Pulmonary Thromboembolic Disease
Raffi Calikyan MD
Venous thromboembolic disease encompasses two closely linked conditions: deep vein thrombosis (DVT) and pulmonary embolism (PE). DVT and PE are significant causes of morbidity and mortality in the United States and in other Western countries. Although the precise incidence in the general population is difficult to determine, it is estimated that venous thromboembolism accounts for approximately 300,000 hospitalizations and as many as 50,000 deaths per year (Anderson & Wheeler, 1991).
Although PE is a frequent, potentially fatal disease, it remains largely underdiagnosed, and the mortality rate has not significantly changed since the 1960s, despite new diagnostic and therapeutic modalities. Efforts to improve this situation should focus on recognizing patients at risk and taking appropriate preventive measures.
ANATOMY, PHYSIOLOGY, AND PATHOLOGY
Anatomy and Physiology
The venous system of the lower extremities can be divided into the superficial and deep vein groups. The superficial venous system includes the greater and lesser saphenous veins and their tributaries. Perforating veins connect the deep and superficial venous systems. The deep veins found in the calf flow into the popliteal vein, which continues into the femoral vein. The femoral vein connects to the iliac venous system and then into the inferior vena cava. The inferior vena cava flows into the right atrium. From the right ventricle the pulmonary artery takes the blood flow into the lungs. Blood supply to the lungs is provided by the pulmonary circulation and by the systemic circulation via the bronchial arteries.
CLINICAL PEARL
The superficial femoral vein is part of the deep venous system.
The venous flow in the lower extremities is directed from the superficial to the deep veins. Bicuspid valves in the veins of the lower extremity direct the flow centrally. The flow from the periphery into the right side of the heart is aided by the contraction of the skeletal muscles, compressing the veins in the extremities. The greater veins are helped by the negative intrathoracic pressure generated during inspiration.
The normal pulmonary arterial tree consists of large elastic arteries, smaller muscular pulmonary arteries, arterioles, and capillaries. The elastic capacity of the pulmonary circulation allows accommodation of increases in blood flow without increasing pulmonary artery pressures (normal range 25/10 mmHg). However, if a significant obstruction of the pulmonary vascular bed occurs (generally >30%), an abrupt increase in the pulmonary artery pressure results; this may be poorly tolerated by the right ventricle, leading to acute decompensation (Elliott, 1992).
Pathology
DEEP VEIN THROMBOSIS
More than 90% of clinically significant PEs arise from thrombosis of veins in the lower extremities. Thrombi can form in the calf veins, popliteal vein, and more proximal veins of the iliofemoral venous system. In the 19th century Virchow defined the pathophysiologic factors promoting the formation of a thrombus: stasis, abnormalities of the vessel wall, and hypercoagulability. Risk factors for venous thromboembolism are based on these conditions.
DVT usually develops in proximity to the venous valves. Initially a “white thrombus” is formed by platelet aggregation, followed by a “red thrombus” with fibrin deposition. The thrombus propagates by continued fibrin and platelet accretion. Subsequently, it either resolves within hours to days by the fibrinolytic system or undergoes organization with re-endothelization. If there is re-endothelization, the venous lumen narrows and distal venous valves become incompetent because of increased intraluminal pressure. Venous flow is directed into the superficial venous system during leg muscle contraction, leading to edema and impaired viability of subcutaneous tissues, and in the most severe cases to ulceration (post-thrombotic syndrome).
Within 7 to 10 days, the actions of fibrinolysis or organization reach a stable state. Thus, the embolic risk is highest within the first few days of thrombus formation (Hirsh & Hoak, 1996).
PULMONARY EMBOLISM
If the whole thrombus or any portion of it dislodges before it organizes, it may be transported in the venous system to the right side of the heart and into the pulmonary circulation. The acute obstruction of pulmonary vessels by an embolus leads to hemodynamic and respiratory consequences. The most important hemodynamic complication is the acute increase in pulmonary vascular resistance, which in extreme cases can lead to acute right-sided heart failure and death. In addition to mechanical obstruction, vasoconstriction occurs as a result of the release of vasoactive amines (serotonin, thromboxane A2) and the stimulation of baroreceptors in the pulmonary artery wall.
