Low doses of IV anesthetics produce sedation, and high doses produce hypnosis (or unconsciousness)
 All IV anesthetics are sedative–hypnotics and produce dose-dependent central nervous system (CNS) depression.
All IV anesthetics are sedative–hypnotics and produce dose-dependent central nervous system (CNS) depression.
 Dexmedetomidine is an α2-agonist with sedative and opioid-sparing effects that is used as an anesthetic adjuvant in the operating room and intensive care unit.
Dexmedetomidine is an α2-agonist with sedative and opioid-sparing effects that is used as an anesthetic adjuvant in the operating room and intensive care unit.
 With the exception of ketamine and dexmedetomidine, intravenous (IV) anesthetics lack intrinsic analgesic properties.
With the exception of ketamine and dexmedetomidine, intravenous (IV) anesthetics lack intrinsic analgesic properties.
 Compared to thiopental and propofol, methohexital produces less depression of electroencephalogram (EEG) activity.
Compared to thiopental and propofol, methohexital produces less depression of electroencephalogram (EEG) activity.
 IV anesthetics in combination with potent opioid analgesics and/or local anesthetics can be used to produce total intravenous anesthesia (TIVA).
IV anesthetics in combination with potent opioid analgesics and/or local anesthetics can be used to produce total intravenous anesthesia (TIVA).
 Propofol possesses unique antiemetic and appetite-stimulating properties.
Propofol possesses unique antiemetic and appetite-stimulating properties.
 Midazolam possesses prominent amnestic and anxiolytic properties.
Midazolam possesses prominent amnestic and anxiolytic properties.
 Etomidate produces less cardiovascular depression than the barbiturates and propofol.
Etomidate produces less cardiovascular depression than the barbiturates and propofol.
 Ketamine possesses both analgesic and psychomimetic properties.
Ketamine possesses both analgesic and psychomimetic properties.
Multimedia
 Drug Concentration and Effect
Drug Concentration and Effect
INTRODUCTION
In general, low doses of  IV anesthetics produce sedation, and high doses produce hypnosis (or unconsciousness). The concept of intravenous (IV) anesthesia has evolved from primarily induction of general anesthesia to total IV anesthesia (TIVA).1 TIVA has assumed increasing importance for therapeutic, as well as diagnostic, procedures in both adults and children.2 IV anesthetic techniques are used for procedures in the operating room (OR) and in sites remote from the OR. In many centers in Europe and South America, TIVA has become more popular for general anesthesia than classic “balanced anesthesia” or volatile anesthetic-based techniques. For superficial surgical procedures, lower dosages of IV anesthetics are increasingly being used for sedation during local and regional anesthesia, so-called monitored anesthesia care (MAC) (see Chapter 29 Monitored Anesthesia Care).2 This change has been a result of: (1) the development of rapid, short-acting IV hypnotic, analgesic, and muscle relaxant drugs; (2) the availability of pharmacokinetic and dynamic-based IV delivery systems; and (3) the development of the electroencephalogram (EEG)-based cerebral monitoring devices, which measure the hypnotic component of the anesthetic state.
IV anesthetics produce sedation, and high doses produce hypnosis (or unconsciousness). The concept of intravenous (IV) anesthesia has evolved from primarily induction of general anesthesia to total IV anesthesia (TIVA).1 TIVA has assumed increasing importance for therapeutic, as well as diagnostic, procedures in both adults and children.2 IV anesthetic techniques are used for procedures in the operating room (OR) and in sites remote from the OR. In many centers in Europe and South America, TIVA has become more popular for general anesthesia than classic “balanced anesthesia” or volatile anesthetic-based techniques. For superficial surgical procedures, lower dosages of IV anesthetics are increasingly being used for sedation during local and regional anesthesia, so-called monitored anesthesia care (MAC) (see Chapter 29 Monitored Anesthesia Care).2 This change has been a result of: (1) the development of rapid, short-acting IV hypnotic, analgesic, and muscle relaxant drugs; (2) the availability of pharmacokinetic and dynamic-based IV delivery systems; and (3) the development of the electroencephalogram (EEG)-based cerebral monitoring devices, which measure the hypnotic component of the anesthetic state.  All IV anesthetics are sedative–hypnotics and produce dose-dependent central nervous system (CNS) depression. This chapter focuses on the pharmacologic properties and clinical uses of the currently available IV anesthetics.
All IV anesthetics are sedative–hypnotics and produce dose-dependent central nervous system (CNS) depression. This chapter focuses on the pharmacologic properties and clinical uses of the currently available IV anesthetics.
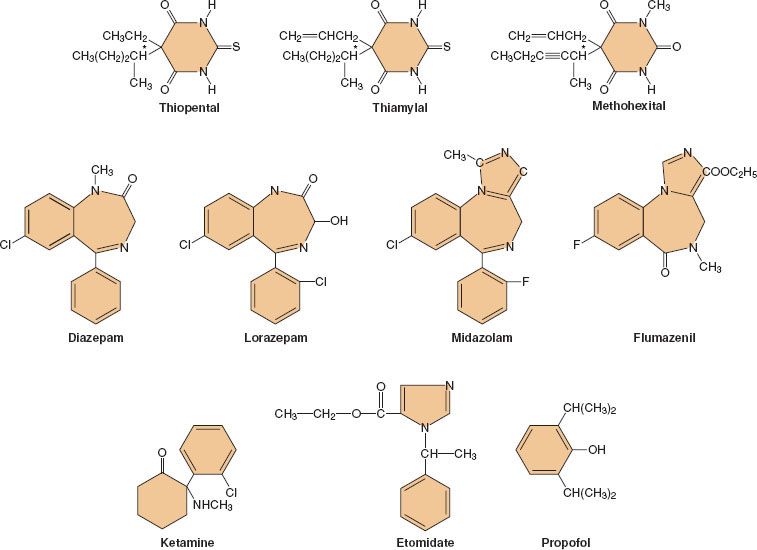
Following its introduction into clinical practice, thiopental quickly became the gold standard of IV anesthetics against which all the newer IV drugs were compared. Many different hypnotic drugs are currently available for use during IV anesthesia (Fig. 18-1). Although propofol (currently the most widely used IV anesthetic) is both safe and effective, the “ideal” IV anesthetic is yet to be developed. The physical and pharmacologic properties that an ideal IV anesthetic would possess include the following:
FIGURE 18-1. Chemical structures of currently available nonopioid intravenous anesthetics.
1. Drug compatibility (water-solubility) and stability in solution.
2. Lack of pain on injection, veno-irritation, and local tissue damage following extravasation.
3. Low potential to release histamine or precipitate hypersensitivity reactions.
4. Rapid and smooth onset of hypnotic action without excitatory activity.
5. Rapid metabolism to pharmacologically inactive metabolites.
6. A steep dose-response relationship to enhance titratability and minimize tissue accumulation.
7. Lack of acute cardiovascular and respiratory depression.
8. Decreases in cerebral metabolism and intracranial pressure.
9. Rapid and smooth return of consciousness and cognitive skills with residual analgesia.
10. Absence of postoperative nausea and vomiting, amnesia, psychomimetic reactions, dizziness, headache, or prolonged sedation (“hangover” effects).
HISTORICAL PERSPECTIVE
Despite thiopental’s proven clinical usefulness, safety, and widespread use over many decades of use, it has been supplanted by a variety of agents from different pharmacologic drug groups. The sedative–hypnotic drugs that have been more recently introduced into clinical practice (e.g., midazolam, ketamine, etomidate, propofol) have proven to be extremely valuable in specific clinical situations. These newer compounds combine many of the characteristics of the ideal IV anesthetic, but fail in aspects where the other drugs succeed. For some of these IV sedative–hypnotics, disadvantages have led to “restricted” indications (e.g., ketamine, etomidate). Although propofol is clearly the most popular of the sedative–hypnotic drug class, the optimal pharmacologic properties are not equally important in every clinical situation. Therefore, the anesthesiologist must make the choice of the IV anesthetic drug that best fits the needs of the individual patient and the operative (or diagnostic) procedure.
GENERAL PHARMACOLOGY OF INTRAVENOUS ANESTHETICS
Mechanism of Action
A widely accepted theory of anesthetic action is that both IV and inhalational anesthetics exert their primary sedative and hypnotic effects through an interaction with the inhibitory γ-aminobutyric acid (GABA) neurotransmitter system.3 GABA is the principal inhibitory neurotransmitter within the CNS. The GABA and adrenergic neurotransmitter systems counterbalance the action of excitatory neurotransmitters. The GABA type A (GABAA) receptor is a receptor complex consisting of up to five glycoprotein subunits. When the GABAA receptor is activated, transmembrane chloride conductance increases, resulting in hyperpolarization of the postsynaptic cell membrane and functional inhibition of the postsynaptic neuron. Sedative–hypnotic drugs interact with different components of the GABA-receptor complex (Fig. 18-2). However, the allosteric (structural) requirements for activation of the receptor are different for IV and volatile anesthetics.
FIGURE 18-2. A: This model depicts the postsynaptic site of γ-aminobutyric acid (GABA) and glutamate within the central nervous system (CNS). GABA decreases the excitability of neurons by its action at the GABAA-receptor complex. When GABA occupies the binding site of this complex, it allows inward flux of chloride ion, resulting in hyperpolarizing of the cell and subsequent resistance of the neuron to stimulation by excitatory transmitters. Barbiturates, benzodiazepines, propofol, and etomidate decrease neuronal excitability by enhancing the effect of GABA at this complex, facilitating this inhibitory effect on the postsynaptic cell. Glutamate and its analog N-methyl-D-aspartate (NMDA) are excitatory amino acids. When glutamate occupies the binding site on the NMDA subtype of the glutamate receptor, the channel opens and allows Na+, K+, and Ca2+ to either enter or leave the cell. Flux of these ions leads to depolarization of the postsynaptic neuron and initiation of an action potential and activation of other pathways. Ketamine blocks this open channel and prevents further ion flux, thus inhibiting the excitatory response to glutamate. (Reprinted with permission from: Van Hemelrijck J, Gonzales JM, White PF. Use of intravenous sedative agents. In: Rogers MC, Tinker JH, Covino BG, Longnecker DE, eds. Principles and Practice of Anesthesiology. Mosby: St. Louis; 1992:1131.) B: Schematic model of the GABAA-receptor complex illustrating recognition sites for many of the substances that bind to the receptor. C: Model of the NMDA receptor showing sites for antagonist action. Ketamine binds to the site labeled PCP (phencyclidine). The pentameric structure of the receptor, composed of a combination of the subunits NR 1 and NR 2, is illustrated. (Altered with permission from: Leeson PD, Iversen LL. The glycine site on the NMDA receptor: Structure-activity relationships and therapeutic potential. J Med Chem. 1994;37:4053.)
