A recent study showed that emergent gastrointestinal surgical procedures in older patients did not differ from those in younger patients. However, the morbidity and mortality rates were higher in older patients. Assessing comorbidities, frailty, the diagnosis, and the patient’s wishes and status should help in the decision-making for emergency surgical management [23]. Overall mortality approaches 20–25% in older patients who underwent emergent abdominal surgery. The American Society of Anesthesiologists (ASA) grading, interval from onset of symptoms to admission, presence of mesenteric infarction, palliative bypass, and nontherapeutic laparotomy are predictors of mortality [25]. Twenty-five percent of older patients who have undergone emergency abdominal surgery are faced with local morbidity, and 30% of them are faced with systemic morbidity. The leading local morbidity is wound infection, whereas the leading systemic morbidity is respiratory failure [25].
Another important point is to manage older patients with AAP in a multidisciplinary approach. Recent data have confirmed that multidisciplinary care improves outcomes for older surgical patients [23, 26]. Diagnostic and therapeutic decisions should involve EPs, surgeons, anesthesiologists, and geriatricians. It is recommended that protocol-driven integrating pathways can guide care of older patients effectively, but the critical decisions should be individualized for each patient [23, 26].
15.4 Specific Causes
15.4.1 Acute Mesenteric Ischemia
Acute mesenteric ischemia (AMI) is a disease of older patients with a rate of 0.1–2/1000 acute hospital admissions [27]. It is mostly seen in patients over 70 years old and is more common than abdominal aneurysm in patients older than 75 years of age [28]. Arterial embolism and thrombus are the two most common causes of AMI. Women are affected slightly more frequently, but exact gender ratio differs according to the etiology of AMI (Table 15.1) [29]. Despite many advances in the diagnosis and treatment of AMI in the last 3–4 decades, prognosis is still poor. In-hospital mortality rate ranges from 59 to 93% [29, 30].
Table 15.1
Incidence, mortality, and demographics of patients according to the cause of acute mesenteric ischemia [29]
Etiology | Incidence (%) | Mean mortality (%) | Mean age | Female/male ratio |
|---|---|---|---|---|
Arterial embolism | 34 | 66–71 | 69 | 1.23 |
Arterial thrombus | 34 | 70–87 | 71 | 1.46 |
Mesenteric venous thrombosis | 13 | 44 | 70 | 0.78 |
Non-occlusive mesenteric ischemia | 19 | 70–80 | 69 | 1.17 |
Overall | 64–74 |
AMI can be classified into two main groups: thrombotic and non-thrombotic. Thrombotic AMI can be a result of arterial embolism, thrombosis, and mesenteric venous thrombosis (MVT). Non-thrombotic AMI, mainly named as non-occlusive mesenteric ischemia (NOMI) and secondary to low flow states such as cardiogenic shock, sepsis, and hypovolemia, is primarily seen in patients hospitalized in intensive care units [29–32]. Decreased cardiac output causes diffuse mesenteric vasoconstriction leading to ischemia and necrosis.
Arterial embolism is the most common cause of AMI. Onset is usually rapid. Since superior mesenteric artery (SMA) emerges from the aorta with an acute angle, emboli have an affinity for SMA. Most emboli arise from cardiac origin. Arterial thrombosis has the worst prognosis because it involves the origin of the SMA, and the onset is more insidious. MVT accounts for 10% of AAP. Ninety percent of MVTs have an identifiable cause (Table 15.2) and may be acute or chronic.
Table 15.2
Conditions that predispose to mesenteric venous thrombosis
• Hypercoagulable states (antithrombin III deficiency, protein C/S deficiency, factor V Leiden, PCV, etc.) |
• Direct injury or inflammation (surgery, trauma, pancreatitis, cholecystitis, etc.) |
• Venous stasis (Budd-Chiari, cirrhosis, postsplenectomy, etc.) |
Whatever the cause, the result is a breakdown of mucosal barrier, bacterial translocation, migration of polymorphonuclear leukocytes, and activation of inflammatory pathways. Reperfusion can cause ischemia perfusion injury and worsen the condition. Systemic inflammatory response syndrome, disseminated intravascular coagulation, sepsis, and multiorgan failure are the final expression in these patients.
The most important step in the diagnosis of AMI is clinical suspicion in older patients with chief complaint of AAP. Some risk factors must be recorded such as atrial fibrillation, history of coronary artery disease and myocardial infarction, recent cardiac or vascular surgery, prior embolism, chronic mesenteric ischemia symptoms, history of prior deep venous thrombosis, hypercoagulable states, etc. Only 33% of patients had been suspected of AMI before they died of mesenteric ischemia [29–32].