A major respiratory consequence of PE is increased alveolar dead space, because ventilation continues in lung areas that are not perfused. Both increased dead space and intrapulmonary or
intracardiac right-to-left shunting of blood lead to hypoxemia. Tachypnea is caused by the stimulation of juxtacapillary irritant receptors. Release of serotonin may also cause bronchoconstriction, accounting for the wheezing sometimes detected in patients with PE.
intracardiac right-to-left shunting of blood lead to hypoxemia. Tachypnea is caused by the stimulation of juxtacapillary irritant receptors. Release of serotonin may also cause bronchoconstriction, accounting for the wheezing sometimes detected in patients with PE.
Over several hours, atelectasis may develop as a result of loss of surfactant distal to the occlusion. Development of pulmonary infarction is uncommon because of the dual blood supply of the lungs. Infarction occurs in less than 10% of cases, usually only if concomitant heart or lung disease is present (eg, left ventricular failure, mitral stenosis, chronic obstructive lung disease).
After the acute phase, most emboli undergo resolution by fibrinolysis. Some thrombi fail to resolve, presumably as a result of defects in the intrinsic fibrinolytic system or because they were very well organized before embolization (Elliot, 1992).
EPIDEMIOLOGY
Venous thromboembolism is relatively rare in the general population in the absence of predisposing conditions. Numerous factors are associated with an increased risk for venous thromboembolism, including:
Age older than 40 years
Prior history of venous thromboembolism
Major surgery, especially orthopedic surgery of the lower extremities
Bed rest in excess of 5 days
Malignancy
Fracture of the pelvis, hip, or long bones
Paralytic stroke
Estrogen treatment, such as high-dose oral contraceptives
Pregnancy and the puerperium
Hypercoagulability states (lupus anticoagulant, protein C deficiency, protein S deficiency, antithrombin III deficiency, plasminogen deficiency)
Obesity
Congestive heart failure
Myocardial infarction (Anderson & Wheeler, 1995).
There are some data to suggest that the incidence of venous thromboembolism is higher in African Americans than whites. Asians have the lowest incidence, which may be explained by genetic differences in the control of the coagulation system (Fujimura et al, 1995).
DIAGNOSTIC CRITERIA
Imaging studies establish the diagnosis of DVT or PE. Doppler ultrasound of the legs and nuclear studies of the lungs demonstrate alterations of venous compressibility and lung perfusion, respectively, providing indirect evidence of the presence of a clot. Thrombosis can be demonstrated directly by venography of the lower extremities or by a pulmonary angiogram of the pulmonary arteries.
HISTORY AND PHYSICAL EXAM
In the patient with suspected DVT or PE, the history should focus on risk factors. The most important risk factors are:
Prior history of thromboembolic disease
Recent immobilization or long-distance travel
Estrogen use
Family history of thromboembolic disease or hypercoagulable states
Recent trauma.
The most common complaint of the patient with DVT is calf pain. DVT is suggested by unilateral leg swelling, warmth, or erythema. There may be tenderness or increased tissue turgor along the course of the involved vein. Increased resistance or pain during dorsiflexion of the foot (Homan’s sign) is an unreliable diagnostic test.
The symptoms of the patient with PE are often nonspecific. Dyspnea and pleuritic chest pain are the most usual symptoms, although apprehension and cough may also be present. Hemoptysis and syncope occur in less than one third of the patients. On the physical exam, the most common presenting signs are tachypnea and tachycardia. The pulmonary component of the second heart sound may be accentuated. In massive PE, signs of acute right ventricular failure, as well as hypotension and cyanosis, are encountered.
