The Physics Teacher
1969
Magnesium is an essential element for the utilization of energy in the organic world. In green plants, magnesium is the heart of the chlorophyll molecule that captures the energy in sunlight in order to produce oxygen and carbohydrates (i.e., photosynthesis). Aerobic organisms then use the oxygen to release the energy stored in organic nutrients (including carbohydrates), and this energy is stored as adenosine triphosphate (ATP). The release of energy from ATP requires magnesium, which is an essential cofactor for the ATPase enzymes that hydrolyze ATP. Therefore, magnesium is essential for providing us with energy, and for allowing us to utilize this energy to sustain life. Now that’s an element.
More specific roles of magnesium include the proper functioning of the Na+-K+ exchange pump (which is a magnesium-dependent ATPase) that generates the electrical gradient across cell membranes. As a result, magnesium plays an important role in the activity of electrically excitable tissues (1–4). Magnesium also regulates the movement of calcium into smooth muscle cells, which gives it a pivotal role in the maintenance of cardiac contractile strength and peripheral vascular tone (4).
MAGNESIUM BASICS
Distribution
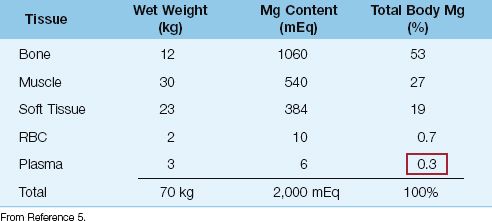
The distribution of magnesium (Mg) in the human body is shown in Table 37.1 (5). The average-sized adult contains approximately 24 g (1 mole, or 2,000 mEq) of magnesium; a little over half is located in bone, whereas less than 1% is located in plasma. This lack of representation in the plasma limits the value of the plasma Mg as an index of total body magnesium (similar to plasma potassium). This is particularly true in patients with magnesium deficiency, in whom plasma Mg levels can be normal in the face of total body magnesium depletion (5,6).
Table 37.1 Magnesium Distribution in Adults
Serum Magnesium
Serum is favored over plasma for magnesium assays because the anticoagulant used for plasma samples can be contaminated with citrate or other anions that bind magnesium (5). The reference range for serum Mg depends on the daily magnesium intake, which varies according to geographic region. The normal range of serum Mg for healthy adults in the United States is shown in Table 37.2 (7).
Note: The clinical laboratory typically reports the serum Mg concentration in mg/dL (because Mg is partially bound to plasma proteins), while the medical literature typically uses mEq/L for the serum Mg concentration. The conversion is as follows:
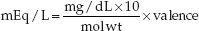 (37.1)
(37.1)
where mol wt is the molecular weight (atomic weight in the case of magnesium) and valence is the number of charges on the atom or molecule. Magnesium has an atomic weight of 24 and a valence of 2, so a serum Mg concentration of 1.7 mg/dL is equivalent to (1.7×10)/24×2 = 1.4 mEq/L.
Ionized Magnesium
About 67% of the magnesium in plasma is in the ionized (active) form, and the remaining 33% is either bound to plasma proteins (19% of the total) or chelated with divalent anions such as phosphate and sulfate (14% of the total) (8). The standard assay for magnesium (i.e., spectrophotometry) measures all three fractions. Therefore, when the serum Mg is abnormally low, it is not possible to determine if the problem is a decrease in the ionized (active) fraction, or a decrease in the bound fractions (e.g., hypoproteinemia) (9). The level of ionized Mg can be measured with an ion-specific electrode (10), but this is not routinely available. However, because only a small amount of magnesium resides in plasma, the difference between the ionized and bound magnesium content may not be large enough to be clinically relevant.
Table 37.2 Reference Ranges for Magnesium

Urinary Magnesium
The normal range for urinary Mg excretion is shown in Table 37.2. Under normal circumstances, only small quantities of magnesium are excreted in the urine (3). When magnesium intake is deficient, the kidneys conserve magnesium, and urinary magnesium excretion falls to negligible levels. This is shown in Figure 37.1. Note that the serum Mg remains in the normal range one week after starting a Mg-3 diet, while the urinary Mg excretion has dropped to negligible levels. This illustrates the relative value of urinary magnesium excretion in the detection of magnesium deficiency.
