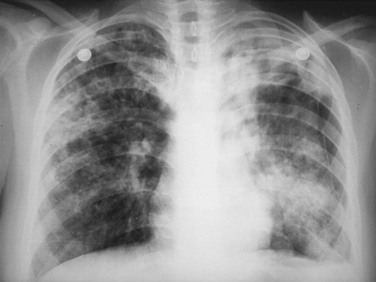
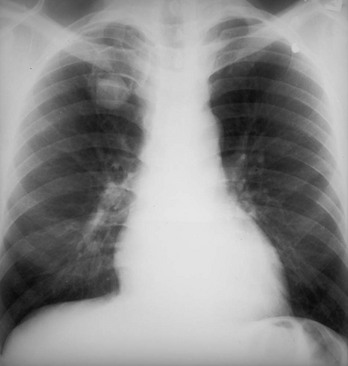
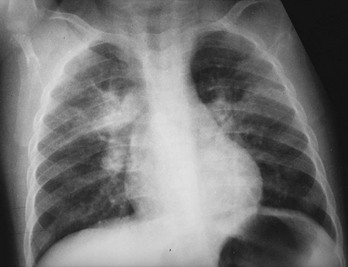
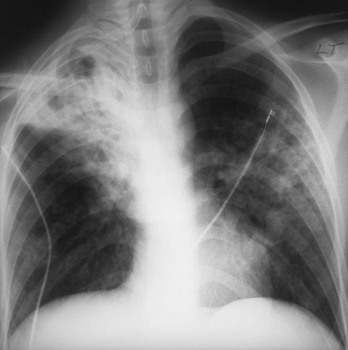
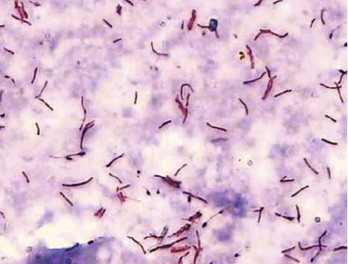
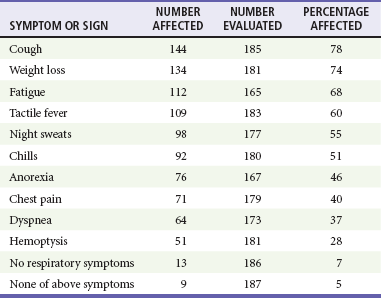
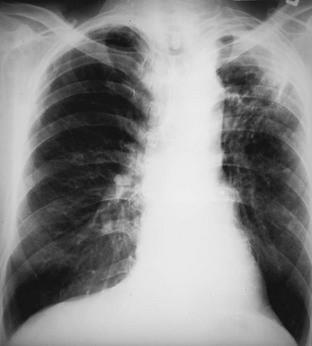
Chapter 135 Tuberculosis (TB) has plagued humankind throughout recorded history. Hippocrates (460-370 BCE) is credited for providing the first accurate clinical description of TB. He coined the term phthisis (“a melting or wasting away”) to describe the wasting character of the disease, which he noted also was associated with fever and incurable lung ulcerations. Overcrowded living conditions, widespread poverty, and poor hygienic conditions were ideal for the epidemic spread of the disease throughout western Europe from the early 1600s through the 1800s. TB gradually became a global epidemic as Europeans colonized North America and explored and colonized other parts of the world.1 The TB epidemic peaked in western Europe in the early 1800s and by 1900 had peaked in the Americas. Globally, the disease still has not reached a peak in some developing countries in Africa and Asia. Laënnec and Schönlein recognized the tubercle as the fundamental anatomic lesion and named the disease tuberculosis. Koch identified the tubercle bacillus in 1882, and Roentgen’s discovery of x-rays in 1895 greatly improved the ability to diagnose TB early in the disease course.2 In 1892 Biggs instituted a comprehensive program of TB control in New York City that served as a model for the U.S. Public Health Service, including public education, systemic surveillance, isolation of patients, nursing follow-up, improved sanitation, and free sputum testing. These types of programs and the subsequent introduction of the antituberculosis drugs led to an impressive decline in the incidence of TB throughout the 20th century. Between 1953 and 1985, TB cases decreased by an average of 5.8% per year. The decline of the infrastructure dedicated to TB control during the late 1980s and early 1990s was compounded by new problems, including the epidemic of human immunodeficiency virus infection and acquired immunodeficiency syndrome (HIV/AIDS), increasing immigration from countries with high TB prevalence, increasing occurrence of TB in institutional living settings, escalating poverty, substance abuse, homelessness, and urban overcrowding. These factors contributed to a resurgence of TB in the United States from 1986 through 2010 and to the emergence of drug-resistant strains of Mycobacterium tuberculosis (MTB). TB is currently the world’s second leading infectious cause of death, and one third of the world’s population has been infected by TB.3 Each year more than 8 million people acquire active TB infection, and nearly 2 million die of the disease.4 Elderly persons who harbor dormant infection that is reactivated constitute an important reservoir of MTB in the United States. Debilitating disease or immunosuppression may predispose the affected person to reactivation. Nursing homes are particularly vulnerable to TB outbreaks because reactivation of disease in remotely infected persons may be followed by epidemic spread among the many susceptible hosts living in close quarters. Emigration from endemic countries is another major factor.1,3 As recently reported, a majority of U.S. TB cases occur among foreign-born persons.5 The largest numbers of people with TB originate from Africa and Asia, specifically the Philippines, Vietnam, India, and China, accounting for more than half of all TB cases among foreign-born persons.6 Homelessness also has contributed to the spread of TB in major urban centers. Homelessness often is associated with conditions that decrease resistance to TB, such as malnutrition, alcoholism, and substance abuse. MTB infection in the homeless population may quickly progress to active TB infection. The HIV/AIDS epidemic has had the greatest impact on the reemergence of TB in the United States. The pandemic of HIV-related TB has led to an increase in TB cases among non–HIV-infected people owing to the higher numbers of source cases in the community. The rate of TB among patients who are HIV infected