DIAGNOSTIC STUDIES
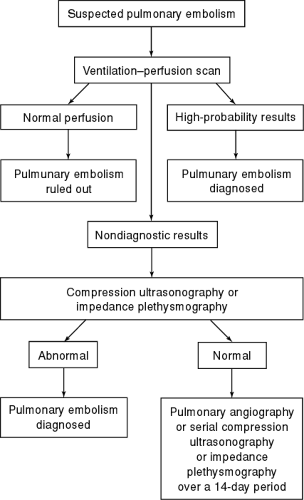
An algorithmic approach to the diagnostic workup for DVT and PE is illustrated in Figures 77-1 and 77-2.
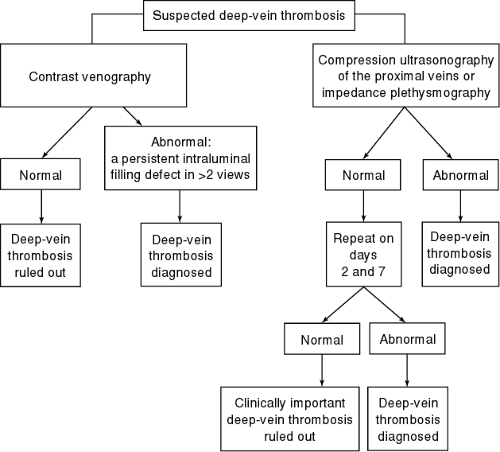 FIGURE 77-1 An approach to patients with clinically suspected deep-vein thrombosis. ( J.S. Ginsberg. (1996). Management of venus thromboembolism. New England Journal of Medicine, 335(24), 1816–1828. ) |
Deep Vein Thrombosis
Routine diagnostic tests available for evaluation of DVT include Doppler ultrasonography, impedance plethysmography (IPG), and contrast venography.
Doppler ultrasonography of the lower extremities is highly sensitive (>90%) for detecting clots in the popliteal and femoral veins but is less sensitive for detecting calf vein thrombosis (about 50%). It is a preferred method because it is noninvasive and can be performed at the bedside.
IPG is a noninvasive test that can detect the changes in blood volume resulting from delayed venous emptying, which occurs if a thrombus is obstructing proximal veins. However, the test is limited in obese or edematous patients, and false-positive results can be seen because of the lack of discrimination between intravascular obstruction and extravascular compression. The sensitivity of IPG is low for nonobstructing or infrapopliteal thrombi. Because of these limitations, ultrasound is a more accurate test for suspected DVT (Burke et al, 1995).
Contrast venography is considered the gold standard for the diagnosis of DVT. However, it is not used as a first-line diagnostic tool because of patient discomfort during venous cannulation and because of the risk of a reaction to the contrast material. Venography is used only if accurate information cannot be obtained with noninvasive tests.
Magnetic resonance imaging can be used to evaluate the pelvic and lower extremity veins. Because of its expense, its use as a first-line test is limited mostly to suspected disease in the upper extremities.
Blood tests for the diagnosis of DVT have not yet been widely adopted. Measurement of plasma levels of D-dimer, a specific product of fibrinolysis, has been used as a complementary test with IPG or ultrasound. If the level of D-dimer is below a certain cutoff point (usually 500 mg/L) and an imaging study is negative, the negative predictive value of the combination is 98.5% (Ginsberg et al, 1997; Bounameux et al, 1994). D-dimer
has also been investigated as a diagnostic tool for PE. Its sensitivity for the diagnosis of PE is 83% to 96.8%, but the specificity is low (45.1% to 68%). This precludes the use of the assay as a screening tool for patients with suspected PE, but it may be useful for patients with inconclusive nuclear scans.
has also been investigated as a diagnostic tool for PE. Its sensitivity for the diagnosis of PE is 83% to 96.8%, but the specificity is low (45.1% to 68%). This precludes the use of the assay as a screening tool for patients with suspected PE, but it may be useful for patients with inconclusive nuclear scans.