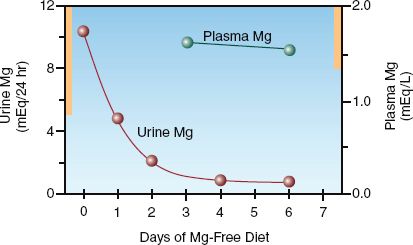
FIGURE 37.1 Urinary magnesium excretion and plasma magnesium levels in a healthy volunteer placed on a magnesium-free diet. Solid bars on the vertical axes indicate the normal range for each variable. Adapted from Shils ME. Experimental human magnesium deficiency. Medicine 1969;48:61–82.
MAGNESIUM DEFICIENCY
Hypomagnesemia is reported in as many as 65% of patients in ICU’s (1–3). Because magnesium depletion may not be accompanied by hypomagnesemia, the incidence of magnesium depletion is probably higher. In fact, magnesium depletion has been described as “the most underdiagnosed electrolyte abnormality in current medical practice” (11).
Predisposing Conditions
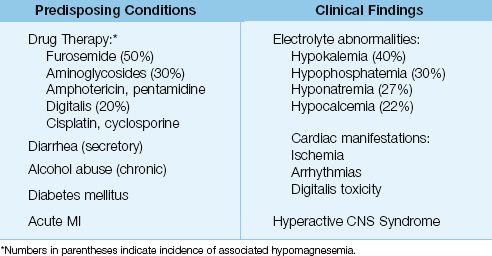
Because serum Mg levels have a limited ability to detect magnesium depletion, recognizing the conditions that predispose to magnesium depletion may be the only clue of an underlying electrolyte imbalance. The common predisposing conditions for magnesium depletion are listed in Table 37.3.
Table 37.3 Markers of Possible Magnesium Depletion
Diuretic Therapy
Diuretics are the leading cause of magnesium deficiency. Diuretic-induced inhibition of sodium reabsorption also interferes with magnesium reabsorption, and the resultant urinary magnesium losses can parallel urinary sodium losses. Urinary magnesium excretion is most pronounced with the loop diuretics (furosemide and ethacrynic acid). Magnesium deficiency has been reported in 50% of patients receiving chronic therapy with furosemide (12). The thiazide diuretics show a similar tendency for magnesium depletion, but only in elderly patients (13). Magnesium depletion does not occur with “potassium-sparing” diuretics (14).
Antibiotic Therapy
The antibiotics that promote magnesium depletion are the aminoglycosides, amphotericin and pentamidine (15,16). The aminoglycosides block magnesium reabsorption in the ascending loop of Henley, and hypomagnesemia has been reported in 30% of patients receiving aminoglycoside therapy (16).
Other Drugs
There are several case reports indicating that prolonged use of proton pump inhibitors (14 days to 13 years) can be associated with severe hypomagnesemia (17), possibly due to diminished magnesium absorption in the GI tract. Other drugs associated with magnesium depletion include digitalis, epinephrine, and the chemotherapeutic agents cisplatin and cyclosporine (15,18). The first two agents shift magnesium into cells, whereas the latter two promote renal magnesium excretion.
Alcohol-Related Illness
Hypomagnesemia is reported in 30% of hospital admissions for alcohol abuse, and in 85% of admissions for delirium tremens (19,20). The magnesium depletion is due to a number of factors, including generalized malnutrition and chronic diarrhea. In addition, there is an association between magnesium deficiency and thiamine deficiency (21). Magnes-ium is required for the transformation of thiamine into thiamine pyrophosphate, so magnesium deficiency can promote thiamine deficiency in the face of adequate thiamine intake. For this reason, the magnesium status should be monitored periodically in patients receiving daily thiamine supplements.
Secretory Diarrhea
Secretions from the lower GI tract are rich in magnesium (10–14 mEq/L) (22), and secretory diarrhea can be accompanied by profound magnesium depletion (20). Upper GI tract secretions are not rich in magnesium (1–2 mEq/L), so vomiting does not pose a risk for magnesium depletion.
Diabetes Mellitus
Magnesium depletion is common in insulin-dependent diabetic patients, probably as a result of urinary Mg losses that accompany glycosuria (23). Hypomagnesemia is reported in only 7% of admissions for diabetic ketoacidosis, but the incidence increases to 50% over the first 12 hours after admission (24), probably as a result of insulin-induced movement of magnesium into cells.
Acute Myocardial Infarction
Hypomagnesemia is reported in as many as 80% of patients with acute myocardial infarction (25). The mechanism is unclear, but may be due to an intracellular shift of Mg from excess catecholamines.
