Fig. 18.1
Symptoms and signs of acute respiratory failure
Age-related changes of the cardiorespiratory system should be taken into account in the assessment and management of dyspnoea. Figure 18.2 shows the main changes secondary to ageing in the pulmonary, cardiovascular and central nervous systems. Changes due to ageing in both the pulmonary and cardiovascular systems make older people more vulnerable to significant acute injury and less likely to present with the classical symptoms and signs of dyspnoea, which are associated with a worse prognosis.
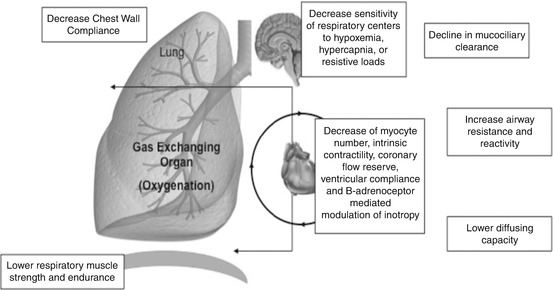

Fig. 18.2
Age-related changes of the cardiorespiratory system
18.3 Aetiology
Dyspnoea is the main clinical presentation of cardiac and respiratory disorders, although many other potential aetiologies may also be present [4, 5]. Dyspnoea is caused by chest wall and pleural abnormalities, lung and airway disease, acute heart failure (AHF) or central and peripheral nervous system condition alterations. In the EPIDASA study, AHF (43%), pneumonia (35%), COPD exacerbations (32%) and pulmonary embolism (PE) (18%) were the main causes of dyspnoea in older patients, being associated with a high mortality [3]. Acute dyspnoea in the older is more likely to be due to multiple causes (50% had more than two diagnoses), including non-cardiopulmonary factors (i.e. metabolic acidosis, acute intra-abdominal or renal pathology and sepsis) and therefore require a more extensive work-up [3].
18.4 Diagnosis
Dyspnoea is due to multiple causes and the diagnostic work-up of emergency physicians involves wide differential diagnoses (Table 18.1). Diagnosis by emergency physicians is even more complex and less accurate in older patients [3]. The appropriate initial diagnosis and treatment of patients attended for dyspnoea in the ED are associated with shorter hospital and intensive care unit (ICU) stay and a better short-term prognosis [3].
Table 18.1
Aetiology of dyspnoea in older patients in the emergency departmenta
Lung and airway disease |
• Acute exacerbation of COPD |
• Pneumonia |
• Acute asthma |
• Upper airway obstruction |
• Lung cancer |
• Pulmonary fibrosis |
• Bronchiectasis |
• Tuberculosis |
Acute heart failure |
• Ischaemic disease |
• Heart valve disease |
• Arrhythmia |
Vessels |
• Hypertension |
• Pulmonary embolism |
Chest wall and pleural abnormalities |
• Kyphoscoliosis |
• Chest wall trauma |
• Pleural effusion |
• Pneumothorax |
• Muscle abnormalities |
Central and peripheral nervous system |
• Drugs: Sedatives |
• CNS: Stroke, trauma, etc. |
• PNS: Guillain-Barre syndrome, myelitis, tetanus, spinal cord trauma, amyotrophic lateral sclerosis, myasthenia, botulism, etc. |
Other |
• Shock |
• Anaemia |
The differential diagnosis of dyspnoea in older patients attended in the ED is usually achieved according to the clinical history, physical examination, 12-lead electrocardiogram, chest X-ray and laboratory tests. Further tests such as determination of biomarkers, arterial or venous blood gas, ultrasound, computed tomography, ventilation/perfusion scan or other tests should be performed taking into account previous findings. The diagnosis of pneumonia is based on chest X-rays [6], while the diagnosis of heart failure is based on electrocardiogram, chest radiographs and natriuretic peptide type B values [7], and the diagnosis of pulmonary embolism is based on prediction rules (Wells rule and revised Geneva score), D-dimer (age-adjusted cut-offs) and computerized tomography [8]. There are no definitive diagnostic or imaging tests to diagnose an exacerbation of COPD or asthma. Moreover, asthma present in the most advanced ages is often diagnosed as COPD, thereby leading to under or inappropriate treatment [9].
