Key Concepts
 The primary clinical use of cholinesterase inhibitors, also called anticholinesterases, is to reverse nondepolarizing muscle blockade.
The primary clinical use of cholinesterase inhibitors, also called anticholinesterases, is to reverse nondepolarizing muscle blockade.
 Acetylcholine is the neurotransmitter for the entire parasympathetic nervous system (parasympathetic ganglions and effector cells), parts of the sympathetic nervous system (sympathetic ganglions, adrenal medulla, and sweat glands), some neurons in the central nervous system, and somatic nerves innervating skeletal muscle.
Acetylcholine is the neurotransmitter for the entire parasympathetic nervous system (parasympathetic ganglions and effector cells), parts of the sympathetic nervous system (sympathetic ganglions, adrenal medulla, and sweat glands), some neurons in the central nervous system, and somatic nerves innervating skeletal muscle.
 Neuromuscular transmission is blocked when nondepolarizing muscle relaxants compete with acetylcholine to bind to nicotinic cholinergic receptors. The cholinesterase inhibitors indirectly increase the amount of acetylcholine available to compete with the nondepolarizing agent, thereby reestablishing neuromuscular transmission.
Neuromuscular transmission is blocked when nondepolarizing muscle relaxants compete with acetylcholine to bind to nicotinic cholinergic receptors. The cholinesterase inhibitors indirectly increase the amount of acetylcholine available to compete with the nondepolarizing agent, thereby reestablishing neuromuscular transmission.
 In excessive doses, acetylcholinesterase inhibitors can paradoxically potentiate a nondepolarizing neuromuscular blockade. In addition, these drugs prolong the depolarization blockade of succinylcholine.
In excessive doses, acetylcholinesterase inhibitors can paradoxically potentiate a nondepolarizing neuromuscular blockade. In addition, these drugs prolong the depolarization blockade of succinylcholine.
 Any prolongation of action of a nondepolarizing muscle relaxant from renal or hepatic insufficiency will probably be accompanied by a corresponding increase in the duration of action of a cholinesterase inhibitor.
Any prolongation of action of a nondepolarizing muscle relaxant from renal or hepatic insufficiency will probably be accompanied by a corresponding increase in the duration of action of a cholinesterase inhibitor.
 The time required to fully reverse a nondepolarizing block depends on several factors, including the choice and dose of cholinesterase inhibitor administered, the muscle relaxant being antagonized, and the extent of the blockade before reversal.
The time required to fully reverse a nondepolarizing block depends on several factors, including the choice and dose of cholinesterase inhibitor administered, the muscle relaxant being antagonized, and the extent of the blockade before reversal.
 A reversal agent should be routinely given to patients who have received nondepolarizing muscle relaxants unless full reversal can be demonstrated or the postoperative plan includes continued intubation and ventilation.
A reversal agent should be routinely given to patients who have received nondepolarizing muscle relaxants unless full reversal can be demonstrated or the postoperative plan includes continued intubation and ventilation.
 In monitoring a patient’s recovery from neuromuscular blockade, the suggested end points are sustained tetanus for 5 sec in response to a 100-Hz stimulus in anesthetized patients or sustained head lift in awake patients. If neither of these end points is achieved, the patient should remain intubated and ventilation should be continued.
In monitoring a patient’s recovery from neuromuscular blockade, the suggested end points are sustained tetanus for 5 sec in response to a 100-Hz stimulus in anesthetized patients or sustained head lift in awake patients. If neither of these end points is achieved, the patient should remain intubated and ventilation should be continued.
 Sugammadex exerts its effects by forming tight complexes in a 1:1 ratio with steroidal neuromuscular blocking agents.
Sugammadex exerts its effects by forming tight complexes in a 1:1 ratio with steroidal neuromuscular blocking agents.
 Cysteine causes inactivation of gantacurium via metabolic degradation and adduct formation.
Cysteine causes inactivation of gantacurium via metabolic degradation and adduct formation.
Cholinesterase Inhibitors & Other Pharmacologic Antagonists to Neuromuscular Blocking Agents: Introduction
Incomplete reversal of neuromuscular blocking agents and residual post-procedure paralysis are associated with morbidity; therefore, careful evaluation of neuromuscular blockade and appropriate pharmacologic antagonism are strongly recommended whenever muscle relaxants are administered.  The primary clinical use of cholinesterase inhibitors, also called anticholinesterases, is to reverse nondepolarizing muscle blockade. Some of these agents are also used to diagnose and treat myasthenia gravis. More recently, newer agents, such as cyclodextrins and cysteine, with superior ability to reverse neuromuscular blockade from specific agents, are being investigated with promising results. This chapter reviews cholinergic pharmacology and mechanisms of acetylcholinesterase inhibition and presents the clinical pharmacology of commonly used cholinesterase inhibitors (neostigmine, edrophonium, pyridostigmine, and physostigmine). It concludes with a brief description and mechanisms of action of some unique reversal agents.