Clinical Manifestations
There are no specific clinical manifestations of magnesium deficiency, but the following clinical findings can suggest an underlying magnesium deficiency.
Other Electrolyte Abnormalities
Magnesium depletion is often accompanied by depletion of potassium, phosphate, and calcium (see Table 37.3) (26).
HYPOKALEMIA: Hypokalemia is reported in 40% of cases of magnesium depletion (26). Furthermore, the hypokalemia that accompanies magnesium depletion can be refractory to potassium replacement therapy, and magnesium replacement is often necessary before the hypokalemia can be corrected (27).
HYPOCALCEMIA: Magnesium depletion can cause hypocalcemia as a result of impaired parathormone release (28) combined with an impaired end-organ response to parathormone (29). As with the hypokalemia, the hypocalcemia from magnesium depletion is difficult to correct unless magnesium deficits are corrected.
HYPOPHOSPHATEMIA: Phosphate depletion is a cause rather than effect of magnesium depletion. The mechanism is enhanced renal magnesium excretion (30).
Arrhythmias
As mentioned earlier, magnesium is required for proper function of the membrane pump on cardiac cell membranes. Magnesium depletion will depolarize cardiac cells and promote tachyarrhythmias. Because both digitalis and magnesium deficiency act to inhibit the membrane pump, magnesium deficiency will magnify the digitalis effect and promote digitalis cardiotoxicity. Intravenous magnesium can suppress digitalis-toxic arrhythmias, even when serum Mg levels are normal (31). Intravenous magnesium can also abolish refractory arrhythmias (i.e., unresponsive to traditional antiarrhythmic agents) in the absence of hypomagnesemia (32). This effect may be due to a membrane-stabilizing effect of magnesium that is unrelated to magnesium repletion.
One of the serious arrhythmias associated with magnesium depletion is torsade de pointes (see Figure 15.8). The role of magnesium in this arrhythmia is discussed in Chapter 15.
Neurologic Findings
The neurologic manifestations of magnesium deficiency include altered mentation, generalized seizures, tremors, and hyperreflexia. All are uncommon, nonspecific, and have little diagnostic value.
A neurologic syndrome described recently that can abate with magnesium therapy deserves mention. The clinical presentation is characterized by ataxia, slurred speech, metabolic acidosis, excessive salivation, diffuse muscle spasms, generalized seizures, and progressive obtundation (33). The clinical features are often brought out by loud noises or bodily contact, and thus the term reactive central nervous system magnesium deficiency has been used to describe this disorder. This syndrome is associated with reduced magnesium levels in cerebrospinal fluid, and it resolves with magnesium infusion. The prevalence of this disorder is unknown at present.
Diagnosis
As mentioned several times, the serum Mg level is an insensitive marker of magnesium depletion. When magnesium depletion is due to nonrenal factors (e.g., diarrhea), the urinary magnesium excretion is a more sensitive test for magnesium depletion (34). However, most cases of magnesium depletion are due to enhanced renal magnesium excretion, so the diagnostic value of urinary magnesium excretion is limited.
Magnesium Retention Test
In the absence of renal magnesium wasting, the urinary excretion of magnesium in response to a magnesium load may be the most sensitive index of total body magnesium stores (35,36). This method is outlined in Table 37.4. The normal rate of magnesium reabsorption is close to the maximum rate (Tmax), so most of an infused magnesium load will be excreted in the urine when magnesium stores are normal. However, when magnesium stores are deficient, the magnesium reabsorption rate is much lower than the Tmax, so more of the infused magnesium will be reabsorbed and less will be excreted in the urine. Magnesium deficiency is likely when less than 50% of the infused Mg is recovered in the urine, and is unlikely when more than 80% of the infused Mg is excreted in the urine. This test can be also be valuable for identifying the end-point of magnesium replacement therapy (see later). It is important to emphasize that this test will be unreliable in patients with impaired renal function or when there is ongoing renal magnesium wasting.
Table 37.4 Renal Magnesium Retention Test

Magnesium Replacement
The magnesium preparations available for oral and parenteral use are listed in Table 37.5 (37,38). The oral preparations can be used for daily maintenance therapy (5 mg/kg in normal subjects). However, because intestinal absorption of oral magnesium is erratic, parenteral magnesium is advised for managing hypomagnesemia.