and have a positive result of TB skin testing is approximately 200 to 800 times higher than that estimated for the U.S. population overall.7 The reemergence of TB also has affected children. Between 1962 and the mid-1980s, the rates of childhood TB in the United States decreased an average of 6% a year. This trend has reversed along with the young urban adult trend, and the number of cases reported in children 4 years of age or younger increased 36% from 1985 through 1992. This increase reflected ongoing transmission of TB in the community because TB in young children must result from recent infection. In 2010, the Centers for Disease Control and Prevention (CDC) reported 11,181 new cases of TB in the United States, a rate of 3.6 cases per 100,000 population. More than half of these cases were in foreign-born persons.7 Multidrug-resistant TB was reported in 113 cases, and only one case of extensively drug-resistant TB was reported. In contrast, 9 million persons are newly diagnosed with TB each year throughout the world. Approximately 2 million persons die each year in countries outside the United States.8 TB is transmitted with rare exception by the respiratory route, including both droplet spread and as true aerosolization into microparticles. Patients with active disease expel MTB in liquid droplets during coughing, sneezing, and vocalizing. A single cough or 5 minutes of talking can produce 3000 infectious droplets, and sneezing can produce an even higher number.1 The droplets rapidly evaporate, and the desiccated bacilli circulate airborne for prolonged periods. These infective particles, or droplet nuclei, measure 1 to 5 µm in diameter, contain one to three tubercle bacilli, and, when inhaled, can travel to the distal alveoli. Transmission by nonrespiratory routes, such as direct inoculation, occurs primarily among health care workers. The risk for TB transmission increases when source patients have airway and cavitary disease. Infectivity correlates with the number of organisms seen on sputum smear, the extent of pulmonary disease, and the frequency of cough. After 2 weeks of chemotherapy, patients with initially AFB-negative sputum smears can be considered noncontagious. However, after 2 weeks of treatment, patients who initially were smear positive may still have viable MTB detectable in their post-treatment sputum cultures, and patients with extensive disease may still have AFB detectable on their post-treatment sputum smears; these two groups should be considered contagious. There is currently no clear epidemiologic evidence to better define contagiousness of patients after they have started effective therapy.9 The CDC has published guidelines requiring presence of three negative smears on different days as the criterion for removal of a patient from respiratory isolation, but debate about this recommendation is ongoing.9 Extrapulmonary TB also may be infectious, but only if it is in the oral cavity or an open skin lesion.9 Transmission of MTB to health care workers caring for patients with skin ulcers and draining tuberculous abscesses has been reported.10 Irrigation of the abscess may aerosolize the bacilli, forming infectious droplet nuclei. When infectious droplet nuclei are inhaled, the airflow through the bronchial tree tends to deposit them in the midlung zone on the respiratory surface of the alveoli. The deposition launches a complex series of immunologic events. Dannenberg has organized the complex pathogenesis of TB into four stages.11 When the alveolar macrophage is unable to destroy the inhaled tubercle bacilli, the bacilli replicate until the macrophage lyses. Circulating monocytes are attracted to the site of infection by the released bacilli, cellular debris, and various chemotactic factors. The monocytes differentiate into macrophages and ingest the free bacilli. Initially, these new macrophages are not activated and cannot destroy or inhibit the mycobacteria. The bacilli multiply logarithmically within macrophages and accumulate at the primary focus of infection, now called a tubercle.12 The infected macrophages also may be transported through lymphatics to regional lymph nodes, from which they can reach the bloodstream, with