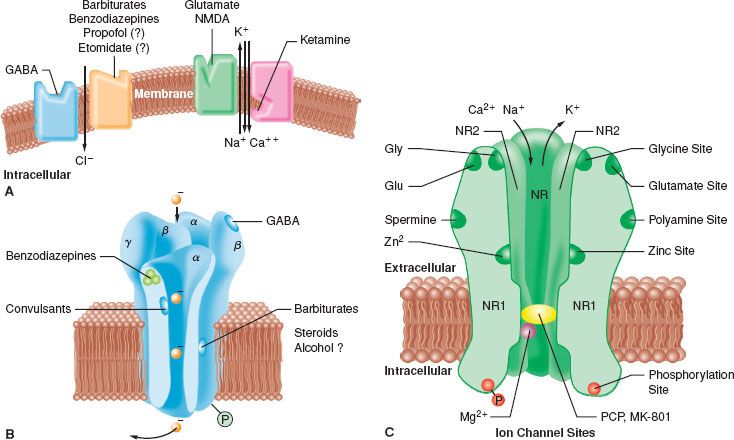
Benzodiazepines bind to specific receptor sites that are part of the GABAA-receptor complex. The binding of benzodiazepines to their receptor site increases the efficiency of the coupling between the GABA receptor and the chloride ion channel. The degree of modulation of the GABA-receptor function is limited, which explains the maximal “ceiling effect” produced by benzodiazepines with respect to CNS depression. The dose-dependent CNS depressant effect of benzodiazepines produce hypnosis, sedation, anxiolysis, amnesia, and anticonvulsant effects.1 These CNS effects are presumed to be associated with stimulation of different receptor subtypes and/or concentration-dependent receptor occupancy.3 For example, it has been suggested that benzodiazepine receptor occupancy of 20% provides anxiolysis, while 30% to 50% receptor occupancy is associated with amnesia to sedation, and 60% receptor occupancy is required for hypnosis (or unconsciousness).1
The interaction of barbiturates and propofol with specific membrane structures appears to decrease the rate of dissociation of GABA from its receptor, thereby increasing the duration of the GABA-activated opening of the chloride ion channel (Fig. 18-2). Barbiturates can also mimic the action of GABA by directly activating the chloride channels. The proposed mechanism of action of thiopental relates to its ability to function as a competitive inhibitor at the nicotinic acetylcholine receptors in the CNS.4 Etomidate augments GABA-gated chloride currents (i.e., indirect modulation) and at higher concentrations evokes chloride currents in the absence of GABA (i.e., direct activation). Although the mechanism of action of propofol is similar to that of the barbiturates (i.e., enhancing the activity of the GABA-activated chloride channel), it also possesses ion channel-blocking effects in cerebral cortex tissue and nicotinic acetylcholine receptors, as well as an inhibitory effect on lysophosphatidate signaling in lipid mediator receptors.5
Ketamine produces a functional dissociation between the thalamocortical and limbic systems, a state that has been termed dissociative anesthesia. Ketamine depresses neuronal function in the cerebral cortex and thalamus, while simultaneously activating the limbic system. The effect of ketamine on the medial medullary reticular formation may be involved in the affective component of its nociceptive activity. The CNS effects of ketamine appear to be primarily related to its antagonistic activity at the N-methyl-D-aspartate (NMDA) receptor (Fig. 18-2). Unlike the other IV anesthetics, ketamine does not interact with GABA receptors; however, it binds to non-NMDA glutamate receptors and nicotinic, muscarinic, monoaminergic, and even kappa-opioid receptors. In addition, it also inhibits neuronal sodium channels (producing a modest local anesthetic action) and calcium channels (causing cerebral vasodilatation).
The centrally  active α2-adrenergic receptor agonists, clonidine and dexmedetomidine, have potent sedative and opioid analgesic-sparing properties. These drugs also have significant effects on the peripheral α2 receptors involved in regulating the cardiovascular system by inhibiting norepinephrine release. This class of anesthetic adjuvants can also reduce blood pressure and heart rate by decreasing the tonic levels of sympathetic outflow from the CNS and augmenting cardiac vagal activity, respectively.6,7 However, dexmedetomidine failed to block the acute hyperdynamic response to electroconvulsive therapy (ECT) when administered as an adjuvant to methohexital anesthesia.8 Earlier studies with clonidine demonstrated that this α2 agonist–antagonist could also reduce the IV9 and volatile10 anesthetic requirements, as well as the postoperative opioid analgesic requirement.
active α2-adrenergic receptor agonists, clonidine and dexmedetomidine, have potent sedative and opioid analgesic-sparing properties. These drugs also have significant effects on the peripheral α2 receptors involved in regulating the cardiovascular system by inhibiting norepinephrine release. This class of anesthetic adjuvants can also reduce blood pressure and heart rate by decreasing the tonic levels of sympathetic outflow from the CNS and augmenting cardiac vagal activity, respectively.6,7 However, dexmedetomidine failed to block the acute hyperdynamic response to electroconvulsive therapy (ECT) when administered as an adjuvant to methohexital anesthesia.8 Earlier studies with clonidine demonstrated that this α2 agonist–antagonist could also reduce the IV9 and volatile10 anesthetic requirements, as well as the postoperative opioid analgesic requirement.
Pharmacokinetics and Metabolism
An understanding of basic pharmacokinetic principles is integral to the understanding of the pharmacologic actions and interactions of IV anesthetic and adjunctive drugs, and will allow the anesthesiologist to develop more optimal dosing strategies when using IV techniques (see Chapter 7 Basic Principles of Clinical Pharmacology). Although lipid solubility facilitates diffusion of IV anesthetics across cellular membranes, including the blood–brain barrier, only the nonionized form of the compound is able to readily cross neuronal membranes. The ratio of the unionized-to-ionized fraction depends on the pKa of the drug and the pH of the body fluids.
The rapid onset of the CNS effect of most IV anesthetics can be explained by their high lipid solubility and the relatively high proportion of the cardiac output (20%) perfusing the brain. However, a variable degree of hysteresis exists between the blood concentration of the hypnotic drug and its onset of action on the CNS. The hysteresis is related in part to diffusion of these drugs into brain tissue and nonspecific CNS receptor binding. However, the number of CNS binding sites is usually saturable and only a small fraction of the available binding sites needs to be occupied to produce clinical effects. Although the total amount of drug in the blood is available for diffusion, the diffusion rate will be more limited for IV anesthetics with a high degree of plasma protein binding (90%) because only the “free” unbound drug can diffuse across membranes and exert central effects. When several drugs compete for the same binding sites, or when the protein concentration in the blood is decreased by preexisting disease (e.g., hepatic failure, malnutrition), a higher fraction of the unbound drug will be available to exert an effect on the CNS. Since only unbound drug is available for uptake and metabolism in the liver, highly protein-bound drugs may have a lower rate of hepatic metabolism as a result of their decreased hepatic extraction ratio (i.e., the fraction of the hepatic blood flow that is cleared of the drug).
The pharmacokinetics of IV hypnotics are characterized by rapid distribution and subsequent redistribution into several hypothetical compartments (determined by their effect on blood flow to various tissues), followed by elimination (Table 18-1). The initial pharmacologic effects are related to the activity of the drug in the central compartment. The primary mechanism for terminating the central effects of IV anesthetics administered for induction of anesthesia is redistribution from the central highly perfused compartment (e.g., brain) to the larger, but less well perfused “peripheral” compartments (e.g., muscle, fat). Even for drugs with a high hepatic extraction ratio (e.g., propofol), elimination does not always play a major role in terminating the drug’s sedative–hypnotic effects because elimination of the drug can occur only from the central compartment. The rate of elimination from the central compartment, the amount of drug present in the peripheral compartments, and the rate of redistribution from the peripheral compartments “back” into the central compartment determine the time necessary to eliminate the drug from the body and can directly influence intermediate and late recovery times.
TABLE 18-1. PHARMACOKINETIC VALUES FOR THE CURRENTLY AVAILABLE INTRAVENOUS SEDATIVE–HYPNOTIC DRUGS
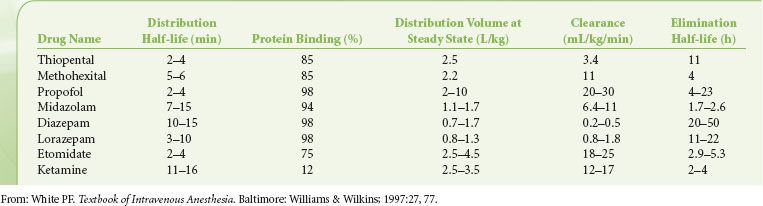
Most IV anesthetic agents are eliminated via hepatic metabolism followed by renal excretion of more water-soluble metabolites. Some metabolites have pharmacologic activity and can produce prolonged drug effects (e.g., oxazepam, desmethyldiazepam, norketamine). Moreover, there is considerable interpatient variability in the clearance rates for commonly used IV anesthetic drugs. The elimination clearance is the distribution volume cleared of drug over time and is a measure of the efficacy of the elimination process. The slow elimination of some anesthetics is partly due to their high degree of protein binding that reduces their hepatic extraction ratio. Other drugs may have a high hepatic extraction ratio and elimination clearance despite extensive plasma protein binding (e.g., propofol), indicating that protein binding is not always a rate-limiting factor.