The standard intravenous magnesium preparation is magnesium sulfate (MgSO4). Each gram of MgSO4 has 8 mEq (4 mmol) of elemental magnesium (4). A 50% magnesium sulfate solution (500 mg/mL) has an osmolarity of 4,000 mosm/L (43), so it must be diluted to a 10% (100 mg/mL) or 20% (200 mg/mL) solution for intravenous use. Ringer’s solutions should not be used as the diluent for MgSO4 because the calcium in Ringer’s solutions will counteract the actions of the infused magnesium.
Table 37.5 Oral and Parenteral Magnesium Preparations

The following magnesium replacement protocols are recommended for patients with normal renal function (39).
Mild, Asymptomatic Hypomagnesemia
The following guidelines can be used for a serum Mg of 1–1.4 mEq/L with no apparent complications (44):
1. Assume a total magnesium deficit of 1–2 mEq/kg.
2. Because 50% of the infused magnesium can be lost in the urine, assume that the total magnesium requirement is twice the magnesium deficit.
3. Replace 1 mEq/kg for the first 24 hours, and 0.5 mEq/kg daily for the next 3–5 days.
Moderate Hypomagnesemia
The following protocol is recommended for a serum Mg <1 mEq/L, or for a low serum Mg that is accompanied by other electrolyte abnormalities:
1. Add 6 g MgSO4 (48 mEq of Mg) to 250 or 500 mL isotonic saline and infuse over 3 hours.
2. Follow with 5 g MgSO4 (40 mEq of Mg) in 250 or 500 mL isotonic saline infused over the next 6 hours.
3. Continue with 5 g MgSO4 every 12 hours (by continuous infusion) for the next 5 days.
Life-Threatening Hypomagnesemia
The following is recommended for hypomagnesemia associated with serious cardiac arrhythmias (e.g., torsade de pointes) or generalized seizures:
1. Infuse 2 g MgSO4 (16 mEq of Mg) intravenously over 2–5 minutes.
2. Follow with 5 g MgSO4 (40 mEq of Mg) in 250 or 500 mL isotonic saline infused over the next 6 hours.
3. Continue with 5 g MgSO4 every 12 hours (by continuous infusion) for the next 5 days.
Monitoring Replacement Therapy
Serum Mg levels will rise after the initial magnesium bolus, but will begin to fall after 15 minutes. Therefore, it is important to follow the bolus dose with a continuous magnesium infusion. Serum Mg levels may normalize after 1 to 2 days, but it will take several days to replenish the total body magnesium stores.
The magnesium retention test in Table 37.4 can be valuable for identifying the end-point of potassium replacement therapy; i.e., magnesium replacement is continued until urinary magnesium excretion is ≥80% of the infused magnesium load.
Hypomagnesemia and Renal Insufficiency
Hypomagnesemia is not common in renal insufficiency but can occur when severe or chronic diarrhea is present and the creatinine clearance is >30 mL/minute. When magnesium is replaced in the setting of renal insufficiency, no more than 50% of the magnesium in the standard replacement protocols should be administered (39), and the serum Mg should be monitored carefully.
MAGNESIUM EXCESS
Magnesium accumulation occurs much less frequently than magnesium depletion. In one survey, hypermagnesemia (i.e., serum Mg >2 mEq/L) was observed in 5% of hospitalized patients (40).
Predisposing Conditions
Renal Insufficiency
Most cases of hypermagnesemia are the result of impaired renal magnesium excretion, which occurs when the creatinine clearance falls below 30 mL/minute (41). However, hypermagnesemia is not a prominent feature of renal insufficiency unless magnesium intake is increased.
Hemolysis
The Mg concentration in erythrocytes is approximately three times greater than in serum (42), so hemolysis can increase the serum Mg. The serum Mg is expected to rise by 0.1 mEq/L for every 250 mL of erythrocytes that lyse completely (46), so hypermagnesemia is expected only with massive hemolysis.
Other Conditions
Other conditions that can predispose to mild hypermagnesemia are diabetic ketoacidosis (transient), adrenal insufficiency, hyperparathyroid-ism, and lithium intoxication (41).
Clinical Features
The clinical consequences of progressive hypermagnesemia are listed be-low (42).
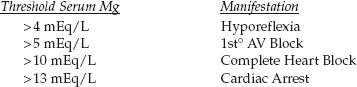
Magnesium has been described as nature’s physiologic calcium blocker (43), and most of the serious consequences of hypermagnesemia are due to calcium antagonism in the cardiovascular system. Most of the cardiovascular depression is the result of cardiac conduction delays. Depressed contractility and vasodilation are not prominent.
