CHAPTER 6
Corticosteroids: Indications, Pharmacology, and Risks in Interventional Pain Management
Corticosteroids have been a mainstay in the area of interventional pain management because of their clinical effectiveness in treating numerous inflammatory subacute and chronic pain conditions. These conditions often include significant inflammatory components as seen in certain neuraxial, intra-articular and extra-articular disorders.
INDICATIONS FOR CORTICOSTEROIDS IN INTERVENTIONAL PAIN MANAGEMENT
Hollander introduced the concept that injected corticosteroids can have a positive effect on inflammatory pain conditions in 1950. Since Hollander’s study, the role of corticosteroids has expanded into treating many other types of acute and chronic pain conditions such as degenerative and inflammatory conditions of the peripheral joints and the spine.6
The common indications for corticosteroids in interventional pain medicine can be broadly divided into the following categories:
• Peripheral joints
• Extra-articular tissue (eg, bursa)
• Neuraxial structures and spaces (eg, facet joints, epidural space)
Epidural steroids were introduced into the United States in the 1960s and are the most commonly performed spinal interventions in the United States with an annual estimated Medicare spending of 175 million dollars in 2001. Studies have shown that epidural steroid can be effective in the treatment of painful radiculopathy from:
• Disc herniations
• Spondylolysis
• Spinal stenosis
• Annular tears
• Degenerative conditions of cervical and lumbar spine
Other diseases that may benefit from corticosteroid injections include:
• Osteoarthritis of the spine and peripheral joints
• Rheumatoid arthritis and extra-articular disorders
• Tendinitis
• Bursitis
• Ligament sprain
• Tenosynovitis
• Other overuse syndromes
CONTRAINDICATIONS FOR CORTICOSTEROIDS IN INTERVENTIONAL PAIN MANAGEMENT
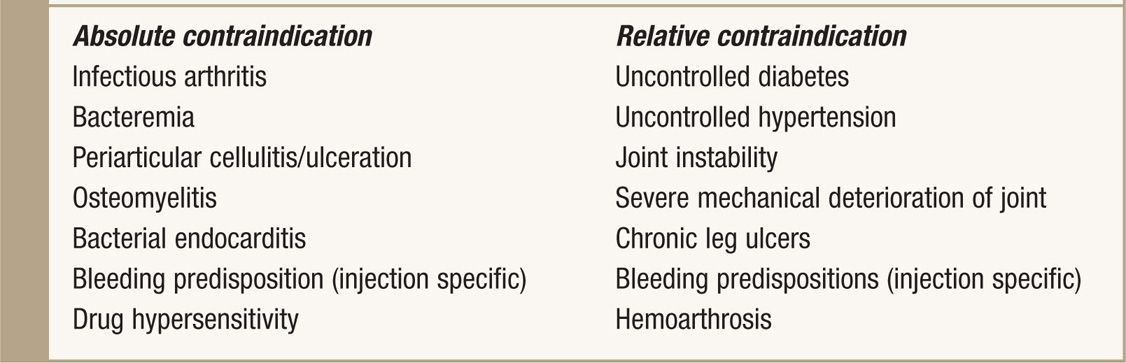
The contraindications are more a function of the specific injection being performed rather than whether if cortisone should be included in the injectate. Active infection at the needle insertion site must be considered before all injections although this at times may prove difficult to differentiate from noninfectious arthropathies. Bleeding disorders are especially important in neuraxial procedures as they can lead to paralysis if the physician is not appropriately vigilant. The absolute and relative contraindications for corticosteroid injections are listed in Table 6-1.
TABLE 6-1. Absolute and Relative Contraindications to Corticosteroid Injection
PHYSIOLOGY
• Cortisol is an endogenous glucocorticoid required for normal cellular function synthesized in the zona fasciculata of the adrenal cortex.
• It is essential for normal metabolism, wound healing, gluconeogenesis, lipolysis, and immunologic activity.
• It has significant anti-inflammatory actions that are beneficial in the treatment of chronic pain conditions.
• Cortisol is under the control of the hypothalamic-pituitary-adrenal axis (HPA).
• In the presence of a stressor, the hypothalamus releases corticotropin releasing hormone (CRH).
• CRH induces the anterior pituitary gland to release adrenal corticotropic hormone (ACTH), which in turn stimulates the adrenal cortex to release cortisol, androgen, and aldosterone.
• Normally, the human body produces 10-12 mg of cortisol per m2 of body surface area daily.
• This amount equates to a glucocorticoid secretion of 20-30 mg/day of oral hydrocortisone or 5-7 mg/day of oral prednisone.