AAP is the most common symptom of AMI. At the beginning, ischemia causes cramping abdominal pain, but tenderness is minimal. When intestinal necrosis develops, diffuse peritonitis appears with a rigid abdomen and sepsis (Fig. 15.2a). Bowel sounds are usually absent but may be present at the beginning. Depending on the cause, other symptoms such as vomiting, diarrhea, distention, and blood in stool may be present. Older patients may have less frequent abdominal pain and more often symptoms such as tachypnea and mental status changes [29–31].
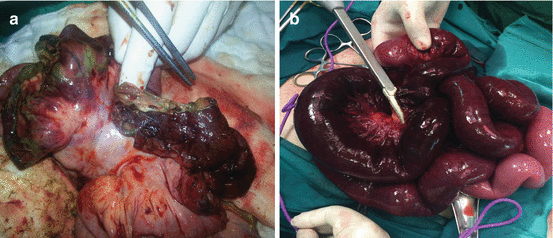

Fig. 15.2
(a) Acute mesenteric ischemia resulted in diffuse necrosis and perforation in jejunal segments. (b) Mesenteric venous thrombosis causing necrosis, which required surgical treatment
Laboratory findings are increased WBC (usually > 15,000/mm3), D-dimer, lactate levels, and metabolic acidosis is observed. Metabolic alkalosis may also be seen because of vomiting. However, sensitivity and specificity of those markers range from 38–96% to 40–84%, respectively. Intestinal fatty acid binding protein has promising results with 90% sensitivity and 89% specificity. Patients may have elevated amylase levels, abnormal liver enzymes, and hyperphosphatemia. Electrocardiography may reveal arrhythmia [29–32].
Pneumatosis intestinalis , free air, air-fluid levels, portal venous gas, thumb printing, or thickening of the bowel wall may be seen on abdominal X-ray, but 25% of patients with AMI have normal findings. Although in classical textbooks, conventional angiography is still accepted as the gold standard for diagnosing AMI, CT angiography has replaced it with a sensitivity and specificity of 94 and 95%. In patients with peritoneal signs, early surgery is indicated because these signs reflect presence of infarction [30, 31]. However, conventional mesenteric angiography may still be the first choice in NOMI [27].
Aggressive volume resuscitation, urinary catheterization, nasogastric decompression, blood sampling for CBC, renal function test, liver function tests, arterial blood gas analysis, and crossmatch should be done. The patient must be stabilized as much as possible before the transfer to the operating room. EPs should consider broad spectrum antibiotic treatment and first dose of anticoagulation therapy in the ED.
Revascularization, assessment of intestinal viability, and resection of necrotic bowel are the main principles of the surgical treatment. The first 6 h of management is critical to decrease the mortality. MVT and NOMI are usually treated nonsurgically. Surgical treatment is indicated in patients with bowel necrosis occurring in 20% of MVT (Fig. 15.2b). Treatment of underlying cause is the mainstay therapy of NOMI. Papaverine infusion via the intra-arterial catheter may be beneficial in resolving vasoconstriction without the need for surgery.
15.4.2 Abdominal Aortic Aneurysm
Abdominal aortic aneurysm is defined by an aortic diameter greater than 3 cm. Prevalence of AAA varies between 4 and 9% in males and 0.2 and 2% in females [33]. Risk factors include old age, male gender, smoking, positive family history, high height, coronary artery disease, hypercholesterolemia, hypertension, and atherosclerosis [33].
AAA is usually asymptomatic. It is frequently found in imaging studies in the ED or in outpatient clinics. However, sometimes patients go to the ED with AAP related to life-threatening rupture. About one half of the patients with aneurysm rupture cannot reach the ED following the rupture. Hypertension, smoking, female gender, larger diameter of the aneurysm, growth rate of the aneurysm, and wall tension of the aneurysm are risk factors for AAA rupture [33, 34].
Older patients with AAA can present with sudden, severe, and unchanging back or abdominal pain depending on the growth rate of the aneurysm. Abdominal pain can be unclear in such patients, and nonspecific symptoms such as syncope, paralysis in the lower extremities, and sensation of fullness in flanks can be the presenting symptoms. Patients who have atypical clinical expression can be misdiagnosed as renal colic, constipation, gas distension, diverticulitis, or NSAP.
Ultrasonography is the first choice imaging study for AAA. Sensitivity of USG approaches 95% and its specificity is 100% for the diagnosis of AAA. Detection of the intra-abdominal free fluid and hemorrhage at bedside are additional advantages in older patients with suspicion of rupture [35].