The primary clinical use of cholinesterase inhibitors, also called anticholinesterases, is to reverse nondepolarizing muscle blockade. Some of these agents are also used to diagnose and treat myasthenia gravis. More recently, newer agents, such as cyclodextrins and cysteine, with superior ability to reverse neuromuscular blockade from specific agents, are being investigated with promising results. This chapter reviews cholinergic pharmacology and mechanisms of acetylcholinesterase inhibition and presents the clinical pharmacology of commonly used cholinesterase inhibitors (neostigmine, edrophonium, pyridostigmine, and physostigmine). It concludes with a brief description and mechanisms of action of some unique reversal agents.
Cholinergic Pharmacology
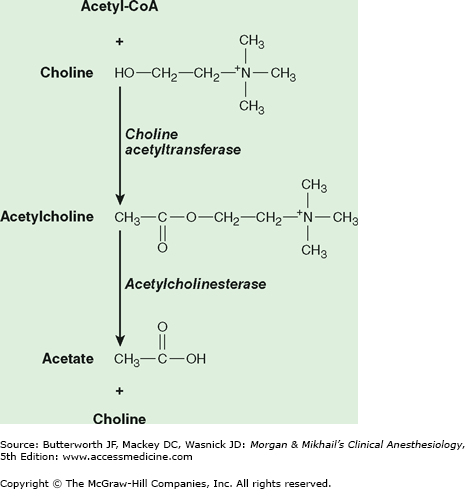
The term cholinergic refers to the effects of the neurotransmitter acetylcholine, as opposed to the adrenergic effects of noradrenaline (norepinephrine). Acetylcholine is synthesized in the nerve terminal by the enzyme cholineacetyltransferase, which catalyzes the reaction between acetylcoenzyme A and choline (Figure 12-1). After its release, acetylcholine is rapidly hydrolyzed by acetylcholinesterase (true cholinesterase) into acetate and choline.
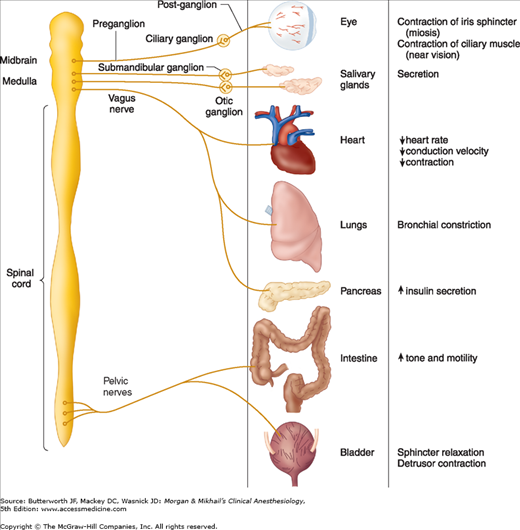
 Acetylcholine is the neurotransmitter for the entire parasympathetic nervous system (parasympathetic ganglions and effector cells), parts of the sympathetic nervous system (sympathetic ganglions, adrenal medulla, and sweat glands), some neurons in the central nervous system, and somatic nerves innervating skeletal muscle (Figure 12-2).
Acetylcholine is the neurotransmitter for the entire parasympathetic nervous system (parasympathetic ganglions and effector cells), parts of the sympathetic nervous system (sympathetic ganglions, adrenal medulla, and sweat glands), some neurons in the central nervous system, and somatic nerves innervating skeletal muscle (Figure 12-2).
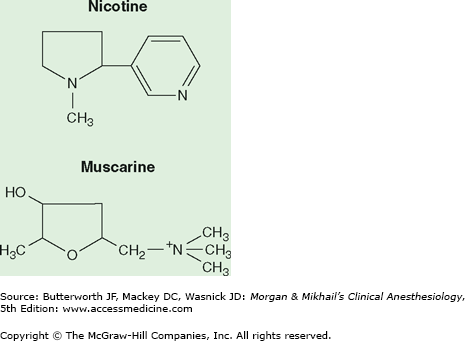
Cholinergic receptors have been subdivided into two major groups based on their reaction to the alkaloids muscarine and nicotine (Figure 12-3). Nicotine stimulates the autonomic ganglia and skeletal muscle receptors (nicotinic receptors), whereas muscarine activates end-organ effector cells in bronchial smooth muscle, salivary glands, and the sinoatrial node (muscarinic receptors). The central nervous system has both nicotinic and muscarinic receptors. Nicotinic receptors are blocked by muscle relaxants (also called neuromuscular blockers), and muscarinic receptors are blocked by anticholinergic drugs, such as atropine. Although nicotinic and muscarinic receptors differ in their response to some agonists (eg, nicotine, muscarine) and some antagonists (eg, vecuronium vs atropine), they both respond to acetylcholine (Table 12-1). Clinically available cholinergic agonists resist hydrolysis by cholinesterase. Methacholine and bethanechol are primarily muscarinic agonists, whereas carbachol has both muscarinic and nicotinic agonist activities. Methacholine by inhalation has been used as a provocative test in asthma, bethanechol is used for bladder atony, and carbachol may be used topically for wide-angle glaucoma.