subsequent spread. During this lymphohematogenous dissemination, the pathogens tend to distribute preferentially to lymph nodes, kidney, epiphyses of long bones, vertebral bodies, meningeal areas, and apical posterior areas of the lungs. These sites may be favored because of a high oxygen tension or impaired clearance mechanisms from poor lymph flow. The third stage of TB begins 2 to 3 weeks after the initial infection, with development of the immune response that terminates the unimpeded growth of MTB. Cell-mediated immunity occurs through CD4+ helper T cells. When the T cell encounters mycobacterial antigens, it is activated and produces an expanded population of specific T cells. These T cells secrete cytokines (e.g., interferon-γ [IFN-γ], tumor necrosis factor) that attract and activate monocyte-macrophages. Once activated, the macrophages, containing previously ingested mycobacteria and their progeny, kill the bacilli. The destruction of the mycobacteria is associated with the formation of epithelioid cell granulomas and clearance of the organisms.12 Delayed-type hypersensitivity is mediated by cytotoxic CD8+ suppressor T cells. The cytotoxic cells kill nonactivated macrophages laden with mycobacteria and thus cause local tissue destruction. Delayed-type hypersensitivity results in the formation of caseating necrotic granulomas. This stops bacillary growth; mycobacteria, now extracellular, cannot multiply in this acidic, anoxic, extracellular environment. Tubercle bacilli can survive dormant in this solid caseous material for years.12 The host’s resistance determines whether the disease remains dormant or becomes active. The less-resistant host with weak cell-mediated immunity relies more on delayed-type hypersensitivity to control the infection. The primary lesion is surrounded by nonactivated macrophages so it is not effectively walled off, and the caseous center expands, compromising more lung tissue. If the infection eventually can be contained by the host, any initial manifestations may go unnoticed, and the infection may be evident only radiographically as healed parenchymal calcifications of the primary or Ghon focus and of the regional lymph nodes. If host defenses are unable to contain the primary infection, however, as can occur in infants and immunosuppressed adults, the primary focus may become an area of advancing pneumonia. This process is called primary progressive TB. In addition, this host may be unable to control the infection at the sites of lymphohematogenous spread, resulting in formation of multiple uncontrolled caseous tubercles and development of disseminated TB.12 HIV-infected patients are particularly susceptible to primary progressive TB because HIV specifically targets CD4+ cells and macrophages. The liquefied tubercle serves as an excellent growth medium for the mycobacteria. The large numbers of extracellular bacilli stimulate delayed-type hypersensitivity, which secondarily causes local damage. The tubercle eventually erodes through the bronchial wall and drains its contents, forming a cavity. The liquefied caseous material, teeming with mycobacteria, enters other parts of the lung and the outside environment. The spilling of this liquefied material within the lung may produce a caseous bronchopneumonia.12 The cavity formed at the site of the initial focus remains a significant lesion. Cavities provide optimal conditions for mycobacterial growth. The oxygen tension is increased, and the host’s defenses are ineffective at interrupting multiplication of the mycobacteria within a cavity.12 Emergency Department Presentation Patients with TB may present to the emergency department (ED) with either a primary infection or, more commonly, reactivation of an old infection. In either event, a wide variety of symptoms are features of this disease. Common ED presentations include classic infection in previously undiagnosed TB, TB with atypical symptoms, and known TB history with incomplete or complete drug therapy. The ED physician sometimes may be misled and not consider TB in the presentation of ill patients. Therefore, a high index of suspicion should be maintained and TB included in the differential diagnosis of common