For most drugs, the hepatic enzyme systems are not saturated at clinically relevant drug concentrations, and the rate of drug elimination will decrease as an exponential function of the drug’s plasma concentration (first-order kinetics). However, when high steady state plasma concentrations are achieved with prolonged infusions, hepatic enzyme systems can become saturated and the elimination rate becomes independent of the drug concentration (zero-order  kinetics). The elimination half-life (t1/2β) is the time required for the anesthetic concentration to decrease by 50% during the terminal phase of the plasma decay curve. The t1/2β depends on the volume to be cleared (the distribution volume) and the efficiency of the metabolic clearance system. Because their volumes of distribution are similar, the wide variation in elimination half-life values for the IV anesthetics is a reflection of differences in their clearance values.
kinetics). The elimination half-life (t1/2β) is the time required for the anesthetic concentration to decrease by 50% during the terminal phase of the plasma decay curve. The t1/2β depends on the volume to be cleared (the distribution volume) and the efficiency of the metabolic clearance system. Because their volumes of distribution are similar, the wide variation in elimination half-life values for the IV anesthetics is a reflection of differences in their clearance values.
When a drug infusion is administered without a loading dose, at least three times the t1/2β value may be required to achieve a true “steady state” plasma concentration. The steady state concentration obtained during an anesthetic infusion depends on the rate of drug administration and its clearance rate. When an infusion is discontinued, the rate at which the plasma concentration decreases largely depends on the clearance rate (as reflected by the terminal t1/2β value). For drugs with shorter elimination half-lives, plasma concentration will decrease at a rate that allows for a more rapid recovery (e.g., propofol). Drugs with longer t1/2β values (e.g., thiopental and diazepam) are usually only administered by continuous IV infusion when the medical condition requires long-term treatment (e.g., elevated intracranial pressure [ICP] as a result of brain injury or prolonged sedation in the intensive care unit [ICU] because of respiratory failure).
Careful titration of an anesthetic drug to achieve the desired clinical effect is necessary to avoid drug accumulation and the resultant prolonged CNS effects after the infusion has been discontinued. Although the value of the t1/2β indicates how fast a drug is eliminated from the body, a more useful indicator of the acceptability of a hypnotic infusion for maintenance of anesthesia or sedation is the so-called context-sensitive half-time, a value derived from computer simulations of drug infusions.11 The context-sensitive half-time is defined as the time necessary for the effect-compartment (i.e., effect site) concentration to decrease by 50% in relation to the duration of the infusion. The context-sensitive half-time becomes particularly important in determining recovery after prolonged infusions of sedative–hypnotic drugs. Drugs (e.g., propofol) may have a relatively short context-sensitive half-time despite the fact that a large amount of drug remains present in the “deep” (less well-perfused) tissue compartment. The slow return of the anesthetic from the deep compartment contributes little to the concentration of drug in the central compartment from which it is rapidly cleared. Therefore, the concentration in the central compartment rapidly declines below the hypnotic threshold after discontinuation of the infusion, contributing to short emergence times despite the fact that a substantial quantity of anesthetic drug may remain in the body.
Marked interpatient variability exists in the pharmacokinetics of IV sedative–hypnotic drugs. Factors that can influence anesthetic drug disposition include the degree of protein binding, the efficiency of hepatic and renal elimination processes, physiologic changes with aging, preexisting disease states, the operative site, body temperature, and drug interactions (e.g., coadministration of volatile anesthetics). For example, increased age, lean body (muscle) mass, and total body water decrease result in an increase in the steady state volume of distribution of most IV anesthetics. The increased distribution volume and decreased hepatic clearance leads to a prolongation of their t1/2β values. Moreover, a decrease of the volume of the central compartment may result in higher initial drug concentrations and can at least partially explain the decreased induction requirement in the elderly. Additionally, the slower redistribution from the vessel-rich tissues to intermediate compartments (e.g., muscles) also contributes to the age-related decrease in the induction dose requirements.11 Although prolongation of the elimination half-time in the elderly does not provide an explanation for the decreased induction dose requirement, it is responsible for producing higher steady state plasma concentrations of IV anesthetics at any given infusion rate when administered for maintenance of general anesthesia or sedation, contributing to a slower recovery from the sub-hypnotic (residual) CNS depressant effects.
The hepatic clearance of IV anesthetics with a high (e.g., etomidate, propofol, ketamine) or intermediate (e.g., methohexital, midazolam) extraction ratio largely depends on hepatic blood flow, with most of the drug being removed from the blood as it flows through the liver (so-called perfusion-limited clearance). The elimination rate of drugs with low hepatic extraction ratios (e.g., thiopental, diazepam, lorazepam) depends on the enzymatic activity of the liver and is less dependent of hepatic blood flow (so-called capacity-limited clearance). Hepatic blood flow decreases during upper abdominal and laparoscopic surgery and, as a result, higher blood levels of drugs with perfusion-limited clearance are achieved at any given infusion rate. With aging, a decreased cardiac output and a redistribution of blood flow can partly explain the lower clearance rate for drugs with perfusion-limited clearance. Although concomitant administration of volatile anesthetics (which are known to decrease liver blood flow) has little influence on the elimination of thiopental, they can decrease the clearance of etomidate, ketamine, methohexital, and propofol. Other factors that decrease hepatic blood flow include hypocapnia, congestive heart failure, intravascular volume depletion, acute alcohol intoxication, circulatory collapse, increase intra-abdominal pressure, β-adrenergic blockade, and norepinephrine administration.
Hepatic disease can influence the pharmacokinetics of drugs by: (1) altering the plasma protein content and changing the degree of protein binding, (2) decreasing hepatic blood flow and producing intrahepatic shunting, and (3) depressing the metabolic enzymatic activity of the liver. Therefore, the influence of hepatic disease on pharmacokinetics and dynamics of IV anesthetics is difficult to predict. Renal disease can also alter the concentration of plasma and tissue proteins, as well as the degree of protein binding, thereby producing changes in free drug concentrations. Because IV anesthetic agents are primarily metabolized by the liver, renal insufficiency has little influence on their rate of metabolic inactivation or elimination of the primary compound.
Pharmacodynamic Effects
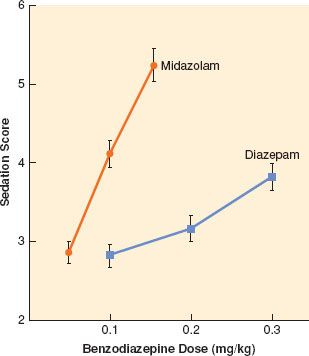
The principal pharmacologic effect of IV anesthetics is to produce progressively increasing sedation and ultimately hypnosis as a result of dose-dependent CNS depression. However, all sedative–hypnotics also directly or indirectly affect other major organ systems. The relationship between the dose of a sedative–hypnotic and its CNS effects can be defined by dose-response curves. Although most IV anesthetics are characterized by steep dose-response curves, the slopes of the curves are not always parallel (Fig. 18-3). However, the characteristics of a dose-response curve can only be interpreted in relation to the specific response for which it was constructed.
FIGURE 18-3. Dose-response relationships for sedation with midazolam (•) and diazepam. The level of sedation (2 = awake and alert to 6 = asleep and unarousable) was assessed 5 minutes after bolus doses of midazolam (0.05, 0.1, or 0.15 mg/kg) or diazepam (0.1, 0.2, or 0.3 mg/kg). Values represent mean values ± SEM. (Reprinted with permission from: White PF, Vasconez LO, Mathes SA, et al. Comparison of midazolam and diazepam for sedation during plastic surgery. J Plast Reconstruct Surg. 1988;81:703.)
When steady state plasma concentrations are achieved, one can assume that the plasma concentration is in quasi-equilibrium with the effect-site concentration. Under these circumstances, it is possible to describe the relationship between drug and effect using a concentration-effect curve (Fig. 18-4). Because of the pharmacodynamic variability that exists among individuals, the plasma drug concentration necessary to obtain a particular effect is often described in terms of an effective concentration range, the so-called therapeutic window. Efficacy of an IV anesthetic relates to the maximum effect that can be achieved with respect to some measure of CNS function. Depending on the drug effect under consideration, the efficacy of sedative–hypnotics may appear to be <100%. Compared to thiopental and propofol, it is extremely difficult to produce an isoelectric [silent] EEG pattern with a benzodiazepine. Potency, on the other hand, relates to the quantity of drug necessary to obtain the maximum CNS effect. The relative potency of sedative–hypnotics also varies depending on the end point chosen. In the presence of an antagonist drug (e.g., flumazenil), the maximal response that can be obtained with a benzodiazepine agonist is further reduced because of competition for the same CNS receptor binding sites.
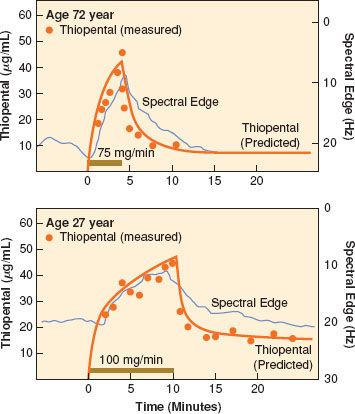
FIGURE 18-4. The concentration of thiopental versus time and spectral edge in an elderly patient (top) and in a younger patient (bottom). Solid horizontal bars represent the length of thiopental infusion. Filled circles represent the measured thiopental concentration (linear scale), and the solid line next to them represents the fitted data from the pharmacokinetic model. The axis for spectral edge has been inverted for visual clarity. (Reprinted with permission from: Homer TD, Stanski DR. The effect of increasing age on thiopental disposition and anesthetic requirement. Anesthesiology. 1985;62:714.)
