Chapter 16 Chest trauma is a frequent etiology for admission of patients to critical care units. In North America, blunt thoracic injury is responsible for about 8% of all trauma admissions and chest trauma (blunt and penetrating) account for one in four of all deaths from trauma, and is a contributing factor in another 25%.1, 2 Early deaths result from hemorrhage, or associated head, or abdominal trauma. Many of these patients die after reaching the hospital, and of those deaths, many might have been prevented with prompt diagnosis and treatment. The vast majority of thoracic injuries (90% blunt and 70–85% penetrating) can be managed without open surgical thoracotomy.3 As such, the skills for dealing with thoracic trauma are well within the scope of intensivists who care for victims of trauma. All practitioners must be alert to the myriad of possible injuries that can occur in the chest, and facile in their treatment as recognition and treatment can be life-saving. The intensivist caring for patients with thoracic trauma should be skilled in establishing a patent airway, providing adequate ventilation and oxygenation, evacuation of fluid or air from the chest, diagnosing and temporizing cardiac tamponade (pericardiocentesis), and providing vascular access for fluid administration. Recognition of when to seek surgical consultation/intervention in these patients is also an important principle for the critical care practitioner. Generally, thoracic trauma may be classified into two broad groups: penetrating and blunt. It is important to remember that trauma to the chest is often not an isolated event and other structures can also sustain potentially life-threatening injuries as well.4, 5 A penetrating missile entering inferior to the nipple line can produce diaphragmatic, intrathoracic, or abdominal injuries. Similarly, blunt injuries may disrupt intrathoracic contents as well as intraabdominal contents either directly or indirectly through fractures of the lower ribs, which then puncture intra-abdominal organs such as the spleen, liver, and stomach. It is imperative when caring for these patients not to get “tunnel vision” and to rule in or out other potential life-threatening injuries outside of the chest. A more practical (clinically relevant) method of classifying torso trauma involves two categories. The first category consists of injuries that are immediately life threatening and thus require immediate intervention because of cardiorespiratory or hemodynamic compromise. The other category includes injuries in a relatively hemodynamically normal patient that have the potential for adverse outcome if not promptly recognized and treated. As seen in Table 1, these two categories can be collectively referred to as the “Deadly Dozen” injuries of the chest which are further divided into the “Fearsome Fatal Five” and the “Serious Seven”. The critical care practitioner caring for these patients must have a broad understanding of the diagnosis and treatment of all of these potential injuries. While one would hope that the recognition and initial treatment of the “Fearsome Fatal Five” would have occurred prior to a patient being admitted to the intensive care unit, the intensivist may be the one to make the diagnosis and in a position to render initial treatment, and will most certainly be involved in managing the aftermath of these conditions and their treatment. Additionally the intensivist may play a very important role in helping to identify and treat the “Serious Seven” injuries which may not be immediately apparent, and have the potential to be potentially life threatening if left unattended. It must also be kept in mind that some of these life-threatening thoracic injuries may result from complications of procedures done in the intensive care unit (ICU), e.g., simple or tension pneumothorax or hemothorax from central line insertion; rib fractures from chest compressions; cardiac tamponade from attempted pericardiocentesis; etc. The next sections will provide a detailed description of the multitude of injuries that can occur in the chest, the diagnostic adjuncts and the specific treatments and critical care considerations following treatment. In all cases it is imperative that the intensivist ensure a patent airway and adequate intravenous access. Table 1. The “Deadly Dozen” injuries of the chest.