presenting complaints, such as isolated fever, weakness, weight loss, failure to thrive, and night sweats.13 The initial infection with TB most often is asymptomatic in otherwise healthy persons. Mild fever and malaise may develop in association with the immune response at 4 to 6 weeks, but the primary infection is generally insignificant clinically.14 Conversion to a positive PPD skin test result may be the only means of diagnosis of the infection. Clinically active TB develops in 8 to 10% of otherwise healthy PPD converters who do not take prophylactic agents: 3 to 5% in the first 2 years (acute primary TB) and another 5% during the remainder of life (reactivation TB). By contrast, in persons also infected with HIV, progression to acute primary TB occurs at a rate of 37% within 6 months and then to active TB at a rate of 7 to 10% per year.5 Reactivation of dormant foci is responsible for the major clinical manifestations of TB.14,15 Exogenous reinfection of patients with well-documented previous TB infection causes clinical disease indistinguishable from reactivation TB.15 Because it may be incorrect to label all late-onset cases as reactivation disease, postprimary TB is the preferred term. Postprimary TB is active or chronic disease in a patient previously infected. In the United States and other developed countries, reactivation is thought to be the primary mechanism of postprimary TB. A high index of suspicion should be maintained for elders.8,16 Exogenous reinfection has played a role in circumstances in which contagion levels are high, as in outbreaks of TB in developing countries, and in immunocompromised hosts.15,16 Clinically significant pulmonary TB often is indolent, and signs and symptoms are absent or minimal until the disease advances. Systemic reaction, thought to be mediated by cytokines, especially tumor necrosis factor alpha, causes the constitutional symptoms of anorexia, weight loss, fatigue, irritability, malaise, weakness, headache, chills, and, most commonly, fever.14,17 The fever usually develops in the afternoon; defervescence occurs during sleep, leading to the classic night sweats of TB.14 Cough is the most common symptom of pulmonary TB. It may initially be nonproductive, but as caseation necrosis and liquefaction develop, mucopurulent and nonspecific sputum typically is produced.14,17 Hemoptysis, caused by caseous sloughing or endobronchial erosion, usually is minor but often indicates extensive lung involvement. Many asymptomatic patients present for medical attention because they are alarmed by the hemoptysis. Patients also may complain of pleuritic chest pain, which is caused by parenchymal inflammation adjacent to the pleural surface. Dyspnea with chest pain may indicate a spontaneous pneumothorax. Shortness of breath from parenchymal lung involvement is unusual, however, and if present indicates extensive parenchymal disease or tracheobronchial obstruction.14 Table 135-1 shows the frequency of symptoms found in one study of patients with culture-proven pulmonary TB. The clinical manifestation of TB in patients presenting to the ED may be especially confusing. In one study, only one third of ED patients with active pulmonary TB had pulmonary chief complaints, only 64% ever reported a cough, and only 8% had hemoptysis.18 Table 135-1 Frequency of Symptoms and Signs in Pulmonary Tuberculosis From Barnes PF, et al: Chest roentgenogram in pulmonary tuberculosis: New data on an old test. Chest 94:316, 1988. Any vague systemic disorder or fever of unknown etiology may represent TB. Atypical presentations are particularly common in infants, elders, and immunocompromised persons. In infants and young children, the development of large hilar lymph nodes is common; such nodes may compress a bronchus, leading to atelectasis and possibly obstructive pneumonia; the child may have a “brassy cough.” A node also may erode through the bronchial wall, causing symptomatic endobronchial disease and allowing endobronchial spread of tuberculous pneumonia to other areas of the lungs. By contrast, fewer elders present with respiratory symptoms. The diagnosis may be masked by coexistent disease and nonspecific presenting symptoms.19 Pulmonary TB