The second imaging study to be preferred for the diagnosis of AAA or its rupture is CT scan with a sensitivity of approximately 100%. Furthermore, CT scan provides valuable information about the diameter of the aorta, aortic area affected from the aneurism, visceral artery involvement, retroperitoneal bleeding, and renal abnormalities [33]. Detailed information about the aorta and its branches can be obtained with CT angiography, which can provide all information required for the treatment plan of AAA [36].
15.4.3 Intestinal Obstruction
Intestinal obstruction may affect the bowel or the colon. Adhesions as a result of previous surgery and hernias are the most common cause of intestinal obstruction, whereas malignancy is most common in colonic obstruction. Other causes include gallstone ileus, sigmoid volvulus, and diverticulitis. The gallstone ileus is unique to the older population and can cause intestinal obstruction in 20% of patients older than 65 years with a high rate of mortality [16]. Patients usually present with classical findings of intestinal obstruction, air in the biliary tree, and calculus on plain abdominal radiographs [32].
The other signs are colicky abdominal pain with nausea and vomiting. Colonic obstructions may be more insidious in onset [16]. Obstruction in any localization may be complete or incomplete depending on the degree of obstruction. Physical examination usually reveals altered bowel sounds, signs of hypovolemia, distention, and tenderness. Peritoneal signs may be present if necrosis or perforation has developed.
Imaging which shows distended small bowel loops, air-fluid levels, and absence of air in the large bowel should indicate intestinal obstruction. The dilated large bowel with haustral markings is the classical finding of colonic obstruction [16, 32].
The first step in the management of obstruction is the evaluation of vital signs and hydration status. Intravenous hydration, nasogastric decompression, correction of any electrolyte abnormality, urinary catheterization, and diagnostic work-up must be done quickly in the ED. Definitive treatment depends on clinical presentation. If peritoneal findings are present, urgent laparotomy may be necessary. Otherwise, patients may be treated conservatively, and the decision will be made depending on the findings in the follow-up period.
15.4.4 Acute Diverticulitis
Diverticulosis is seen in 10% of the population younger than 40 years of age. Incidence increases with age; 70% of patients older than 80 years have a diverticulosis. The most commonly affected side of the colon is the sigmoid region. About 10–30% of the patients with diverticulosis may have painful diverticular disease or diverticulitis [16, 37].
Painful diverticular disease is characterized by attacks of abdominal pain without inflammatory findings. When fever, leukocytosis, and peritoneal irritation signs are present, it is called diverticulitis, which is associated with microperforation in one of the diverticula. Beyond microperforation, patients may have diverticular perforation causing localized or generalized peritonitis, or abscess [32].
Depending on the extension of the inflammation, clinical signs and management may differ from oral antibiotics in outpatient clinics to urgent laparotomy and supportive care in the intensive care unit. Diagnosis can be made by history and clinical findings. A CT scan may be necessary to make diagnosis and determine the extension of the inflammation. Colonoscopy and barium enema should be avoided in the acute phase of the disease [32].
15.4.5 Acute Appendicitis
Older patients with acute appendicitis (AA) present frequently to the ED with generalized pain, rigidity, hypoactive bowel sounds, longer period of complaints, and a mass. However, it has been shown that this difference is not due to a difference in process of AA in older patients but rather to the late presentation to the ED [16].
Although anorexia, nausea, vomiting, and pain in the right lower quadrant are the main symptoms of patients with AA, clinical findings depend on the stage of the disease at the admission. Findings may not be specific at the beginning, whereas other signs include tachycardia, tachypnea, hypotension, oliguria, and acute abdomen such as rigidity and rebound tenderness (localized or generalized). Fever may not be present.
Ultrasonography is the first choice for the diagnosis of AA. However, since it is operative dependent, it may not always be accurate nor decrease rate of negative appendectomies. In patients with suspected diagnosis or in conditions needed to eliminate other causes of abdominal pain, CT scan with contrast enhanced may be the choice (sensitivity 91–98.5% and specificity 90–98%) [16, 38, 39]. A recent meta-analysis demonstrated that magnetic resonance imaging (MRI) for the diagnosis of AA has a sensitivity and specificity of 96% and 96%, respectively, for the general population and 94 and 97% for pregnant patients [39]. The use of MRI for diagnosis of AA can be used in older patients, but the availability 24 h a day could be a major limitation. Further investigations are needed in older patients.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