| Nicotinic | Muscarinic | |
|---|---|---|
| Location |
|
|
| Agonists |
|
|
| Antagonists | Nondepolarizing relaxants |
|
When reversing neuromuscular blockade, the primary goal is to maximize nicotinic transmission with a minimum of muscarinic side effects.
 Normal neuromuscular transmission critically depends on acetylcholine binding to nicotinic cholinergic receptors on the motor end-plate. Nondepolarizing muscle relaxants act by competing with acetylcholine for these binding sites, thereby blocking neuromuscular transmission. Reversal of blockade depends on gradual diffusion, redistribution, metabolism, and excretion from the body of the nondepolarizing relaxant (spontaneous reversal), often assisted by the administration of specific reversal agents (pharmacological reversal). Cholinesterase inhibitors indirectly increase the amount of acetylcholine available to compete with the nondepolarizing agent, thereby reestablishing normal neuromuscular transmission.
Normal neuromuscular transmission critically depends on acetylcholine binding to nicotinic cholinergic receptors on the motor end-plate. Nondepolarizing muscle relaxants act by competing with acetylcholine for these binding sites, thereby blocking neuromuscular transmission. Reversal of blockade depends on gradual diffusion, redistribution, metabolism, and excretion from the body of the nondepolarizing relaxant (spontaneous reversal), often assisted by the administration of specific reversal agents (pharmacological reversal). Cholinesterase inhibitors indirectly increase the amount of acetylcholine available to compete with the nondepolarizing agent, thereby reestablishing normal neuromuscular transmission.
Cholinesterase inhibitors inactivate acetylcholinesterase by reversibly binding to the enzyme. The stability of the bond influences the duration of action. The electrostatic attraction and hydrogen bonding of edrophonium are short-lived; the covalent bonds of neostigmine and pyridostigmine are longer lasting.
Organophosphates, a special class of cholinesterase inhibitors, form very stable, irreversible bonds to the enzyme. They are used in ophthalmology and more commonly as pesticides. The clinical duration of the cholinesterase inhibitors used in anesthesia, however, is probably most influenced by the rate of drug disappearance from the plasma. Differences in duration of action can be overcome by dosage adjustments. Thus, the normally short duration of action of edrophonium can be partially overcome by increasing the dosage. Cholinesterase inhibitors are also used in the diagnosis and treatment of myasthenia gravis.
Mechanisms of action other than acetylcholinesterase inactivation may contribute to the restoration of neuromuscular function. Edrophonium seems to have prejunctional effects that enhance the release of acetylcholine. Neostigmine has a direct (but weak) agonist effect on nicotinic receptors. Acetylcholine mobilization and release by the nerve may also be enhanced (a presynaptic mechanism).
 In excessive doses, acetylcholinesterase inhibitors paradoxically potentiate a nondepolarizing neuromuscular blockade. Standard dogma states that neostigmine in high doses may cause receptor channel blockade; however, clinical evidence of this is lacking. In addition, these drugs prolong the depolarization blockade of succinylcholine. Two mechanisms may explain this latter effect: an increase in acetylcholine (which increases motor end-plate depolarization) and inhibition of pseudocholinesterase activity. Neostigmine and to some extent pyridostigmine display some limited pseudocholinesterase-inhibiting activity, but their effect on acetylcholinesterase is much greater. Edrophonium has little or no effect on pseudocholinesterase. In large doses, neostigmine can cause a weak depolarizing neuromuscular blockade.
In excessive doses, acetylcholinesterase inhibitors paradoxically potentiate a nondepolarizing neuromuscular blockade. Standard dogma states that neostigmine in high doses may cause receptor channel blockade; however, clinical evidence of this is lacking. In addition, these drugs prolong the depolarization blockade of succinylcholine. Two mechanisms may explain this latter effect: an increase in acetylcholine (which increases motor end-plate depolarization) and inhibition of pseudocholinesterase activity. Neostigmine and to some extent pyridostigmine display some limited pseudocholinesterase-inhibiting activity, but their effect on acetylcholinesterase is much greater. Edrophonium has little or no effect on pseudocholinesterase. In large doses, neostigmine can cause a weak depolarizing neuromuscular blockade.