The influence of sedative–hypnotics on cerebral metabolism, cerebral hemodynamics, and ICP is of particular importance during neuroanesthesia. In patients with reduced cerebral compliance, a small increase in cerebral blood volume can cause a life-threatening increase in ICP. Most sedative–hypnotic drugs cause a proportional reduction in cerebral metabolism (CMRO2) and cerebral blood flow (CBF), resulting in a decrease in ICP. Although a decrease in CMRO2 probably provides only a modest degree of protection against CNS ischemia or hypoxia, some hypnotics appear to possess cerebroprotective potential (e.g., thiopental, propofol) (see also Chapter 36 Anesthesia for Neurosurgery). Explanations for the alleged neuroprotective effects of these compounds include a biochemical role as free radical scavengers and membrane stabilizers (barbiturates and propofol) or NMDA-receptor antagonists (ketamine). With the exception of ketamine, all sedative–hypnotics also lower intraocular pressure. The changes in intraocular pressure generally reflect the effects of the IV agent on systemic arterial pressure and intracranial hemodynamics. However, none of the available sedative–hypnotic drugs protect against the transient increase in intraocular pressure that occurs with laryngoscopy and tracheal intubation.
Most IV hypnotics have similar EEG effects. Activation of high-frequency EEG activity (15 to 30 Hz) is characteristic of low concentrations (so-called sedative doses) of IV anesthetics. At higher concentrations, an increase in the relative contribution of the lower frequency higher amplitude waves is observed. At high concentrations, a burst-suppressive pattern develops with an increase in the isoelectric periods. Most sedative–hypnotic drugs have been reported to cause occasional EEG seizure-like myoclonic activity. Interestingly, these same drugs also possess anticonvulsant properties.12,13 When considering possible epileptogenic properties of CNS-depressant drugs, it is important to differentiate between true epileptogenic activity (e.g., methohexital) and myoclonic-like phenomena (e.g., etomidate, ketamine, propofol). Myoclonic activity is generally considered to be the result of an imbalance between excitatory and inhibitory subcortical centers, produced by an unequal degree of suppression of these brain centers by low concentrations of hypnotic drugs. Epileptic activity refers to a sudden alteration in CNS seizure-like activity resulting from a high-voltage electrical discharge at either cortical or subcortical sites, with subsequent spreading to the thalamic and brainstem centers. As a result of its vasoconstrictive effects on the cerebral vasculature, propofol may be useful for treatment of intractable migraine headaches.14
Although some induction drugs can increase airway sensitivity, coughing and airway irritation (e.g., bronchospasm) are usually a result of manipulation of the airway during “light” (inadequate) levels of IV anesthesia rather than to a direct drug effect. With the exception of ketamine (and to a lesser extent, etomidate), IV anesthetics produce dose-dependent respiratory depression, which is enhanced in patients with chronic obstructive pulmonary disease. The respiratory depression is characterized by a decrease in tidal volume and minute ventilation, as well as a transient rightward shift in the CO2 response curve. Following the rapid injection of a large bolus dose of an IV anesthetic, transient apnea lasting 30 to 90 seconds is usually produced. Ketamine causes minimal respiratory depression when administered in the usual induction doses, while etomidate is associated with less respiratory depressant effects than the barbiturate compounds or propofol. The α2-agonist dexmedetomidine has minimal depressant effects on respiratory function.15 The sympatholytic effects of dexmedetomidine when administered for premedication may increase the incidence of intraoperative hypotension and bradycardia.16
Many different factors contribute to the hemodynamic changes associated with IV induction of anesthesia, including the patient’s preexisting cardiovascular and fluid status, resting sympathetic nervous system tone, chronic cardiovascular drugs, preanesthetic medication, the speed of drug injection, and the onset of unconsciousness. In addition, cardiovascular changes can be attributed to the direct pharmacologic actions of anesthetic and analgesic drugs on the heart and peripheral vasculature. IV anesthetics can depress the CNS and peripheral nervous system responses, blunt the compensatory baroreceptor reflex mechanisms, produce direct myocardial depression, and lower peripheral vascular resistance (and/or dilate venous capacitance vessels), thereby decreasing venous return. Profound hemodynamic effects occur at induction of anesthesia in the presence of hypovolemia because a higher than expected drug concentration is achieved in the central compartment. Not surprisingly, the acute cardiocirculatory depressant effects of all IV anesthetics are accentuated in the elderly, as well as in the presence of preexisting cardiovascular disease (e.g., coronary artery disease, chronic hypertension).
The effects of IV anesthetics on neuroendocrine function are also influenced by the surgical stimuli. Surgery-induced increases in stress hormones (e.g., vasopressin [ADH], catecholamines) can result in increased peripheral vascular resistance, and a reduction of urine output. Similarly, glucose tolerance appears to be decreased by surgical stress, resulting in elevations in the glucose concentration.  Unlike ketamine and dexmedetomidine, most IV sedative–hypnotic drugs lack intrinsic analgesic activity. In fact, thiopental has been alleged to possess so-called antianalgesic activity (i.e., appearing to lower the pain threshold). Although propofol possesses dose-dependent effects on thalamocortical transfer of nociceptive information, pain-evoked cortical activity remains intact after loss of consciousness.17
Unlike ketamine and dexmedetomidine, most IV sedative–hypnotic drugs lack intrinsic analgesic activity. In fact, thiopental has been alleged to possess so-called antianalgesic activity (i.e., appearing to lower the pain threshold). Although propofol possesses dose-dependent effects on thalamocortical transfer of nociceptive information, pain-evoked cortical activity remains intact after loss of consciousness.17
Hypersensitivity (Allergic) Reactions
Allergic or hypersensitivity-type reactions to IV anesthetics are rare but can be severe and even life-threatening. IV drug administration bypasses the normal “protective barriers” against entrance of foreign molecules into the body. With the exception of etomidate, all IV induction agents have been alleged to cause some histamine release. However, the incidence of severe anaphylactic reactions is extremely low with the currently available IV induction agents. The high frequency of allergic reactions to the Cremophor EL–containing formulations led to the early withdrawal of IV anesthetics containing this solubilizing agent (e.g., propofol EL, propanidid, Alphadione [Althesin]). The possible mechanisms for immunologic reactions include: (1) direct action on mast cells, (2) classic complement activation after previous exposure and antibody formation, (3) complement activation through the alternative pathway without previous antigen exposure, (4) antigen-antibody reactions, and (5) the “mixed type” of anaphylactoid reactions.
Severe anaphylactic reactions to IV anesthetics are extremely uncommon; however, profound hypotension attributed to nonimmunologically mediated histamine release has been reported with thiopental use. Although anaphylactic reactions to etomidate have been reported, it does not appear to release histamine, and is considered to be the most “immunologically safe” IV anesthetic. Propofol does not normally trigger histamine release, but life-threatening anaphylactoid reactions have been reported in patients with a previous history of multiple-drug allergies. Barbiturates can also precipitate episodes of acute intermittent porphyria and their use is contraindicated in predisposed patients. Although benzodiazepines, ketamine, and etomidate are reported to be safe in humans, these drugs have been shown to be porphyrogenic in animal models. The most common causes of profound hypotension following IV induction of anesthesia are unexpected drug interactions and/or unrecognized hypovolemia.
COMPARATIVE PHYSICOCHEMICAL AND CLINICAL PHARMACOLOGIC PROPERTIES OF INTRAVENOUS AGENTS
Barbiturates
The most commonly used barbiturates are thiopental (5-ethyl-5-[1-methylbutyl]-2-thiobarbituric acid), methohexital (1-methyl-5-allyl-5-[1-methyl-2-pentanyl] barbituric acid), and thiamylal (5-allyl-5-[1-methylbutyl]-2-thiobarbituric acid). Thiopental (Pentothal) and thiamylal (Surital) are thiobarbiturates, while methohexital (Brevital) is an oxybarbiturate. Thiamylal is slightly more potent than thiopental but has a similar pharmacologic profile. Although the l-isomers of thiopental and thiamylal are twice as potent as the d-isomers, both hypnotics are commercially available as racemic mixtures. Because methohexital has two asymmetric centers, it has four stereoisomers. The β-l-isomer is 4 to 5 times more potent than the α-l-isomer, but it produces excessive motor responses. Therefore, methohexital is marketed as the racemic mixture of the two α-isomers.
All three barbiturates are available as sodium salts and must be dissolved in isotonic sodium chloride (0.9%) or water to prepare solutions of 2.5% thiopental, 1% to 2% methohexital, and 2% thiamylal. If refrigerated, solutions of the thiobarbiturates are stable for up to 2 weeks. Solutions of methohexital are stable for up to 6 weeks. When barbiturates are added to Ringer lactate or an acidic solution containing other water-soluble drugs, precipitation will occur and can occlude the IV catheter. Although the typical solution of thiopental (2.5%) is highly alkaline (pH 9) and can be irritating to the tissues if injected extravenously, it does not cause pain on injection and venoirritation is rare. In contrast, a 1% methohexital solution frequently causes discomfort when injected into small veins. Intra-arterial injection of thiobarbiturates is a serious complication as crystals can form in the arterioles and capillaries, causing intense vasoconstriction, thrombosis, and even tissue necrosis. Accidental intra-arterial injections should be treated promptly with intra-arterial administration of papaverine and lidocaine (or procaine), as well as a regional anesthesia-induced sympathectomy (stellate ganglion block, brachial plexus block) and heparinization.
Thiopental is metabolized in the liver to hydroxythiopental and the carboxylic acid derivative, which are more water soluble and have little CNS activity. When high doses of thiopental are administered, a desulfuration reaction can occur with the production of pentobarbital, which has long-lasting CNS-depressant activity. The low elimination clearance of thiopental (3.4 mL/ kg/min) contributes to a long elimination half-life (t1/2β of 11 hours). Preexisting hepatic and renal disease result in decreased plasma protein binding, thereby increasing the free fraction of thiopental and enhancing its CNS and cardiovascular-depressant properties. During prolonged continuous administration of thiopental, the concentration in the tissues approaches the concentration in the central compartment, with termination of its CNS effects becoming solely dependent on elimination by nonlinear hepatic metabolism. Methohexital is metabolized in the liver to inactive hydroxyderivatives. The clearance of methohexital (11 mL/kg/min) is higher and more dependent on hepatic blood flow than thiopental, resulting in a shorter elimination half-life (t1/2β 4 hours).