should be considered in elderly patients with chronic cough and failure to thrive.5 Clinical manifestations of TB in patients coinfected with HIV are even more subtle and nonspecific, especially because these patients are vulnerable to opportunistic infections and neoplasms that can cause the same constitutional symptoms as in TB. A synergy between MTB and HIV leads to a greatly increased viral load. Active TB with HIV coinfection has been associated with an increased risk for opportunistic infections and death. Patients with advanced HIV infection also commonly have extrapulmonary involvement (seen in 30%) as well as combined pulmonary and extrapulmonary TB (in 32%).17 All patients in the ED who have been coughing should be screened for the presence of TB risk factors (Box 135-1). One of the most important risk factors is HIV/AIDS with CD4+ levels below 500 cells/µL.20 Overseas, coinfection with HIV and TB is common and results in increased mortality of TB. Risks for acquiring TB may also be stratified by age. Because infants and toddlers have poorly developed cell-mediated immunity, they have a much higher incidence of TB than adults do. Children 5 to 10 years of age are relatively resistant to TB. Infants and toddlers commonly have extrapulmonary disease and acute lower and midlung bronchopneumonia that rarely progresses to cavitary disease. Young adults show the adult pattern of apical pulmonary disease, including cavity formation, suggesting reactivation. Because of decreased immunocompetence, elders typically have disease manifestations similar to those in young children. Patients with a history of PPD conversion should be asked about the presence of medical conditions associated with increased risk for the development of active postprimary disease through reactivation (Box 135-2). Household contacts also have increased risk of TB infection.21 Patients with a history of active TB should be asked about all antituberculosis medications previously or currently being taken and about compliance. Failure to improve after 2 months with an appropriate regimen may signal nonadherence to therapy or the presence of a resistant strain.22 Examination of the chest is unlikely to establish the extent of disease. Over areas of infiltration, rales may be heard when the patient breathes in after a short cough (post-tussive rales), and bronchial breath sounds may be present over areas of lung consolidation. Distant, hollow breath sounds (amphoric breath sounds) may be heard over cavities. One ED study reported abnormal physical examination findings in 80% of patients with active pulmonary TB.18 Spontaneous pneumothorax is not common (affecting less than 5% of patients with severe cavitary disease) but may occur when a tuberculous cavity ruptures and creates a bronchopleural fistula or when a bleb ruptures into the pleural space (Fig. 135-1). Delayed tube thoracostomy and suction result in progressive infection and fibrosis of the pleura that leads to air trapping within affected lung. Extensive TB infection often heals with open cavities and areas of bronchiectasis. Superinfection may occur with a wide variety of organisms, including Aspergillus fumigatus.14 The characteristic finding on chest radiographs is the aspergilloma or “fungus ball” (Fig. 135-2). Aspergillomas are of particular clinical significance because they may cause massive and fatal hemoptysis.14 Mild hemoptysis is a common complication of acute infection. TB also causes massive hemoptysis. The destruction of lung parenchyma leads to rupture of blood vessels. An uncommon complication is the erosion of a tuberculous lesion or cavity into a pulmonary artery, leading to pseudoaneurysm formation (Rasmussen’s aneurysm), with potentially fatal hemoptysis. Alternatively, superinfection of cavities by invasive organisms or tumor development in scarred lung may cause erosion of bronchial or pulmonary vessels, with resultant major hemorrhage. Affected patients often require emergency surgical resection or selective embolization.23 Primary tuberculous pericarditis usually results from direct extension of infection from the tracheobronchial tree, mediastinal or hilar lymph nodes, sternum, or spine. Pericardial