18.4.1 Symptoms and Signs
Regarding dyspnoea, it is important to ascertain the time of presentation, precipitating factors and additional treatment given, the baseline functional status and exercise tolerance, the clinical history (comorbidities, tobacco consumption, use of home oxygen, number of previous hospital admissions or ED visits, and ventilation in an ICU) as well as the usual medical treatment.
Questions about associated symptoms such as chest pain, palpitations, diaphoresis, paroxysmal nocturnal dyspnoea, orthopnoea, leg swelling, fever, cough and changes in sputum or haemoptysis should also be considered. The older tends to present more atypical (i.e. confusion, fall, functional impairment or decompensation of chronic disease) and fewer typical symptoms (i.e. paroxysmal nocturnal dyspnoea in heart failure, chest pain in myocardial infarction (MI), fever, cough, sputum or dyspnoea in pneumonia) compared to younger adults.
Physical examination has a twofold objective. The first is to identify high-risk patients in the initial assessment even before obtaining the clinical history. Severe respiratory distress includes the inability to complete sentences in one breath or lie supine, the use of accessory muscles, agitation or other altered mental status, diaphoresis, hypotension, tachypnoea, tachycardia and silent chest. Signs of imminent respiratory failure include confusion or coma, cyanosis and feeble respiratory effort. The level of consciousness or vital signs in older people may be altered by age-related changes, comorbidities or medications. Elevated jugular venous pressure and pulsus paradoxus can occur in some conditions that compromise right heart filling. The second objective of the physical examination is to provide essential elements for differential diagnosis, although no finding on physical examination can rule out a significant condition. Cardiac and pulmonary auscultation may reveal abnormal (inspiratory or expiratory stridor, wheezing or rales) or diminished breath or heart sounds and murmurs. On physical examination of older patients, there is a greater probability of detecting less specific (i.e. rales, oedema) and reproducible (i.e. elevated jugular venous pressure) signs and a lesser probability of identifying specific (i.e. third heart sound) signs [10–12].
The presence of breathlessness, fatigue or tiredness increased time to recover after exercise or confusion, and a past history of chronic heart failure or coronary disease suggest an episode of acute heart failure. Silent acute MI is well recognized in older people, especially in those over 85 years old. Acute presentation of dyspnoea, especially if accompanied by tachypnoea, tachycardia, pleuritic chest pain, haemoptysis or syncope, with recent immobilization (i.e. bed rest, recent trauma or surgery or recent hospitalization) or hypercoagulability (i.e. cancer), indicates the possible presence of a pulmonary embolism. Asthma and COPD exacerbations are frequently accompanied by cough, tachypnoea, intermittent chest pain, sputum production, wheezing and decreased or absent breath sounds. The most complex task is to distinguish between asthma and COPD [9]. The presentation of pneumonia is usually more subtle. It is important to consider spontaneous pneumothorax or pulmonary embolism in patients showing a sudden deterioration in the ED, especially in those with underlying asthma and COPD.
18.4.2 Electrocardiogram
The electrocardiogram may show new features suggestive of acute cardiac ischaemia (ST segment or T wave), pulmonary embolism (S1Q3T3 and right ventricle overload) as well as other conditions.
18.4.3 Chest Radiography
A chest X-ray is usually diagnostic of pneumonia (infiltrate), pneumothorax (air in pleural space) and AHF (cardiomegaly, redistribution of pulmonary vessels, Kerley lines, peribronchial cuffing, consolidation, cotton wool appearance, pleural effusion). Chest X-ray has showed a low sensitivity and negative predictive value in identifying pulmonary opacities. A chest X-ray may be unremarkable in many patients with mildly or moderately severe COPD, in most patients with asthma, in almost 20% patients with heart failure and in 30% of patients with pneumonia, being more frequent in the early phase or when accompanied by dehydration or neutropenia. Thus, on suspicion of pneumonia the radiography should be repeated at 24–48 h, or a computerized tomography should be performed. Rib fractures may be found in older patients with an altered mental status or a history of falls.