Management
Hemodialysis is the treatment of choice for severe hypermagnesemia. Intravenous calcium gluconate (1 g IV over 2 to 3 minutes) can be used to antagonize the cardiovascular effects of hypermagnesemia temporarily, until dialysis is started (44). If fluids are permissible and some renal function is preserved, aggressive volume infusion combined with furosemide may be effective in reducing the serum magnesium levels in less advanced cases of hypermagnesemia.
A FINAL WORD
Magnesium often takes a back seat to sodium and potassium, but as mentioned in the introduction to this chapter, magnesium is required for the release of energy stored in ATP, and is also required for the proper functioning of the membrane Na-K exchange pump that allows the transmission of electrical impulses in excitable tissues. Therefore, magnesium de-serves more attention that it receives.
The following are some specific points about magnesium that warrant emphasis:
1. The serum Mg can be normal in patients who are magnesium depleted.
2. Hypomagnesemia is reported in over 50% of ICU patients, and the frequency of magnesium depletion is likely to be even higher. Diuretic therapy with furosemide is the leading cause of magnesium depletion in ICUs.
3. Magnesium depletion should be suspected in any patient with diuretic-induced hypokalemia, and especially when hypokalemia is refractory to potassium replacement.
4. Magnesium replacement will correct the serum Mg before total body stores of magnesium are replenished. The best indicator of magnesium repletion is the urinary retention test (see Table 37.4)
REFERENCES
Introduction
1. Noronha JL, Matuschak GM. Magnesium in critical illness: metabolism, assessment, and treatment. Intensive Care Med 2002; 28:667–679.
2. Tong GM, Rude RK. Magnesium deficiency in critical illness. J Intensive Care Med 2005; 20:3–17.
3. Martin KJ, Gonzalez EA, Slatpolsky E. Clinical consequences and management of hypomagnesemia. J Am Soc Nephrol 2009; 20:2291–2295.
4. White RE, Hartzell HC. Magnesium ions in cardiac function. Regulator of ion channels and second messengers. Biochem Pharmacol 1989; 38:859–867.
Magnesium Balance
5. Elin RJ. Assessment of magnesium status. Clin Chem 1987; 33:1965–1970.
6. Reinhart RA. Magnesium metabolism. A review with special reference to the relationship between intracellular content and serum levels. Arch Intern Med 1988; 148:2415–2420.
7. Lowenstein FW, Stanton MF. Serum magnesium levels in the United States, 1971-1974. J Am Coll Nutr 1986; 5:399–414.
8. Altura BT, Altura BM. A method for distinguishing ionized, complexed and protein-bound Mg in normal and diseased subjects. Scand J Clin Lab Invest 1994; 217:83–87.
9. Kroll MH, Elin RJ. Relationships between magnesium and protein concentrations in serum. Clin Chem 1985; 31:244–246.
10. Alvarez-Leefmans FJ, Giraldez F, Gamino SM. Intracellular free magnesium in excitable cells: its measurement and its biologic significance. Can J Physiol Pharmacol 1987; 65:915–925.
Magnesuim Depletion
11. Whang R. Magnesium deficiency: pathogenesis, prevalence, and clinical implications. Am J Med 1987; 82:24–29.
12. Dyckner T, Wester PO. Potassium/magnesium depletion in patients with cardiovascular disease. Am J Med 1987; 82:11–17.
13. Hollifield JW. Thiazide treatment of systemic hypertension: effects on serum magnesium and ventricular ectopic activity. Am J Cardiol 1989; 63:22G–25G.
14. Ryan MP. Diuretics and potassium/magnesium depletion. Directions for treatment. Am J Med 1987; 82:38–47.
15. Atsmon J, Dolev E. Drug-induced hypomagnesaemia: scope and management. Drug Safety 2005; 28:763–788.
16. Zaloga GP, Chernow B, Pock A, et al. Hypomagnesemia is a common complication of aminoglycoside therapy. Surg Gynecol Obstet 1984; 158:561–565.
17. Hess MW, Hoenderop JG, Bindeis RJ, Drenth JP. Systematic review: hypomagnesemia induced by proton pump inhibition. Ailement Pharmacol Ther 2012; 36:415–413.