• With stressful conditions, cortisol synthesis can increase up to 5-10 fold to 100 mg/m2/day.1
• The highest cortisol concentration is early in the morning and the lowest cortisol concentration is at evening hours.
PHARMACOLOGY
• Cortisol is a derivative of cholesterol that exists in both bound and unbound (active) forms with 90% being carried on cortisol-binding globulin.
• Its basic chemical structure is a 17-carbon skeleton with 6-carbon hexane rings and one 5-carbon pentane ring.
• The different analogues that are produced and utilized in practice are created by altering the arrangement of the steroid molecule to enhance certain anti-inflammatory properties as well as to decrease the mineralocorticoid activity.
Cortisol has a half-life of about 90 minutes and is metabolized in the liver by its conversion to a water-soluble compound that can be excreted by the kidney. Any change in the physical structure of the compound, such as with the commonly used particulate preparations of steroids, will result in slower metabolism of the product and thus a longer half-life and duration of action.
• Corticosteroids reduce inflammation by decreasing the permeability of capillaries at the affected site.
• They reduce the amount of protein and fluids that are extravasated from the vessel, thereby reducing edema.
• Corticosteroids decrease the inflammatory component of pain by interfering with the synthesis and release of proinflammatory substances by its inhibition of phospholipase-2 activity.
The role of corticosteroids in interrupting the inflammatory pathway is shown in Figure 6-1.
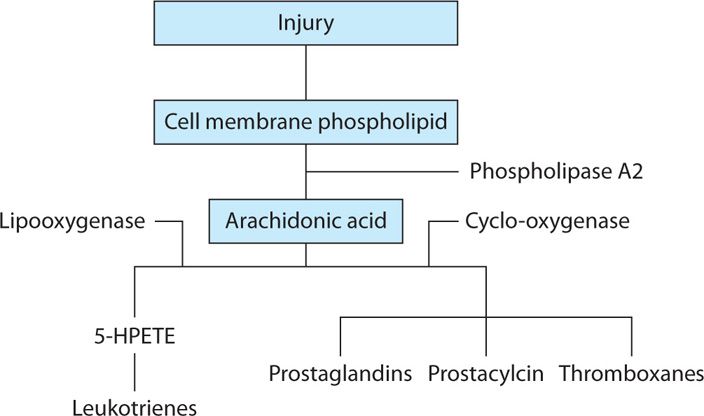
Figure 6-1. Inflammatory pathway.
Pain relief after injection of a neuroma with corticosteroids can be due to:
• Softening of the scar that enclosed the nerve fibrils
• Reducing the mechanical transmission that would produce pain
Corticosteroids act at the nerve fibers and affect the transmission of pain. A study in which the sciatic nerves of rats were ligated and then treated with either triamcinolone hexacetonide, triamcinolone diacetate, or dexamethasone demonstrated a statistically significant reduction in the incidence of spontaneous discharge of the A beta fibers in a neuroma compared to the saline control. The application of corticosteroids to the cut nerves prevented ectopic neuroma hyperexcitability and decreased the discharge in active neuromas that had been present for 3 to 10 days postoperatively. A study by Johnasson of corticosteroids on exposed rat nerves interestingly suggested that its application reduced the transmission of normal unmyelinated C-fibers that carry nociceptive impulses.33
With inflammatory, neuralgic, and paresthetic pain, the initial insult results in:
• Lowered activation threshold
• Increased sensitivity to various stimuli
• Prolonged impulse discharge
• Corticosteroids can normalize the abnormally lowered activation threshold and reduce impulse amplitude and propagation
COMPLICATIONS OF CHRONIC CORTICOSTEROID THERAPY
• Weight gain
• Hyperglycemia
• Congestive heart failure
• Adrenal insufficiency
• Cushing syndrome
Adrenal Insufficiency
Adrenal Insufficiency is a significant concern with frequent exogenous corticosteroid administration because of the interaction with the HPA axis which functions on a feedback mechanism. Symptoms include:
• Hypotension
• Orthostatic Hypotension
• Generalized weakness
• Weight loss
• Anorexia
• Lethargy
• Depression
• Abdominal symptoms
• Amenorrhea
The degree of adrenal suppression is affected by the dose, frequency, chronicity and intervals between glucocorticoid administration. However, an acute suppression of ACTH after a single dose of epidural trimacinolone and suppression of cortisol levels following an intra-articular and intramuscular injection have also been shown. Prompt recognition and treatment are essential. The duration of adrenal suppression with the commonly used depot corticosteroids is shown in Table 6-2. Adrenal suppression has been reported to take up to 9 to 12 months to recover after chronic use of supraphysiologic doses of glucocorticoids.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