18.4.4 Laboratory Tests
In reference to routine laboratory test immediately performed in ED, it is recommended to request a blood count and biochemistry including electrolytes, renal and liver function tests and thyroid hormones. Inadequate inflammatory response as a consequence of immunosenescence may condition C-reactive protein (CRP) and leukocyte results, underestimating the severity of the infectious process.
18.4.5 Biomarkers
Troponin, natriuretic peptides, procalcitonin and D-dimer are markers, which can significantly contribute to the aetiologic diagnosis of an acute dyspnoea. Troponin, natriuretic peptides and procalcitonin (PCT) in conjunction with lactate are also prognostic markers, and these parameters are valuable for stratifying disease severity according to initial values and plasma kinetics during the clinical course of the disease.
Troponin is a marker of myocardial damage and may be useful for the aetiologic diagnosis of an acute coronary event but also in the short-term prognostic stratification of AHF and PE [13]. Serial measurements of cardiac biomarkers are necessary to rule out an acute coronary syndrome. Nonetheless, troponin has limited specificity and may be elevated in other conditions (i.e. sepsis).
D-dimer values may be obtained in cases with a low to intermediate risk of PE (i.e. Wells or Geneva criteria) since a negative D-Dimer value allows PE to be ruled out. D-dimer values are more likely to be falsely increased in older patients (sensitivity of ELISA D-dimer tests is around 100% irrespective of age; however the specificity is lower than 15% in older people with frailty because of multiple underlying comorbidities). Recent evidence suggests using age-adjusted cut-offs to improve the performance of D-dimer testing in older people [8, 14].
The addition of a single natriuretic peptide type B measurement improves the diagnostic accuracy and prognosis of AHF in the ED compared to standard clinical judgement in ED patients with dyspnoea [15]. BNP and NT-proBNP are both accurate in the diagnosis of AHF and are recommended to exclude cardiac causes of dyspnoea [15]. It is known that these values rise with age, sex, renal function, nutritional state and in other associated acute conditions (atrial fibrillation, pulmonary thromboembolism, sepsis, anaemia, ischaemic heart disease, myocardiopathies, etc.) [16]. In general, it is considered that BNP < 100 pg/mL and NT-proBNP < 300 pg/mL almost certainly rule out the diagnosis of AHF. To the contrary, from a diagnostic point of view, different cut-offs of NT-proBNP have been described based on the age group (<50 years, 450 pg/mL; 50–75 years, 900 pg/mL; >75 years, 1.800 pg/mL) [15]. With regard to the prognostic value of NT-proBNP, it is known that a cut-off of ≥5.180 pg/mL is associated with 30-day mortality in the older population attended for AHF in a hospital ED, regardless of the presence of other factors which may influence interpretation such as the severity of the acute episode or a reduction in glomerular filtration (Martin-Sanchez et al. 2012).
PCT is the prehormone of calcitonin, which is normally secreted by the thyroid gland in response to hypercalcemia and is induced by bacterial infection. Elevated PCT concentrations (higher than 0.25 ng/mL) may indicate an infectious aetiology of dyspnoea [17]. In suspected lower respiratory tract infections (CAP or COPD exacerbation), determination of PCT concentrations was useful as a guide to reduce the total antibiotic exposure and antibiotic prescriptions on admission [4, 5]. The BACH trial showed that patients with a diagnosis of AHF and elevated PCT concentrations had a worse outcome if not treated with antibiotics. PCT may aid in the decision to administer antibiotics to patients presenting for AHF and in whom there is clinical uncertainty regarding the involvement of a superimposed bacterial infection [18].
The panel of markers (troponin I, myoglobin, creatinine kinase-myocardial band isoenzyme, D-dimer, and B-type natriuretic peptide) did not improve the area under the ROC curve for diagnosing the combined set of clinical conditions (AHF, PE, MI) [19].
18.4.6 Arterial Blood Gas
Oxygenation can usually be measured using transcutaneous pulse oximetry, but in some cases (delirium, hypothermia, hypoperfusion or carbon monoxide poisoning) it can be more difficult. Arterial blood gas allows the assessment of oxygen saturation and CO2 retention. Only PaO2 and SaO2 changed clinically significantly with increasing age, whereas the other analyses were virtually independent of age [20].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