18. Whang R, Oei TO, Watanabe A. Frequency of hypomagnesemia in hospitalized patients receiving digitalis. Arch Intern Med 1985; 145:655–656.
19. Balesteri FJ. Magnesium metabolism in the critically ill. Crit Care Clin 1985; 5:217–226.
20. Martin HE. Clinical magnesium deficiency. Ann N Y Acad Sci 1969; 162: 891–900.
21. Dyckner T, Ek B, Nyhlin H, et al. Aggravation of thiamine deficiency by magnesium depletion. A case report. Acta Med Scand 1985; 218:129–131.
22. Kassirer J, Hricik D, Cohen J. Repairing Body Fluids: Principles and Practice. 1st ed. Philadelphia, PA: WB Saunders, 1989; 118–129.
23. Sjogren A, Floren CH, Nilsson A. Magnesium deficiency in IDDM related to level of glycosylated hemoglobin. Diabetes 1986; 35:459–463.
24. Lau K. Magnesium metabolism: normal and abnormal. In: Arieff AI, DeFronzo RA, eds. Fluids, electrolytes, and acid base disorders. New York, NY: Churchill Livingstone, 1985; 575–623.
25. Abraham AS, Rosenmann D, Kramer M, et al. Magnesium in the prevention of lethal arrhythmias in acute myocardial infarction. Arch Intern Med 1987; 147:753–755.
Clinical Manifestations
26. Whang R, Oei TO, Aikawa JK, et al. Predictors of clinical hypomagnesemia. Hypokalemia, hypophosphatemia, hyponatremia, and hypocalcemia. Arch Intern Med 1984; 144:1794–1796.
27. Whang R, Flink EB, Dyckner T, et al. Magnesium depletion as a cause of refractory potassium repletion. Arch Intern Med 1985; 145:1686–1689.
28. Anast CS, Winnacker JL, Forte LR, et al. Impaired release of parathyroid hormone in magnesium deficiency. J Clin Endocrinol Metab 1976; 42:707–717.
29. Rude RK, Oldham SB, Singer FR. Functional hypoparathyroidism and parathyroid hormone end-organ resistance in human magnesium deficiency. Clin Endocrinol 1976; 5:209–224.
30. Dominguez JH, Gray RW, Lemann J, Jr. Dietary phosphate deprivation in women and men: effects on mineral and acid balances, parathyroid hormone and the metabolism of 25-OH-vitamin D. J Clin Endocrinol Metab 1976; 43:1056–1068.
31. Cohen L, Kitzes R. Magnesium sulfate and digitalis-toxic arrhythmias. JAMA 1983; 249:2808–2810.
32. Tzivoni D, Keren A. Suppression of ventricular arrhythmias by magnesium. Am J Cardiol 1990; 65:1397–1399.
33. Langley WF, Mann D. Central nervous system magnesium deficiency. Arch Intern Med 1991; 151:593–596.
34. Fleming CR, George L, Stoner GL, et al. The importance of urinary magnesium values in patients with gut failure. Mayo Clin Proc 1996; 71:21–24.
35. Clague JE, Edwards RH, Jackson MJ. Intravenous magnesium loading in chronic fatigue syndrome. Lancet 1992; 340:124–125.
36. Hebert P, Mehta N, Wang J, et al. Functional magnesium deficiency in critically ill patients identified using a magnesium-loading test. Crit Care Med 1997; 25:749–755.
Magnesium Replacement Therapy
37. DiPalma JR. Magnesium replacement therapy. Am Fam Physician 1990; 42:173–176.
38. Trissel LA. Handbook on injectable drugs. 13th ed. Bethesda, MD: Amer Soc Health System Pharmcists, 2005.
39. Oster JR, Epstein M. Management of magnesium depletion. Am J Nephrol 1988; 8:349–354.
Magnesium Excess
40. Whang R, Ryder KW. Frequency of hypomagnesemia and hypermagnesemia. Requested vs routine. JAMA 1990; 263:3063–3064.
41. Van Hook JW. Hypermagnesemia. Crit Care Clin 1991; 7:215–223.
42. Elin RJ. Magnesium metabolism in health and disease. Dis Mon 1988; 34:161–218.
43. Iseri LT, French JH. Magnesium: nature’s physiologic calcium blocker. Am Heart J 1984; 108:188–193.
44. Mordes JP, Wacker WE. Excess magnesium. Pharmacol Rev 1977; 29:273–300.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