involvement also may result from hematogenous spread secondary to acute miliary TB or from another focus elsewhere in the body.24 In the United States, TB is the leading cause of pericarditis among HIV-infected patients. The predominant symptoms are cough, chest pain, and dyspnea, and the most common signs are cardiomegaly, audible rub, fever, and tachycardia.25 Complications of pericardial TB include pericardial effusion, constrictive pericarditis, myocarditis, and cardiac tamponade.24 Cardiac tamponade may result from accumulation of pericardial fluid but also may occur if enlarging lymph nodes rupture into the pericardium. Emergency echocardiography reliably confirms the presence of pericardial fluid.26 Routine laboratory studies generally are not useful in suggesting or establishing the diagnosis in the ED.14 Normochromic normocytic anemia, elevated erythrocyte sedimentation rate and serum globulin level, hyponatremia, and hypercalcemia can occur in active pulmonary TB, but these findings are nonspecific.14,27 Tuberculin skin testing is important epidemiologically as a modality for diagnosis of MTB infection but is of limited value for detection of active clinical disease. At least 20% of patients with active TB have false-negative results on tuberculin skin testing. Chest radiographic manifestations of primary disease in adults often are not recognized as TB.28 Primary tuberculous infiltrates can occur in any lobe. With any age group, a pneumonic infiltrate with enlarged hilar or mediastinal nodes should strongly suggest the diagnosis. The infiltrate usually is homogeneous and most commonly involves a single lobe. Thus primary TB may appear radiographically identical to a bacterial pneumonia, with associated lymphadenopathy, if it is present, being the only distinguishing feature. Lymphadenopathy is considered the radiologic hallmark of primary TB in children but is seen less commonly in adults. When it is present, adenopathy usually is unilateral and associated with parenchymal infiltrate (Fig. 135-3). It may occur bilaterally or, more rarely, may be an isolated finding on chest radiography.14 Massive hilar adenopathy is more common in young children. As a result, atelectasis, resulting from airway compression by adjacent enlarged nodes, is a likely finding in children younger than 2 years but is less common among older children and adults. Other primary TB chest radiographic findings include a moderate to large pleural effusion, which often is an isolated finding whose prevalence increases with age; miliary TB (characterized by the presence of innumerable, 1- to 3-mm noncalcified nodules dispersed throughout both lungs with mild basilar predominance), which is mainly a threat to children younger than 2 years, immunocompromised patients, and elders; and tuberculomas, well-circumscribed nodular lesions of the parenchyma thought to be a result of healed primary TB.29 When the healed primary focus is visible on the chest radiograph as a calcified scar, it is known as the Ghon focus. Calcified secondary foci of infection are known as Simon’s foci. A Ghon focus associated with calcified hilar nodes is called a Ranke complex. A right-sided predominance in the distribution of Ghon foci and Ranke complexes is well recognized and probably reflects the higher statistical probability that an airborne infection will affect the right lung. Calcification seen on the chest radiograph indicates healing, but viable bacilli may still exist in a partially calcified lesion. Postprimary TB typically appears as an upper lung infiltrate or consolidation, with or without cavitation. The lesion may be small or extensive and usually is located in the apical or posterior segment of the upper lobe but may appear in the superior segment of the lower lobe.29 Postprimary disease also occurs in the lower lung. In addition, bronchogenic spread can occur, leading to involvement of multiple lobes (Fig. 135-4). The patient with bilateral upper lobe disease is extremely likely to have TB. The other important, recognizable characteristics of postprimary disease are fibrosis and cavitation. Cavitation should alert ED personnel to the potential high infectivity of the patient and the