The usual induction dose of thiopental is 3 to 5 mg/kg in adults, 5 to 6 mg/kg in children, and 6 to 8 mg/kg in infants. Because methohexital is approximately 2.7 times more potent than thiopental, a dose of 1.5 mg/kg is equivalent to 4 mg/kg of thiopental in adults. The dose of barbiturates necessary to induce anesthesia is reduced in premedicated patients, patients in early pregnancy (7 to 13 weeks’ gestation), and those of more advanced American Society of Anesthesiologists physical status (III or IV). Geriatric patients require a 30% to 40% reduction in the usual adult dose because of a decrease of the volume of the central compartment and slowed redistribution of thiopental from the vessel-rich tissues to lean muscle.18 When the calculation of the induction dose is based on the lean body mass rather than total body weight, dosage adjustments for age, sex, or obesity are not necessary. Thiopental infusion is seldom used to maintain anesthesia because of the long context-sensitive half-time and prolonged recovery period. Plasma thiopental levels necessary to maintain a hypnotic state range between 10 and 20 mg/mL. A typical infusion rate necessary to treat intracranial hypertension or intractable convulsions is 2 to 4 mg/kg/hr. The plasma concentration of methohexital needed to maintain hypnosis during anesthesia ranges between 3 and 5 mg/mL and can be achieved with an infusion rate of methohexital 50 to 120 μg/kg/min.
Barbiturates produce a proportional decrease in CMRO2 and CBF, thereby lowering ICP. The maximal decrease in CMRO2 (55%) occurs when the EEG becomes isoelectric (burst-suppressive pattern). An isoelectric EEG can be maintained with a thiopental infusion rate of 4 to 6 mg/kg/hr (resulting in plasma concentrations of 30 to 50 μg/mL). Because the decrease in systemic arterial pressure is usually less than the reduction in ICP, thiopental should improve cerebral perfusion and compliance. Therefore, thiopental is widely used to improve brain relaxation during neurosurgery and to improve cerebral perfusion pressure (CPP) after acute brain injury. Although barbiturate therapy is widely used to control ICP after brain injury, the results of outcome studies are no better than with other aggressive forms of cerebral antihypertensive therapy.
It has been suggested that barbiturates also possess “neuroprotective” properties secondary to their ability to decrease oxygen demand. Alternative explanations have been suggested, including a reverse steal (“Robin Hood effect”) on CBF, free-radical scavenging, stabilization of liposomal membranes, as well as excitatory amino acid receptor blockade. On the basis of the evidence from experimental studies and a large randomized prospective multi-institutional study,19 experts have concluded that barbiturates have no place in the therapy following resuscitation of a cardiac arrest patient. In contrast, barbiturates are frequently used for cerebroprotection during incomplete brain ischemia (e.g., carotid endarterectomy, temporary occlusion of cerebral arteries, profound hypotension, and cardiopulmonary bypass). By improving the brain’s tolerance of incomplete ischemia in patients undergoing open heart surgery with cardiopulmonary bypass, barbiturates were alleged to decrease the incidence of postbypass neuropsychiatric disorders.20 However, during valvular open heart cardiac surgery, a protective effect of barbiturate loading could not be demonstrated.21 Given the lack of demonstrable neuroprotective effect, use of barbiturates during cardiac surgery is not recommended. Use of moderate degrees of hypothermia (33–34°C) might provide superior neuroprotection to the barbiturates without prolonging recovery.
Barbiturates cause predictable, dose-dependent EEG changes and possess potent anticonvulsant activity. Continuous infusions of thiopental have been used to treat refractory status epilepticus. However, low doses of thiopental may induce spike wave activity in epileptic patients. Methohexital has well-established epileptogenic effects in patients with psychomotor epilepsy. Low-dose methohexital infusions are frequently used to activate cortical EEG seizure discharges in patients with temporal lobe epilepsy.  It is also the IV anesthetic of choice for electroconvulsive therapy22 because, compared to thiopental and propofol, methohexital produces less depression of EEG activity. Since the frequency of epileptiform EEG activity during induction of anesthesia with methohexital is significantly less than that which occurs during normal periods of sleep in epileptic patients, this suggests that higher doses of methohexital produces anticonvulsant activity. Methohexital also causes myoclonic-like muscle tremors and other signs of excitatory activity (e.g., hiccoughing).
It is also the IV anesthetic of choice for electroconvulsive therapy22 because, compared to thiopental and propofol, methohexital produces less depression of EEG activity. Since the frequency of epileptiform EEG activity during induction of anesthesia with methohexital is significantly less than that which occurs during normal periods of sleep in epileptic patients, this suggests that higher doses of methohexital produces anticonvulsant activity. Methohexital also causes myoclonic-like muscle tremors and other signs of excitatory activity (e.g., hiccoughing).
Barbiturates cause dose-dependent respiratory depression.23 However, bronchospasm or laryngospasm following induction with thiopental is usually the result of airway manipulation in “lightly” anesthetized patients. Laryngeal reflexes appear to be more active after induction with thiopental than with propofol. The cardiovascular effects of thiopental and methohexital include decreases in cardiac output, systemic arterial pressure, and peripheral vascular resistance. The depressant effects of thiopental on cardiac output are primarily a result of a decrease in venous return caused by peripheral pooling, as well as a result of a direct myocardial depressant effect, which assumes increasing importance in the presence of hypovolemia and myocardial disease.24 Use of appropriate doses can minimize the cardiodepressant effects of thiopental, even in infants.25 An equipotent dose of methohexital produces even less hypotension than thiopental because of a greater tachycardic response to the blood pressure-lowering effects of the drug. If the blood pressure remains stable, the myocardial oxygen demand/supply ratio remains normal despite the increase in heart rate because of a concurrent decrease in coronary vascular resistance.
Propofol
Propofol (2,6-disopropylphenol), an alkylphenol compound, is virtually insoluble in aqueous solution. The initial Cremophor EL formulation of propofol was withdrawn from clinical testing because of the high incidence of anaphylactic reactions. Subsequently, propofol (10 mg/mL) was reintroduced as an egg lecithin emulsion formulation (Diprivan), consisting of 10% soybean oil, 2.25% glycerol, and 1.2% egg phosphatide. Microbial contamination of the original Diprivan formulation lead the manufacturer to add ethylenediaminetetraacetic acid (EDTA) to the propofol solution. With this formulation, injection pain occurs in 32% to 67% of patients when the drug is injected into small hand veins. This side effect can be minimized by injecting propofol into larger veins and by prior administration of either lidocaine or a potent opioid analgesic (e.g., fentanyl or remifentanil). A wide variety of drugs have been alleged to reduce pain on injection of propofol [e.g., metoprolol,26 granisetron,27 dolasetron,28 and even thiopental29]. Diluting the formulation with additional solvent (Intralipid) or changing the lipid carrier (Lipofundin) also reduced propofol-induced injection pain, probably because of a decrease in the concentration of free propofol in the aqueous phase of the emulsion. A new propofol formulation with sodium metabisulphite (instead of EDTA) as an antimicrobial has been shown to be associated with less severe pain on injection.30 Although the presence of the metabisulphite has raised concerns regarding its use in sulphite-allergic patients, this concern does not appear to be a clinically important problem. Of interest, a 2% formulation is available for long-term sedation to decrease the fluid (and lipid) volume.
A lower-lipid formulation of propofol (Ampofol) was introduced into clinical practice for both general anesthesia31 and sedation.32 The increased “free” fraction of propofol leads to increased pain when it is injected into small veins. Therefore, it is important to add lidocaine to the Ampofol formulation to minimize the pain on injection. A water-soluble prodrug of propofol, fospropofol (aka Aquavan or Lusedra), was approved by the FDA for use during local and regional anesthesia (e.g., conscious sedation and/or monitored anesthesia care [MAC]). This prodrug is rapidly hydrolyzed by plasma alkaline phosphatases in the circulation to release free propofol.33 Therefore, it does not produce the typical pain on injection associated with propofol. However, it has a slower onset than propofol34 and is frequently associated with a transient severe burning sensation in the perineal region immediately following IV injection. A microemulsion formulation of propofol (Aquafol) has been shown to be as effective and safe as the classical propofol formulation with similar pharmokinetics, but is associated with more severe and frequent injection pain.35 A new lipid-free preparation of propofol has been developed containing the drug, sulfobutylether β-cyclodextrin and water.36 Unfortunately, propofol in cyclodextrin (Captisol-enabled propofol) also produced significantly higher injection pain scores than the standard formulation.
Propofol’s pharmacokinetics has been studied using single-bolus dosing and continuous infusions.37 In studies using a two-compartment kinetic model, the initial distribution half-life is 2 to 4 minutes and the elimination half-life is 1 to 3 hours. Using a three-compartment model, the initial and slow distribution half-life values are 1 to 8 minutes and 30 to 70 minutes, respectively. The elimination half-life depends largely on the sampling time after discontinuing the administration of propofol and ranges from 2 to 24 hours. This long elimination half-life is indicative of the existence of a poorly perfused compartment from which propofol slowly diffuses back into the central compartment. Propofol is rapidly cleared from the central compartment by hepatic metabolism and the context-sensitive half-life for propofol infusions up to 8 hours is <40 minutes. Propofol is rapidly and extensively metabolized to inactive, water-soluble sulphate and glucuronic acid metabolites, which are eliminated by the kidneys. The clearance rate of propofol (20 to 30 mL/kg/min) exceeds hepatic blood flow, suggesting that an extrahepatic route of elimination (lungs) also contributes to its clearance. Nevertheless, changes in liver blood flow would be expected to produce marked alterations in the clearance rate of propofol. Surprisingly, few changes in propofol’s pharmacokinetics have been reported in the presence of hepatic or renal disease.