Thoracic Trauma
Col. (retd.) Mark W. Bowyer and Jameel Ali*
Introduction
Classification of Thoracic Trauma
The Fearsome fatal five | The serious seven |
1. Complex Pneumothorax: | 1. Pulmonary Contusion |
a. Tension Pneumothorax | 2. Blunt Cardiac Injury |
b. Open Pneumothorax | 3. Traumatic Aortic Disruption |
c. Massive Pneumothorax | 4. Esophageal Injury |
2. Cardiac Tamponade | 5. Traumatic Diaphragmatic Injury |
3. Massive Hemothorax | 6. Rib Fractures |
4. Traumatic Air Embolism | 7. Simple Hemo or Pneumothorax |
5. Flail Chest |
Thoracic Injuries Requiring Immediate Intervention (The “Fearsome Fatal Five”)
The initial treatment of all trauma patients is to ensure an adequate airway and intravenous access. In addition to upper airway obstruction, the following injuries require immediate recognition and intervention:
(1) Complex Pneumothorax:
(a) Tension Pneumothorax
(b) Open Pneumothorax
(c) Massive Pneumothorax/Broncho-pulmonary Disruption
(2) Cardiac Tamponade
(3) Massive Hemothorax
(4) Traumatic Air Embolism
(5) Flail Chest
Tension pneumothorax (one of the fearsome five)
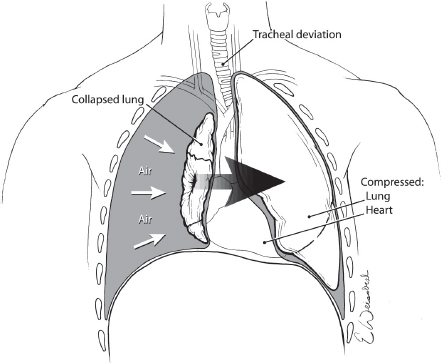
Tension pneumothorax occurs when there is an injury to the lung with resultant air leakage into the chest cavity that cannot escape. This “one-way valve” phenomenon results in each breath increasing both the volume of air and the intrapleural pressure. As the pressure builds, structures in the chest are compressed and displaced with impairment of ventilation and venous return to the heart (see Fig. 1).
The resultant combination of hypoxemia and hypoperfusion can be rapidly fatal necessitating prompt recognition and immediate treatment. Tension pneumothorax is a common cause of preventable death in the pre-hospital environment. The diagnosis of a tension pneumothorax is a clinical one characterized by tachypnea, dyspnea, jugular venous distention, decreased air entry, hyperresonance on the affected side and tracheal deviation to the opposite side in a patient with hypotension. Tension pneumothorax should be considered in all patients with trauma to the thorax and ruled out in the initial assessment of the patient. It is important to note that patients placed on positive pressure ventilation after reaching the ICU can rapidly develop a tension pneumothorax, if they had an unrecognized or untreated occult or simple pneumothorax. Waiting to make or confirm the diagnosis with an X-ray is to be discouraged as the hemodynamic compromise associated with this process is rapidly fatal and delaying treatment for this reason would be a gross breach of standards of care. Additionally, distended neck veins may not be appreciated if the patient has concomitant hypovolemic shock. Likewise, tracheal deviation associated with tension pneumothorax may occur inferior to the jugular notch and may not be obvious, so its absence does not rule out this condition.
Fig. 1. Tension pneumothorax results when air leaking into the chest from an injured lung cannot escape with resultant compression and displacement of mediastinal structures and death if untreated.
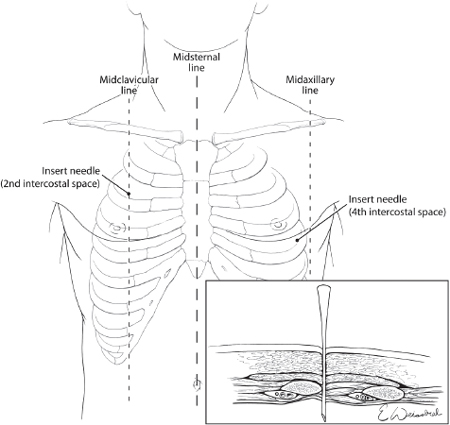
The treatment of tension pneumothorax is immediate decompression of the pleural space, which is accomplished initially by inserting a long large-bore needle into the pleural space at the second intercostal space in the line6 or alternatively the fourth to fifth intercostal space just anterior to the midaxillary line7 (see Fig. 2). The needle should be at least three inches in length in order to completely traverse the chest wall.6 This procedure should be followed by formal insertion of a chest tube.