potential for associated complications, such as bronchogenic spread of TB, when an area of caseous necrosis liquefies, with consequent communication with the bronchial tree (see Fig. 135-4). Cavities usually are multiple and range in diameter from a few millimeters to several centimeters. The walls of the cavities initially are thick and rough and become thinner and smoother with healing (Fig. 135-5). A hazy, parenchymal reaction around a cavity with an ill-defined wall strongly suggests an active lesion. Most cavities heal by obliteration, often leaving a small linear or stellate scar; others remain patent and become thin-walled bullae. Although pulmonary TB usually induces chest radiographic changes, patients with sputum cultures positive for MTB may nevertheless demonstrate normal radiographic findings. Chest radiographs also may be normal in appearance in patients with endobronchial TB. Figure 135-5 A healed, thin-walled cavity can be seen in the left upper lobe on this chest film. (Courtesy John Pearce, MD.) Chest radiographs of patients with pulmonary TB and HIV infection may be atypical in approximately one third of cases. However, the radiographic appearance of disease is heavily influenced by the degree of immunocompromise. Patients with late HIV infection more often demonstrate mediastinal adenopathy or atypical infiltrates and less often have cavitation.30 Severe immunosuppression has been reported to be associated with a miliary pattern of disease on chest radiographs. Conversely, chest radiographs of patients with early HIV infection are more similar to those of patients without HIV infection, with upper lobe infiltrates, more cavitation, and less adenopathy. A normal chest radiographic appearance also is common in patients with HIV infection. If the clinical or chest radiographic findings suggest the diagnosis of pulmonary TB, mycobacteriologic studies of the patient’s sputum should be ordered. Spontaneously produced sputum collected under direct supervision is preferred, and early-morning samples are the best diagnostic specimens. Classically, three initial sputum specimens should be obtained on different days. A positive smear supports a presumptive diagnosis, and the number of bacilli seen correlates with infectivity. For patients who are not producing sputum, nebulized induction of sputum and gastric aspiration of swallowed respiratory secretions are the methods of choice for collection of samples.31 The diagnostic yield is higher for nebulizer-induced sputum samples than for gastric aspirates in adult patients with TB, but in some patients, especially children, gastric aspirates may be the only obtainable specimens. Induction of sputum with nebulization may increase the risk of TB transmission to health care workers and should be performed only in specially ventilated rooms, preferably not in the ED. When sputum is not diagnostic in adults, fiberoptic bronchoscopy with bronchial washings, brushings, and bronchoalveolar lavage or transbronchial biopsy may be necessary for laboratory diagnosis of TB.31 In children, sputum obtained by bronchoscopy has a lower culture yield, so this technique is used less often to obtain sputum from pediatric patients.32 Direct microscopic examination of a stained sputum specimen for AFB (i.e., an AFB smear) is the most rapid laboratory test widely available to support a presumptive diagnosis of TB (Fig. 135-6), and results usually are available within 24 hours.33 Although nontuberculous mycobacteria can cause pulmonary disease, they are less common than MTB and vary by geographic location and population of patients. Fluorochrome stains are more sensitive than the traditional Ziehl-Neelsen or Kinyoun methods for the detection of AFB from clinical specimens.32,33 Negative findings on an AFB smear, however, do not rule out active pulmonary TB because microscopy is relatively insensitive when it is performed on samples with small numbers of bacilli. At least 5000 bacilli/mL of sputum must be present for a positive result by microscopy.33 Because cavitary disease is associated with great numbers of bacilli, the diagnostic yield of microscopy increases with cavitation. Concentrated smears are prepared by