumothorax. Recently, cerebral oximetry monitoring was found to provide brain oxygenation,74–76 and could be particularly useful in high-risk patients undergoing prolonged endoscopic procedures in steep head-up or head-down procedures.32
Fluid Management
Maintenance of optimal intravascular volume or cardiac filling is critical in improving perioperative outcomes.77,78 This is particularly critical during prolonged laparoscopic procedures in the high-risk patient population. However, fluid therapy remains one of the most controversial topics in perioperative management. There is continuing debate regarding the type of fluids used (i.e., crystalloids, colloids, or a type of colloid) as well as the amount of fluid administration (i.e., liberal vs. restricted or goal-directed approach).
It is increasingly becoming clear that intraoperative fluid therapy should be specific to patient characteristics and the type of surgical procedure. For mild to moderately invasive surgical procedures, crystalloid boluses (20 to 40 mL/kg) have been shown to improve outcomes, such as reduced postoperative dizziness and early ambulation.79 For major elective surgical procedures, the use of fluid minimization and a goal-directed fluid administration approach are recommended.77,78 In patients undergoing robotic prostatectomy, fluid minimization is recommended, as steep head-down positioning for prolonged period may result in facial, pharyngeal, and laryngeal edema. Also, large fluid volumes can lead to high urine output and interfere with the surgical procedure.
It is clear that traditional indicators used to guide fluid therapy (e.g., heart rate, arterial blood pressure, central venous pressures, and urine output) are not reliable. Urine output is reduced during laparoscopy, and its use to guide fluid therapy may lead to fluid overload. Therefore, dynamic indicators, such as stroke volume or systolic or pulse pressure variation, are preferred. Optimization of dynamic indicators can be achieved by administration of small fluid boluses.80 Of note, deep general anesthesia and hyperventilation can influence fluid requirements and thus should be avoided.
NAUSEA AND VOMITING PREVENTION
Patients undergoing laparoscopic surgery are at a greater risk for PONV, probably due to intraperitoneal insufflation and bowel manipulation.81 Therefore, aggressive multimodal antiemetic prophylaxis is necessary in this high-risk population. A multimodal approach to PONV prophylaxis could include use of combinations of dexamethasone, 4 to 8 mg administered after induction of anesthesia, and 5-HT3 antagonists (e.g., ondansetron 4 mg) at the end of surgery as well as aggressive hydration, minimal opioid use, and aggressive pain control.81–83 Other antiemetic therapy includes a scopolamine patch applied preferably prior to surgery and neurokinin-1 receptor antagonists administered preoperatively.84,85
PAIN PREVENTION
 Compared to open surgical procedures, pain after laparoscopic procedures is considered to be less intense and of shorter duration.86 Nevertheless, adequate pain control is critical to hasten postoperative recovery and ambulation.55 The origin of pain after most laparoscopic and robotic procedures is predominantly visceral rather than parietal (i.e., from incision site). In addition, shoulder pain secondary to diaphragmatic irritation is also common and can limit the patient’s ability to return to normal activities. The factors that could influence postoperative pain include duration of procedure, degree of IAP,87 and the volume of residual subdiaphragmatic gas after surgery. Optimal pain therapy for patients undergoing laparoscopic and robotic surgeries includes the use of multimodal analgesia techniques.55,88 Because opioid-related adverse effects may delay recovery, nonopioid analgesics should be used when possible.
Compared to open surgical procedures, pain after laparoscopic procedures is considered to be less intense and of shorter duration.86 Nevertheless, adequate pain control is critical to hasten postoperative recovery and ambulation.55 The origin of pain after most laparoscopic and robotic procedures is predominantly visceral rather than parietal (i.e., from incision site). In addition, shoulder pain secondary to diaphragmatic irritation is also common and can limit the patient’s ability to return to normal activities. The factors that could influence postoperative pain include duration of procedure, degree of IAP,87 and the volume of residual subdiaphragmatic gas after surgery. Optimal pain therapy for patients undergoing laparoscopic and robotic surgeries includes the use of multimodal analgesia techniques.55,88 Because opioid-related adverse effects may delay recovery, nonopioid analgesics should be used when possible.