The induction dose of propofol in healthy adults is 1.5 to 2.5 mg/kg, with blood levels of 2 to 6 μg/mL producing unconsciousness depending on the concomitant medications (e.g., opioid analgesics), the patient’s age and physical status, and the extent of the surgical stimulation.38 In the morbidly obese patient, the propofol induction and maintenance dosages should be calculated on the basis of patient’s lean body weight.39 In one of the first reports describing the use of propofol for induction and maintenance of anesthesia with nitrous oxide, an average infusion rate of 120 μg/kg/min was used.40 The recommended maintenance infusion rate of propofol varies between 100 and 200 μg/kg/min for hypnosis and between 25 and 75 μg/kg/min for sedation. Awakening typically occurs at plasma propofol concentrations of 1 to 1.5 μg/mL.41 Because a 50% decrease in the plasma propofol concentration is usually required for awakening, emergence following anesthesia is usually rapid even following more prolonged infusions (>3 hours).
Analogous to the barbiturates, children require higher induction and maintenance doses of propofol on a milligram per kilogram basis as a result of their larger central distribution volume and higher clearance rate. Elderly patients and those in poor health require lower induction and maintenance doses of propofol as a result of their smaller central distribution volume and decreased clearance rate. Although subhypnotic doses of propofol produce sedation and amnesia,41 awareness has been reported even at higher infusion rates when propofol is used as the sole anesthetic.42 Propofol often produces a subjective feeling of well-being (and even euphoria) on emergence, and it has been abused by health care professionals and others as a result of this CNS action.43
Propofol decreases CMRO2 and CBF, as well as ICP.44 However, when larger doses are administered, the marked depressant effect on systemic arterial pressure can significantly decrease CPP. Cerebrovascular autoregulation in response to changes in systemic arterial pressure and reactivity of the cerebral blood flow to changes in carbon dioxide tension are not affected by propofol. Evidence for a possible neuroprotective effect has been reported with in vitro preparations, and the use of propofol to produce EEG burst suppression has been proposed as a method for providing neuroprotection during aneurysm surgery. Its neuroprotective effect may at least partially be related to the antioxidant potential of propofol’s phenol ring structure, which may act as a free radical scavenger, decreasing free radical–induced lipid peroxidation. A recent study reported that this antioxidant activity may offer many advantages in preventing the hypoperfusion–reperfusion phenomenon that can occur during major laparoscopic surgery.45  Although TIVA with propofol and an opioid analgesic is a safe and effective alternative to standard inhalation techniques (i.e., volatile anesthetic with nitrous oxide) for maintenance of anesthesia, concerns have been raised regarding the cost-effectiveness of this technique.46
Although TIVA with propofol and an opioid analgesic is a safe and effective alternative to standard inhalation techniques (i.e., volatile anesthetic with nitrous oxide) for maintenance of anesthesia, concerns have been raised regarding the cost-effectiveness of this technique.46
Propofol produces cortical EEG changes that are similar to those of thiopental. However, sedative doses of propofol increase β-wave activity analogous to the benzodiazepines. Induction of anesthesia with propofol is occasionally accompanied by excitatory motor activity (so-called nonepileptic myoclonia). In a study involving patients without a history of seizure disorders, excitatory movements following propofol were not associated with EEG seizure activity.47 Propofol appears to possess profound anticonvulsant properties.48 Propofol has been reported to decrease spike activity in patients with cortical electrodes implanted for resection of epileptogenic foci and has been used successfully to terminate status epilepticus. The duration of motor and EEG seizure activity following ECT is significantly shorter with propofol than with other IV anesthetics. Propofol produces a decrease in the early components of somatosensory and motor-evoked potentials but does not influence the early components of the auditory-evoked potentials.
Propofol produces dose-dependent respiratory depression, with apnea occurring in 25% to 35% of patients after a typical induction dose. A maintenance infusion of propofol decreases tidal volume and increases respiratory rate. The ventilatory response to carbon dioxide and hypoxia is also significantly decreased by propofol. Propofol can produce bronchodilation in patients with chronic obstructive pulmonary disease and does not inhibit hypoxic pulmonary vasoconstriction.
The cardiovascular depressant effects of propofol are generally considered to be more profound than those of thiopental. Both direct myocardial depressant effects and decreased systemic vascular resistance have been implicated as important factors in producing cardiovascular depression. Direct myocardial depression and peripheral vasodilation are dose- and concentration-dependent. In addition to arterial vasodilation, propofol produces venodilation (caused both to a reduction in sympathetic activity and by a direct effect on the vascular smooth muscle), which further contributes to its hypotensive effect. The relaxation of the vascular smooth muscle may be because of an effect on intracellular calcium mobilization or because of an increase in the production of nitric oxide. Experiments in isolated myocardium suggest that the negative inotropic effect of propofol results from a decrease in intracellular calcium availability secondary to inhibition of transsarcolemmal calcium influx. A recent study in rats has suggested that an altered renin gene is responsible for enhanced sensitivity and hypotensive responses to propofol.49
Propofol also alters the baroreflex mechanism, resulting in a smaller increase in heart rate for a given decrease in arterial pressure.50 The smaller increase in heart rate with propofol may account for the larger decrease in arterial pressure than with an equipotent dose of thiopental. Recent data suggest that induction of anesthesia with propofol attenuates desflurane-mediated sympathetic activation.51 Age enhances the cardiodepressant response to propofol and a reduced dosage is required in studies of the elderly. Patients with limited cardiac reserve seem to tolerate the cardiac depression and systemic vasodilation produced by carefully titrated doses of propofol, and maintenance infusions are increasingly used at the end of cardiac surgery when early extubation is desired.
Propofol appears  to possess antiemetic properties that contribute to a lower incidence of emetic sequelae after general anesthesia.40 In fact, subanesthetic doses of propofol (10 to 20 mg bolus doses) have also been successfully used to treat nausea and emesis in the early postoperative period.52 The postulated mechanisms include antidopaminergic activity, depressant effect on the chemoreceptor trigger zone and vagal nuclei, decreased release of glutamate and aspartate in the olfactory cortex, and reduction of serotonin concentrations in the area postrema. However, the ability of propofol to produce a sense of well-being may also contribute to its antiemetic action. Interestingly, propofol also decreases the pruritus produced by spinal opioids.
to possess antiemetic properties that contribute to a lower incidence of emetic sequelae after general anesthesia.40 In fact, subanesthetic doses of propofol (10 to 20 mg bolus doses) have also been successfully used to treat nausea and emesis in the early postoperative period.52 The postulated mechanisms include antidopaminergic activity, depressant effect on the chemoreceptor trigger zone and vagal nuclei, decreased release of glutamate and aspartate in the olfactory cortex, and reduction of serotonin concentrations in the area postrema. However, the ability of propofol to produce a sense of well-being may also contribute to its antiemetic action. Interestingly, propofol also decreases the pruritus produced by spinal opioids.
Propofol does not trigger malignant hyperthermia and may be considered the induction agent of choice in malignant hyperthermia-susceptible patients. The use of propofol infusions for sedation in the pediatric ICU has been linked to several deaths following prolonged administration because of lipid accumulation and hypotension. Although clinical doses of propofol do not affect cortisol synthesis or the response to adrenocorticotropic hormone stimulation, propofol has been reported to inhibit phagocytosis and killing of bacteria in vitro and to reduce proliferative responses when added to lymphocytes from critically ill patients.53 Because fat emulsions are known to support the growth of microorganisms, contamination can occur as a result of dilution or fractionated use.54
In critically ill children and adults receiving high-dose infusions of propofol, some patients have been reported to experience so-called propofol infusion syndrome (PRIS), which is characterized by cardiac failure, rhabdomyolysis, metabolic acidosis, and renal failure and is often fatal.55 Hyperkalemia, hypertriglyceridemia, and hepatomegaly are also key features. It is associated with high doses and long-term use of propofol (>4 mg/kg/hr for >24 hours). It occurs more commonly in children, and critically ill patients receiving catecholamines and glucocorticoids.
Benzodiazepines
The parenteral benzodiazepines include diazepam (Valium), lorazepam (Ativan), and midazolam (Versed), as well as the antagonist flumazenil (Romazicon). Diazepam and lorazepam are insoluble in water and their formulation contains propylene glycol, a tissue irritant that causes pain on injection and venous irritation. Diazepam is available in a lipid emulsion formulation, which does not cause pain or thrombophlebitis but is associated with a slightly lower bioavailability. Midazolam is a water-soluble benzodiazepine that is available in an acidified (pH 3.5) aqueous formulation that produces minimal local irritation after IV or intramuscular (IM) injection.56 At physiologic pH, an intramolecular rearrangement occurs that changes the physicochemical properties of midazolam such that it becomes more lipid soluble.
Benzodiazepines undergo hepatic metabolism via oxidation and glucuronide conjugation. Oxidation reactions are susceptible to hepatic dysfunction and coadministration of other anesthetic drugs. Diazepam is metabolized to active metabolites (desmethyldiazepam, 3-hydroxydiazepam), which can prolong the residual sedative effects of diazepam because of their long t1/2β values. These metabolites undergo secondary conjugation to form inactive water-soluble glucuronide conjugates. Drugs that inhibit the oxidative metabolism of diazepam include the H2-receptor blocking drug cimetidine. Severe liver disease reduces diazepam’s protein-binding and hepatic-clearance rate, increases its volume of distribution, and thereby further prolongs the t1/2β value. Chronic renal disease decreases protein binding and increases the free drug fraction, resulting in enhanced hepatic metabolism and a shorter t1/2β value. In elderly patients, the clearance rate of diazepam is significantly decreased, with a t1/2β of 75 to 150 hours.
Lorazepam is directly conjugated to glucuronic acid to form pharmacologically inactive metabolites. Age and renal disease have little influence on the kinetics of lorazepam; however, severe hepatic disease decreases its clearance rate. Midazolam undergoes extensive oxidation by hepatic enzymes to form water-soluble hydroxylated metabolites, which are excreted in the urine. However, the primary metabolite, 1-hydroxymethylmidazolam, has mild CNS-depressant activity. The hepatic clearance rate of midazolam is five times greater than lorazepam and ten times greater than diazepam. Although changes in liver blood flow can affect the clearance of midazolam, age has relatively little influence on the elimination half-life of midazolam.
The benzodiazepines used in anesthesia are classified as either short (midazolam, flumazenil), intermediate (diazepam), or long acting (lorazepam). Since the distribution volumes are similar, the large difference in the elimination half-times is because of differences in their differing clearance rates (Table 18-1). The context-sensitive half-times for diazepam and lorazepam are very long; therefore, only midazolam should be used by continuous infusion to avoid excessive accumulation.
All benzodiazepines  produce dose-dependent anxiolytic, anterograde amnestic, sedative, hypnotic, anticonvulsant, and spinally mediated muscle relaxant properties. Benzodiazepines differ in potency and efficacy with regard to their distinctive pharmacologic properties.56 The dose-dependent pharmacologic activity implies that the CNS effects of various benzodiazepine compounds depend on the affinity for receptor subtypes and their degree of receptor binding. Although benzodiazepines can be used as hypnotics, they are primarily used as premedicants and adjuvant drugs because of their anxiolytic, sedative, and amnestic properties. For example, midazolam (0.02 to 0.04 mg/kg IV/IM) is the most commonly used premedicant. In addition, midazolam, 0.4 to 0.8 mg/kg administered orally 10 to 15 minutes before parental separation, is an excellent premedicant in children. In contrast to lorazepam, both diazepam and midazolam can be used to induce anesthesia because they have a relatively short onset time after IV administration. The half-life of equilibration between the plasma concentration of midazolam and its maximal EEG effect is only 2 to 3 minutes. The therapeutic window to maintain unconsciousness with midazolam is reported to be 100 to 200 ng/mL, with awakening occurring at plasma concentrations below 50 ng/mL. However, significant hypnotic synergism occurs when midazolam and opioid analgesics are administered in combination.
produce dose-dependent anxiolytic, anterograde amnestic, sedative, hypnotic, anticonvulsant, and spinally mediated muscle relaxant properties. Benzodiazepines differ in potency and efficacy with regard to their distinctive pharmacologic properties.56 The dose-dependent pharmacologic activity implies that the CNS effects of various benzodiazepine compounds depend on the affinity for receptor subtypes and their degree of receptor binding. Although benzodiazepines can be used as hypnotics, they are primarily used as premedicants and adjuvant drugs because of their anxiolytic, sedative, and amnestic properties. For example, midazolam (0.02 to 0.04 mg/kg IV/IM) is the most commonly used premedicant. In addition, midazolam, 0.4 to 0.8 mg/kg administered orally 10 to 15 minutes before parental separation, is an excellent premedicant in children. In contrast to lorazepam, both diazepam and midazolam can be used to induce anesthesia because they have a relatively short onset time after IV administration. The half-life of equilibration between the plasma concentration of midazolam and its maximal EEG effect is only 2 to 3 minutes. The therapeutic window to maintain unconsciousness with midazolam is reported to be 100 to 200 ng/mL, with awakening occurring at plasma concentrations below 50 ng/mL. However, significant hypnotic synergism occurs when midazolam and opioid analgesics are administered in combination.
The usual induction dose of midazolam in premedicated patients is 0.1 to 0.2 mg/kg IV, with infusion rates of 0.25 to 1.0 μg/kg/min required to maintain hypnosis and amnesia in combination with inhalational agents and/or opioid analgesics. Higher maintenance infusion rates and prolonged administration will result in accumulation and prolonged recovery times. Lower infusion rates are sufficient to provide sedation and amnesia during local and regional anesthesia.57 Patient-controlled administration of midazolam during procedures under local anesthesia is well accepted by patients and associated with few perioperative complications.58
Benzodiazepines decrease both CMRO2 and CBF analogous to the barbiturates and propofol. However, in contrast to these compounds, midazolam is unable to produce a complete burst-suppressive (isoelectric) pattern on the EEG. Accordingly, there is a “ceiling” effect with respect to the decrease in CMRO2 produced by increasing doses of midazolam. Midazolam produces a dose-related decrease in regional cerebral perfusion in the parts of the brain that subserve arousal, attention, and memory. Cerebral vasomotor responsiveness to carbon dioxide is preserved during midazolam anesthesia. In patients with severe head injury, a bolus dose of midazolam may decrease CPP with little effect on ICP. Although midazolam may improve neurologic outcome after incomplete ischemia in animal experiments, benzodiazepines have not been shown to possess neuroprotective activity in humans. Like the other sedative–hypnotic drugs, the benzodiazepines are potent anticonvulsants that are commonly used to treat status epilepticus.
Benzodiazepines produce dose-dependent respiratory depression. In healthy patients, the respiratory depression associated with benzodiazepine premedication is insignificant. However, the depressant effect is enhanced in patients with chronic respiratory disease, and synergistic depressant effects occur when benzodiazepines are coadministered with opioid analgesics. Benzodiazepines also depress the swallowing reflex and decrease upper airway reflex activity.
Both midazolam and diazepam produce decreases in systemic vascular resistance and blood pressure when large doses are administered for induction of anesthesia. However, the cardiovascular depressant effects of benzodiazepines are frequently “masked” by the stimulus of laryngoscopy and intubation. The cardiovascular depressant effects are directly related to the plasma concentration; however, a plateau plasma concentration appears to exist above which little further change in arterial blood pressure occurs. In the presence of heart failure, the decrease in preload and afterload produced by benzodiazepines may be beneficial in improving cardiac output. However, the cardiodepressant effect of benzodiazepines may be more marked in hypovolemic patients.
An investigational short-acting intravenous sedative, Ro 48–6791, is a water-soluble benzodiazepine that has full agonistic activity at CNS benzodiazepine receptors. Compared with midazolam, it is 2- to 2.5-fold more potent, has a higher plasma clearance rate, and has a similar onset and duration of action.59 In a study involving outpatients undergoing endoscopy procedures, the times to ambulation and to recovery from psychomotor impairment were decreased compared to midazolam, although the later recovery end points (e.g., “fitness-for-discharge”) were similar.60
In contrast to all other sedative–hypnotic drugs, there is a specific antagonist for benzodiazepines. Flumazenil, a 1,4-imidazobenzodiazepine derivative, has a high affinity for the benzodiazepine receptor but minimal intrinsic activity.61 Flumazenil’s molecular structure is similar to other benzodiazepines except for the absence of a phenyl group, which is replaced by a carbonyl group. It is water soluble and possesses moderate lipid solubility at physiologic pH. Flumazenil is rapidly metabolized in the liver, and its metabolites are excreted in the urine as glucuronide conjugates. Flumazenil acts as a competitive antagonist in the presence of benzodiazepine agonist compounds. The residual activity of the benzodiazepines in the presence of flumazenil depends on the relative concentrations of the agonist and antagonist drugs. As a result, it is possible to reverse benzodiazepine-induced anesthesia (or deep sedation) either completely or partially, depending on the dose of flumazenil (0.5–1 mg IV). Flumazenil is short acting, with an elimination half-life of ∼1 hour.
Recurrence of the central effects of benzodiazepines (resedation) may occur after a single dose of flumazenil because of residual effects of the more slowly eliminated agonist drug.62 If sustained antagonism is desired, it may be necessary to administer flumazenil as repeated bolus doses or a continuous infusion. In general, 45 to 90 minutes of antagonism can be expected following flumazenil 1 to 3 mg IV. However, the respiratory depression produced by benzodiazepines is not completely reversed by flumazenil.63 Reversal of benzodiazepine sedation with flumazenil is not associated with adverse cardiovascular effects or evidence of an acute stress response.64 Although flumazenil does not appear to change CBF or CMRO2 following midazolam anesthesia for craniotomy, acute increases in ICP have been reported in head-injured patients receiving flumazenil.
Etomidate
Etomidate is a carboxylated imidazole-containing anesthetic compound (R-1-ethyl-1-[a-methylbenzyl] imidazole-5-carboxylate) that is structurally unrelated to any other IV anesthetic. Only the d-isomer of etomidate possesses anesthetic activity. Analogous to midazolam (which also contains an imidazole nucleus), etomidate undergoes an intramolecular rearrangement at physiologic pH, resulting in a closed-ring structure with enhanced lipid solubility. The aqueous solution of etomidate (Amidate) is unstable at physiologic pH and is formulated in a 0.2% solution with 35% propylene glycol (pH 6.9), contributing to a high incidence of pain on injection, venoirritation, and hemolysis. A new lipid emulsion formulation (Etomidate-Lipuro) has been used in Europe for a number of years and appears to be associated with a lower incidence of side effects compared with the original propylene glycol formulation of etomidate.
The standard induction dose of etomidate (0.2 to 0.3 mg/kg IV) produces a rapid onset of anesthesia. Involuntary myoclonic movements are common during the induction period as a result of subcortical disinhibition and are unrelated to cortical seizure activity. The frequency of this myoclonic-like activity can be attenuated by prior administration of opioid analgesics, benzodiazepines, or small sedative doses (0.03 to 0.05 mg/kg) prior to induction of anesthesia.65 Recently, remifentanil reduced etomidate-induced myoclonic activity without increasing side effects like apnea, emesis, or pruritus.66 The notion of utilizing a priming dose to reduce the incidence of myclonus has recently been disproven.67 Emergence time after etomidate anesthesia is dose-dependent but remains short even after administration of repeated bolus doses or continuous infusions. For maintenance of hypnosis, the target concentration is 300 to 500 ng/mL and can be rapidly achieved by administering a two- or three-stage infusion (e.g., 100 mg/kg/min for 10 minutes followed by 10 mg/kg/min or 100 mg/kg/min for 3 to 5 minutes, followed by 20 mg/kg/min for 20–30 minutes, and then 10 mg/kg/min). The pharmacokinetics of etomidate are optimally described by a three-compartment open model.68 The high clearance rate of etomidate (18 to 25 mL/kg/min) is a result of extensive ester hydrolysis in the liver (forming inactive water-soluble metabolites). A significant decrease in plasma protein binding has been reported in the presence of uremia and hepatic cirrhosis. Severe hepatic disease causes a prolongation of the elimination half-life secondary to an increased volume of distribution and a decreased plasma clearance rate.
Analogous to the barbiturates, etomidate decreases CMRO2, CBF, and ICP. However, the hemodynamic stability associated with etomidate will maintain adequate CPP. Etomidate has been used successfully for both induction and maintenance of anesthesia for neurosurgery. The well-known inhibitory effect of etomidate on adrenocortical synthetic function69 limits its clinical usefulness for long-term treatment of elevated ICP. Although clear evidence for a neuroprotective effect in humans is lacking, etomidate is frequently used during temporary arterial occlusion and intraoperative angiography (for the treatment of cerebral aneurysms). Etomidate produces an EEG pattern that is similar to thiopental except for the absence of increased β-activity at lower doses. Etomidate has been alleged to produce convulsion-like EEG potentials in epileptic patients without the appearance of myoclonic or convulsant-like motor activity, a property that has been proven useful for intraoperative mapping of seizure foci. Analogous to methohexital, etomidate possesses anticonvulsant properties and has been used to terminate status epilepticus. Etomidate also produces a significant increase of the amplitude of somatosensory-evoked potentials while only minimally increasing their latency. Consequently, etomidate can be used to facilitate the interpretation of somatosensory-evoked potentials when the signal quality is poor.
Etomidate causes  minimal cardiorespiratory depression even in the presence of cardiovascular and pulmonary disease.70 The drug does not induce histamine release and can be safely used in patients with reactive airway disease. Consequently, etomidate is considered to be the induction agent of choice for poor-risk patients with cardiorespiratory disease, as well as in those situations in which preservation of a normal blood pressure is crucial (e.g., cerebrovascular disease). However, etomidate does not effectively blunt the sympathetic response to laryngoscopy and intubation unless combined with a potent opioid analgesic.
minimal cardiorespiratory depression even in the presence of cardiovascular and pulmonary disease.70 The drug does not induce histamine release and can be safely used in patients with reactive airway disease. Consequently, etomidate is considered to be the induction agent of choice for poor-risk patients with cardiorespiratory disease, as well as in those situations in which preservation of a normal blood pressure is crucial (e.g., cerebrovascular disease). However, etomidate does not effectively blunt the sympathetic response to laryngoscopy and intubation unless combined with a potent opioid analgesic.
Etomidate is associated with a high incidence of postoperative nausea and emesis when used in combination with opioids for brief outpatient procedures. In addition, the increased mortality in critically ill patients sedated with an etomidate infusion has been attributed to its inhibitory effect on cortisol synthesis. Etomidate inhibits the activity of 11-β-hydroxylase, an enzyme necessary for the synthesis of cortisol, aldosterone, 17-hydroxyprogesterone, and corticosterone. Even after a single induction dose of etomidate,71 adrenal suppression persists for 5 to 8 hours. Although the clinical significance of short-term blockade of cortisol synthesis is not known, the use of etomidate for maintenance of anesthesia has been questioned. Recently, etomidate has been reported to inhibit platelet function, resulting in prolongation of the bleeding time.72 In spite of its side effect profile, etomidate remains a valuable induction drug for specific indications (e.g., in patients with severe cardiovascular and cerebrovascular disease).
Ketamine
Ketamine (Ketalar or Ketaject) is an arylcyclohexylamine that is structurally related to phencyclidine.73 Ketamine is a water-soluble compound with a pKa of 7.5 and is available in 1%, 5%, and 10% aqueous solutions. The ketamine molecule contains a chiral center producing two optical isomers. The S(+) isomer of ketamine possesses more potent anesthetic and analgesic properties despite having a similar pharmacokinetic and pharmacodynamic profile as the racemic mixture (or the R[–] isomer).74,75 Although the S(+)-ketamine has been in clinical use in Europe for many years, the most commonly used solution in the remainder of the world remains the racemic mixture of the two isomers. Ketamine is extensively metabolized by hepatic microsomal cytochrome P450 enzymes and its primary metabolite, norketamine, is one-third to one-fifth as potent as the parent compound. The metabolites of norketamine are excreted by the kidney as water-soluble hydroxylated and glucuronidated conjugates. Analogous to the barbiturates and propofol, ketamine has relatively short distribution and redistribution half-life values. Ketamine also has a high hepatic clearance rate (1 L/min) and a large distribution volume (3 L/kg), resulting in an elimination half-life of 2 to 4 hours. The high hepatic extraction ratio suggests that alterations in hepatic blood flow can significantly influence the clearance rate of ketamine.
Ketamine produces  dose-dependent CNS depression leading to a so-called dissociative anesthetic state characterized by profound analgesia and amnesia, even though patients may be conscious and maintain protective reflexes. The proposed mechanism for this cataleptic state includes electrophysiologic inhibition of thalamocortical pathways and stimulation of the limbic system. Although it is most commonly administered parenterally, oral and intranasal administration of ketamine (6 mg/kg) has been used for premedication of pediatric patients. Following benzodiazepine premedication, ketamine 1 to 2 mg/kg IV (or 4 to 8 mg/kg IM) can be used for induction of anesthesia. The duration of ketamine-induced anesthesia is in the range of 10 to 20 minutes after a single induction dose; however, recovery to full orientation may require an additional 60 to 90 minutes. Emergence times are even longer following repeated bolus injections or a continuous infusion. S(+)-ketamine has a shorter recovery time compared with the racemic mixture. The therapeutic window for maintenance of unconsciousness with ketamine is between 0.6 and 2 μg/mL in adults and between 0.8 and 4 μg/mL in children. Analgesic effects are evident at subanesthetic doses of 0.1 to 0.5 mg/kg IV and plasma concentrations of between 85 and 160 ng/mL. Adjunctive use of a low-dose ketamine infusion (1 to 3 μg/kg/min IV) during the 24 to 48 hour period after major surgery produces significant opioid-sparing effects and decreases opioid-related gastrointestinal side effects.76,77
dose-dependent CNS depression leading to a so-called dissociative anesthetic state characterized by profound analgesia and amnesia, even though patients may be conscious and maintain protective reflexes. The proposed mechanism for this cataleptic state includes electrophysiologic inhibition of thalamocortical pathways and stimulation of the limbic system. Although it is most commonly administered parenterally, oral and intranasal administration of ketamine (6 mg/kg) has been used for premedication of pediatric patients. Following benzodiazepine premedication, ketamine 1 to 2 mg/kg IV (or 4 to 8 mg/kg IM) can be used for induction of anesthesia. The duration of ketamine-induced anesthesia is in the range of 10 to 20 minutes after a single induction dose; however, recovery to full orientation may require an additional 60 to 90 minutes. Emergence times are even longer following repeated bolus injections or a continuous infusion. S(+)-ketamine has a shorter recovery time compared with the racemic mixture. The therapeutic window for maintenance of unconsciousness with ketamine is between 0.6 and 2 μg/mL in adults and between 0.8 and 4 μg/mL in children. Analgesic effects are evident at subanesthetic doses of 0.1 to 0.5 mg/kg IV and plasma concentrations of between 85 and 160 ng/mL. Adjunctive use of a low-dose ketamine infusion (1 to 3 μg/kg/min IV) during the 24 to 48 hour period after major surgery produces significant opioid-sparing effects and decreases opioid-related gastrointestinal side effects.76,77
As a result of its NMDA-receptor blocking activity, ketamine should be highly effective for “preemptive” analgesia and opioid-resistant chronic pain states.78 Unfortunately, a well-controlled study failed to demonstrate a preemptive effect when ketamine was administered prior to the surgical incision (vs. intraoperatively).79 Nevertheless, other studies80,81 described a beneficial opioid-sparing effect of small doses of ketamine (75 to 150 μg/kg IV) when administered as an adjuvant during surgery.
An important consideration in the use of ketamine anesthesia relates to the high incidence of psychomimetic reactions (namely, hallucinations, nightmares, altered short-term memory, and cognition) during the early recovery period. The incidence of these reactions is dose-dependent and can be reduced by coadministration of benzodiazepines, barbiturates, or propofol. Ketamine has been traditionally contraindicated for patients with increased ICP or reduced cerebral compliance because it increases CMRO2, CBF, and ICP. However, there is recent evidence that IV induction doses of ketamine actually decrease ICP in traumatic brain injury patients during controlled ventilation with propofol sedation.82 Prior administration of thiopental or benzodiazepines can blunt ketamine-induced increases in CBF. Because ketamine has antagonistic activity at the NMDA receptor, it may possess some inherent protective effects against brain ischemia. However, ketamine can adversely affect neurologic outcome in the presence of brain ischemia despite its NMDA-receptor blocking activity. Cortical EEG recordings following ketamine induction are characterized by the appearance of fast β activity (30 to 40 Hz) followed by moderate-voltage θ activity, mixed with high-voltage δ waves recurring at 3 to 4 second intervals. At higher dosages, ketamine produces a unique EEG burst-suppression pattern (Fig. 18-5).
FIGURE 18-5. Progressive changes in the electroencephalogram (EEG) produced by ketamine. Stages I through III are achieved with racemic ketamine and its S(+)isomer. With R(–)ketamine, Stage II was the maximal EEG depression produced. (Reprinted with permission from: Shüttler J, Stanski DR, White PF, et al. Pharmacodynamic modeling of the EEG effect of ketamine and its enantiomers in man. J Pharmacokinet Biopharm. 1987;15:241.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