decontamination, liquefaction, and centrifugation of sputum, and such smears may be more sensitive than unconcentrated samples.4 Overall, AFB smears have a sensitivity of 20 to 80% and a specificity of 90 to 100%.33 Despite its limitations, microscopy remains an essential diagnostic test because of its ease of performance, low cost, rapid turnaround time, and reasonable diagnostic yield. Sputum culture is more sensitive than microscopy for detection of MTB and is still considered the “gold standard” diagnostic modality. Liquid culture can detect 10 to 100 bacilli/mL, compared with 5000 to 10,000 bacilli/mL for AFB smear. When the presence of mycobacteria is established, the specific identification of MTB may be accomplished by subjecting the initial mycobacteria to various isolation techniques. These include the detection of pigmentation on solid culture media, various biochemical tests, high-performance liquid chromatography, and nucleic acid probes.33 A presumptive diagnosis of TB based on a positive sputum smear usually is confirmed by isolation of MTB by culture. Traditional culture methods using solid media require 3 to 8 weeks for colony formation. The development of liquid culture systems has shortened the detection time to 7 to 14 days. A number of liquid culture systems are available for detection of MTB. For example, the BACTEC (Becton Dickinson, Franklin Lakes, N.J.) method measures carbon dioxide produced by growing mycobacteria when they metabolize the 14C-labeled palmitic acid contained in the system,14 and the mycobacteria growth indicator tube (MGIT) method measures mycobacterial oxygen consumption with a fluorescence assay.33 Because of the emergence of multidrug-resistant MTB, all initial isolates of MTB should be tested for susceptibility to isoniazid, rifampin, and ethambutol.34 Further susceptibility testing should be performed when resistance to one of the three agents is detected or if the patient has had previous TB treatment, has been exposed to a drug-resistant contact or source, or has demonstrated a positive result on sputum cultures beyond 3 months of therapy.34 Conventional susceptibility testing detects selective growth of drug-resistant TB on media containing antituberculosis drugs. For first-line agents, this requires waiting until 4 to 7 days after a positive culture result is obtained; testing for susceptibility to other drugs may require 2 to 3 months. The BACTEC and MGIT systems also can be used for rapid drug susceptibility testing, with results obtained in fewer than 12 days.33 Other new approaches to susceptibility testing include flow cytometry, genotypic sequence-based methods, and bacteriophage viability testing.33,35 The CDC has recently published guidelines for use of IFN-γ release assays and nucleic acid amplification (NAA) tests for the diagnosis of TB.36,37 The QuantiFERON test is the most widely available IFN-γ release assay available. This test was approved in 2001 by the U.S. Food and Drug Administration (FDA). It uses an enzyme-linked immunosorbent assay (ELISA) to measure the amount of IFN-γ released in response to PPD. IFN-γ is a cytokine associated with cell-mediated immunity. Determination of IFN-γ levels also can be used as a diagnostic test for tuberculous pleural effusions, ascites, and pericardial effusions. Clinical studies have reported sensitivity ranges from 90 to 100%. It can rapidly confirm TB infection in an individual in 2 days. This compares with several weeks for a traditional culture.36 However, a normal study does not completely exclude TB; therefore, cultures should be sent when persons thought to have TB have a negative QuantiFERON test result.
Tuberculosis
Perspective
Principles of Disease
Pathogenesis
Stage 2
Stage 3
Stage 4
Clinical Features
Patient’s History
Risk Factors
Physical Findings
Complications
Superinfection
Hemoptysis
Primary Tuberculous Pericarditis
Diagnostic Strategies
Diagnostic Imaging
Primary Tuberculosis
Postprimary Tuberculosis
Microbiology
Direct Microscopy
Culture
Drug Susceptibility Testing
Newer Diagnostic Studies
QuantiFERON Test
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Tuberculosis
Only gold members can continue reading. Log In or Register to continue