Conventional nonsteroidal anti-inflammatory drugs or cyclooxygenase-2–specific inhibitors have been shown to reduce postoperative pain and opioid requirements and thus should be considered part of analgesic therapy.89 These analgesics could be combined with acetaminophen,90 as the combination is superior to either drug alone.91,92 If pain relief remains inadequate, analgesia may be supplemented with weak opioids for low- to moderate-intensity postoperative pain and with strong opioids for moderate- to high-intensity postoperative pain.88 These drugs should be administered at the appropriate time (preoperative or intraoperative) to provide sufficient analgesia in the early recovery period as well as continued in the postoperative period. The use of these medications should depend on assessment of an individual patient’s risks.
Parenteral glucocorticoids (e.g., dexamethasone) should also be considered part of a multimodal analgesia technique because they have been shown to reduce postoperative pain as well as length of hospital stay after abdominal surgery with no evidence of increased complications, including after colorectal surgery.93,94 Although ketamine has some documented benefits,95 its routine use remains controversial due to lack of data on optimal dose and duration.
Infiltration of the surgical wound (laparoscopy portals) with local anesthetics can provide excellent analgesia that outlasts the duration of action of the local anesthetic.88,96,97 For hybrid or laparoscopic-assisted surgical procedures with longer incisions, continuous local anesthetic wound infusion may be used to prolong the duration of analgesia.98 The transversus abdominis plane (TAP) block, which involves administration of local anesthetic between the layers of internal oblique and transversus abdominis muscles, may be beneficial in patients undergoing hybrid or laparoscopic-assisted procedures,99,100 but its role in procedures with small incisions remains controversial.
Intraperitoneal instillation of long-acting local anesthetics (e.g., ropivacaine) has been shown to reduce the intensity of postlaparoscopic pain.101,102 However, the concentration and dose of the local anesthetic as well as optimal timing of administration remain unknown. Therefore, intraperitoneal local anesthetic instillation remains controversial. Recently, intraperitoneal nebulization of ropivacaine has been shown to provide excellent postlaparoscopic pain relief, particularly shoulder pain.103 Intravenous lidocaine infusion in the intraoperative and the postoperative periods has been reported to reduce postoperative pain and opioid requirements and improve bowel function as well as reduce the length of hospital stay.104–106 However, the optimal dose and duration of lidocaine infusion remain unknown. No significant local anesthetic toxicity has been reported in published systematic reviews, and adverse events were not systematically evaluated. Therefore, routine use of lidocaine infusion is not recommended. Nevertheless, lidocaine infusion could be an alternative when other approaches are inadequate.
Although epidural analgesia provides excellent pain relief with improved outcomes after open major abdominal surgery, its role in laparoscopic surgery remains controversial.86,107 Similarly, intrathecal morphine provides excellent pain relief, but it is associated with opioid-related adverse effects such as increased PONV, itching, urinary retention, and respiratory depression.108 For most minimally invasive surgical procedures, neuraxial analgesia is not necessary because of a lack of superiority over other analgesic techniques and the potential risks of adverse effects, such as hemodynamic instability, postural hypotension, and urinary retention as well as delay of early ambulation.109
INTRAOPERATIVE COMPLICATIONS
Intraoperative complications during minimally invasive surgery include those related to creation of pneumoperitoneum through intraperitoneal CO2 insufflation, patient positioning, and surgical instrumentation.110 These include cardiopulmonary compromise, renal dysfunction, and hypothermia. Surgical complications include subcutaneous emphysema, capnothorax, capnomediastinum, capnopericardium, gas embolism, acute hemorrhage, and bowel or bladder perforation, which can occur during the laparoscopic procedure. Procedures with higher incidence of complications include upper abdominal procedures, such as fundoplication, and urologic procedures, particularly in patients with significant comorbidities.111–115
Cardiopulmonary Complications
Hemodynamic complications associated with laparoscopic procedures include dysrhythmias and alterations in arterial blood pressure (i.e., hypotension and hypertension). Bradyarrhythmias are attributed to increased vagal tone following peritoneal stretching, especially associated with lighter levels of anesthesia; while tachyarrhythmias may be due to hypercapnia as a result of intraperitoneal CO2 insufflation. Paroxysmal tachycardia and hypertension followed by ventricular fibrillation have been reported during laparoscopic adrenalectomy.116 Although rare, acute cardiovascular collapse can occur (Table 43-7).
TABLE 43-7. DIFFERENTIAL DIAGNOSIS OF CARDIOVASCULAR COLLAPSE DURING LAPAROSCOPY

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