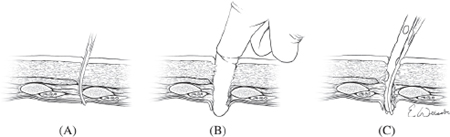
The ability to insert a chest tube is a mandatory skill for all clinicians working in the critical care arena, as surgical expertise is not always immediately available. The technique of chest tube insertion as diagramed in Fig. 3 require an incision of ~3 cm in the 4th to 5th intercostal space just anterior to the midaxillary line. If patient stability and time permits the area should be surgically prepped and draped, local anesthesia infiltrated, and the clinician appropriately gloved and gowned. The landmarks used for finding the site of the incision include the line of the nipple in males and the inframammary crease in females, though a potentially more reliable technique is to place the incision four fingerbreadths below the axillary crease (the width of a palm placed in the axilla). The incision is carried down onto the underlying rib and then a large blunt clamp (Kelley clamp) is popped into the chest above the rib though the intercostal muscle and pleura, enlarging the space by opening the clamp sequentially as it is removed. A gloved finger is then placed into the chest and rotated to first ensure that the chest cavity has been entered, and secondly to sweep away any potential adhesions of the lung up to the chest wall that might impede tube insertion. The chest tube is then inserted into the wound and pleural cavity aiming it towards the posterior apex of the chest cavity, such that the last hole on the chest tube is entirely within the chest (Fig. 3). The tube is then secured in place, dressed, and attached to an underwater seal device with application of suction (generally −20 cm water). The size of the chest tube should be geared to the problem being treated using larger bore chest tubes (36 French) if there is a need to drain blood from the chest, and smaller chest tubes if air is the primary focus. It should be kept in mind that blood drained from a chest tube can potentially be auto-transfused back to the patient and as such if one suspects a large hemothorax prior to placing a chest tube, an autotransfusion bag should be placed on the chest tube in circuit with the underwater seal.
Fig. 2. Emergent treatment of a tension pneumothorax requires needle decompression of the chest using a large bore needle placed over the top of the rib in the 2nd intercostal space at the midclavicular line or alternatively in the 4th intercostal space just anterior to the midaxillary line.
Fig. 3. Chest tube insertion is accomplished by puncturing the pleura of the lung with a clamp over the top of the rib (a) and spreading the clamp to enlarge the space sufficiently for a gloved finger to be passed into the chest (b) which is used to confirm entry into the chest and to sweep down any adhesions to the chest wall. A chest tube is then advanced into the chest (c) until the last hole of the chest tube is entirely within the chest cavity.
Open pneumothorax
The pathophysiology of open pneumothorax is similar to that of a tension pneumothorax with collapse of the lung and shift of the mediastinum resulting in hemodynamic and respiratory compromise. In the case of an open pneumothorax, which is also descriptively called a “sucking chest wound”, there is free communication through a chest wall wound between the pleural space and the atmosphere. With each breath, air is “sucked” into the chest cavity through the wound in proportion to the size of the wound, preventing negative intrapleural pressure with resultant lung collapse. The communication of the pleural space to the atmosphere through the chest defect prevents generation of the negative pressure required for lung expansion during spontaneous inspiration. The net result is progressive collapse of the lung with each subsequent respiratory cycle. The larger the defect in the chest wall, the more pronounced this phenomenon will be. The diagnosis usually is obvious, with a visible open wound in the chest wall and a characteristic loud (sucking) noise created from atmospheric air entry into the pleural space.
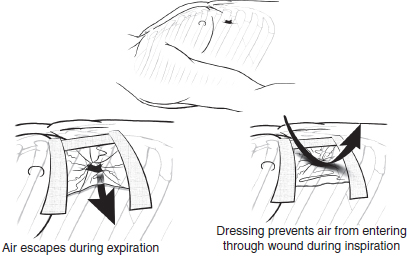
The emergent treatment of a sucking chest wound is to occlude the open wound. Classically this is accomplished by tapping an occlusive dressing down onto the chest on three sides which allows for the egress of air from the plural space without further ingress (Fig. 4). Alternatively, a commercial device such as an Asherman or Bolin seal8 can be placed over the open wound. After the wound has been occluded a chest tube should be placed through an incision separate from the wound as previously described. Wounds too large for simple occlusion will require endotracheal intubation and positive-pressure ventilation until formal surgical repair of the chest wall defect can be accomplished in the operating room (OR).
Fig. 4. The initial treatment of a “sucking” chest wound is to occlude the open wound by tapping an occlusive dressing down onto the chest on three sides which allows air to escape without further ingress.
Massive pneumothorax/tracheobronchial disruption
Massive pneumothorax that occurs as a result of tracheobronchial disruption is a serious life-threatening problem that is reported to occur in about 1% of motor vehicle collision victims of whom 80% die before reaching the hospital. The injury to the bronchus is most commonly found on the right side within 2.5 cm of the carina. The typical presentation of patients surviving to hospital is mediastinal, deep cervical, and subcutaneous emphysema with persistent air leak and pneumothorax in spite of a functioning chest tube with associated hypoxemia and respiratory instability.
Patients with this suspected injury should be immediately placed on 100% oxygen, and for optimal outcome, patients with this injury should be taken emergently to the OR for repair by an experienced surgeon with a Computed Tomography (CT) scan and/or on table bronchoscopy to help delineate the site of the injury if time and patient stability permit. If there is massive destruction of the bronchus with failure to achieve an anastomosis or a high risk of subsequent stenosis, lung resection should be considered. Surgery remains the treatment of choice, however in very selected circumstances, based on strict clinical and endoscopic criteria conservative non-operative management has been advocated.9
Cardiac tamponade
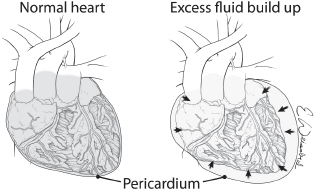
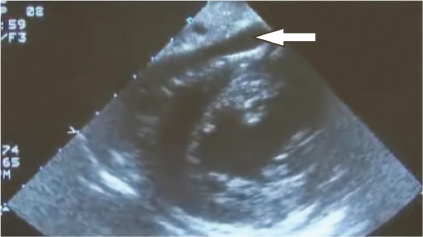
Cardiac or pericardial tamponade results from accumulation of fluid (blood) into the pericardial sac around the heart that interferes with cardiac filling and restricts cardiac activity (Fig. 5). The diagnosis of cardiac tamponade should be considered in any patient who has penetrating or blunt trauma to the chest. Etiology in trauma patients is predominantly from a penetrating wound but can also occur in blunt trauma from cardiac rupture. Blunt cardiac rupture is immediately fatal in greater than 90%, and survivors usually have atrial injuries. The classic physical findings of cardiac tamponade described by Beck (Becks triad) of hypotension, distended neck veins, and muffled heart sounds are not always present and may be difficult to discern. The prevalence of portable ultrasound technology has made tamponade easier to diagnose as visualization of the heart via the epigastric pericardial view or parasternal view is sensitive for small amounts of fluid in the pericardial sac (see Fig. 6). Once the diagnosis of tamponade is made the patient should be taken promptly to the OR for surgical release of tamponade and correction of the underlying pathology which can be done either through an anterolateral thoracotomy or a sternotomy. If surgical expertise is not immediately available consideration should be given to performing a pericardiocentesis as relatively small amounts of blood (100 cc) will cause tamponade and removal of small amounts (15–20 cc) may be enormously beneficial. It must be kept in mind that pericardiocentesis is a temporizing measure only and that definitive treatment requires surgical opening of the pericardium with repair of the underlying injury.10
Fig. 5. Pericardial tamponade results from the accumulation of fluid (blood) in the intact pericardial sac with resultant compromise in cardiac function.
Fig. 6. Ultrasound of the heart demonstrates accumulation of blood (arrow) between the pericardium and the heart confirming the diagnosis of tamponade.
Pericardiocentesis is performed by inserting a long (> 6 inches) large bore (16 to 18 gauge) needle (preferably an angiocath) into the pericardial sac and aspirating the fluid therein with a large empty syringe with a three-way stopcock. The needle is inserted just below the xiphoid process a roughly a 45° angle and advanced cephalad toward the tip of the left scapula or the left nipple (Fig. 7). Gentle aspiration on the syringe is maintained as the needle is advanced with the return of non-clotting blood confirming entry into the pericardial sac. Classically, attaching an alligator clamp to the hub of the needle and then to an electrocardiographic lead is described to detect a “pattern of injury” if the needle is inserted too far and hits the heart. In actual practice the immediate availability of such equipment and the urgent nature of the need to correct tamponade, limits application of this “recommended” adjunct.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree