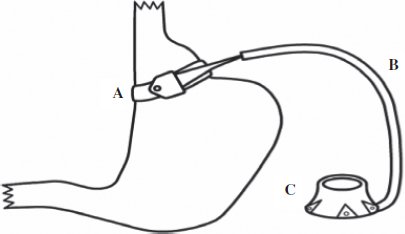
Chapter 32 In discussing management of the bariatric surgery patient two major sets of issues must be addressed: first, the morbid obesity state itself and secondly the bariatric surgical procedures and their effects. Much progress has been made in both these areas to make bariatric surgery relatively safe.1 Most bariatric surgery patients do not require care in the intensive care unit (ICU) but because of the unpredictable course in individual patients, availability of ICU facilities is essential for successful outcome in complicated cases. In this chapter we discuss; (1) general considerations in the morbidly obese which could impact outcome and ICU care and (2) bariatric surgical procedures themselves which carry their own risks. Understanding these concepts should promote better care for these patients if and when this care needs to be provided in the ICU. Obesity is an independent risk factor for heart disease, hypertension, diabetes mellitus, and obstructive sleep apnea,2 all of which could impact on the perioperative care of the bariatric surgery patient. To minimize the effects of these factors, optimize overall care and limit complications. The patient requires a multi-disciplined approach which is an integral part of the bariatric surgical program. Nutritionists, psychologists, dedicated anesthetists, internists (endocrinologists), pulmonologists, gastroenterologists are all part of the care team.3 Laboratory investigations includes complete blood count, coagulation and metabolic profile, thyroid function tests and assessment of vitamin levels such as vitamin B12, fat soluble vitamins, and ferritin levels with preoperative correction of identified deficits including glycemic control and preparation for care of the frequently associated diabetes as well as preparation for continued support in the perioperative period to improve outcome. Cardiorespiratory evaluation includes electrocradiography, stress test to identify occult coronary artery disease, chest X-ray, arterial blood gases, and pulmonary function tests. Sleep apnea is diagnosed by sleep study and where indicated preoperative continuous positive airway pressure (CPAP) is recommended. If gastric pathology is suspected, upper endoscopy is conducted and Helicobacter pylori (H. pylori) infection is treated, if identified. Liver function is assessed by laboratory tests as well as ultrasound and if cirrhosis is a possibility, liver biopsy may be indicated. If gall stones are detected the surgeon may discuss concomitant cholecystectomy as an option. Key elements in preventing perioperative complications and optimizing care of the bariatric surgery patient both in general and specifically in the ICU are anticipation of problems and being prepared to deal with these challenges. Airway control, maintenance of ventilation, cardiovascular, and fluid dynamics as well as pharmacodynamics are areas that require close attention for optimal care of these patients. Being prepared for difficult intubation is always wise in the obese patient and this requires having appropriate medications available as well as regular and more invasive devices including Gum Elastic Bougie, Laryngeal Mask Airway (LMA), fiberoscopic bronchoscope, and the option of a surgical airway. The large tongue, enlarged endopharyngeal mass, limitation of neck movement, and excessive presternal fat limit movement of the laryngoscope, making direct laryngoscopy extremely difficult for intubation. Careful assessment of these factors to determine ease of intubation is necessary, allowing a decision to proceed with general anesthesia induction and intubation or alternatively awake intubation. Fortunately, in most circumstances, general anesthesia induction followed by intubation is possible by using such techniques as the Head Elevated Laryngoscopy Position (HELP).4 In the awake intubation technique, topical anesthesia may be used in the cooperative patient with premedication followed by direct laryngoscopy and intubation, or more commonly by fiberoptic bronchoscopy. The LMA is a good back-up temporary device to allow ventilation until a more definitive airway technique is used. During prolonged attempts at intubation attention must be directed at maintenance of oxygenation by interruption of the intubation attempts to allow bag valve mask ventilation. In some circumstances the technique of apneic oxygenation5 may be helpful to maintain oxygenation through passive means by way of continuous oxygen delivery using an LMA, bronchoscope, or nasal cannula. The morbidly obese patient is generally considered to be at greater risk for aspiration of gastric contents because of increased abdominal pressure, increased incidence of gastro-esophageal reflux with or without hiatus hernia, and generally have a higher gastric residual after fasting. These factors together with the increased ventilator pressures and difficult mask ventilation predispose to regurgitation from gastric insufflation. Because of these factors many anesthetists use prokinetic agents, H2 receptor antagonists, or proton pump inhibitors to minimize the effect of aspirated contents. If rapid sequence intubation appears safe this technique could minimize the chance of aspiration by decreasing the induction time. Decreased chest wall compliance from the excessively fatty chest and abdominal wall tissue, decreased lung compliance from increased pulmonary blood flow, and viscosity all make ventilation difficult. The decreased functional residual capacity (FRC) which is worsened in the supine position may become lower than the closing volume (see chapter on Perioperative Respiratory Dysfunction) leading to airway closure and hypoxemia. These factors together with the increased oxygen demand from the increased metabolic needs of the obese patient lead to arterial desaturation not only in the operating room but also in the ICU. Based on the aforementioned considerations, techniques for optimizing gas exchange in the obese patient includes head up position, larger tidal volumes, positive end expiratory pressure (PEEP), and high inspired oxygen concentrations, recognizing the potential deleterious hemodynamic effects particularly on cardiac output of these maneuvers.6,7 As mentioned in the preceding paragraphs, many morbidly obese patients suffer from obstructive sleep apnea and these patients could desaturate particularly in the immediate post intubation period when residual anesthetic and analgesia decrease the respiratory drive leading to hypoventilation and adverse events including cardiac arrest. It is recommended that these patients bring their CPAP device to the hospital and that CPAP is available in the recovery room as a standby to deal with this possibility.8 As indicated earlier, morbid obesity is a high risk factor for coronary artery disease and its sequelae including myocardial ischemic events and cardiac dysrhythmia. Close cardiac monitoring and preoperative cardiac investigation, as well as preparedness to deal with these events in the perioperative setting including the ICU are essential elements in the care of the bariatric surgery patient. The obese patient develops cardiac dysfunction from a multitude of causes9 including chronic ventricular stress from the increased circulating blood volume and metabolic demands, and later pulmonary hypertension. Patients deemed at greater risk based on assessment are ideally managed in an ICU setting where close monitoring and timely intervention including pharmacologic support is immediately available. The obese patient metabolizes most drugs differently from the non-obese patient making drug management very challenging. There is increased sensitivity to respiratory depressant effects of sedatives such as benzodiazepines. Drug dosing based on total body weight results in over dosing and prolonged effects which could be very serious in respiratory depressant and myocardial depressant drugs.10 Soluble inhalation agents accumulate in adipose tissue leading to longer clearance time and prolonged effects in the obese patient. Analgesia regimens can also prove very challenging in the obese patients because of increased sensitivity to the sedative effects of opioids. For open procedures, epidural analgesia supplemented by Non-steroidal Anti-inflammatory Drugs (NSAIDs) avoid the side effects of opioids. Other useful techniques include local anesthetic delivery through wound catheters and intravenous patient-controlled analgesia in a monitored setting. The bariatric surgery patient has increased risk for thromboembolic complications and thromboembolic prophylactic measures as described elsewhere in this text are important considerations in their management. Most of the surgical procedures are conducted laporoscopically with documented advantages in pain control, and other outcome parameters. Pneumoperitoneum itself predisposes to atelectasis and hypoxemia with decrease in FRC and increased compliance but there is no difference in the hemodynamic and partial pressure of carbon dioxide (PCO2) effects between the obese and non-obese patients.11,12 However, the uniform drop in renal and hepatic perfusion with pneumoperitoneum should be considered in the bariatric surgery patient some of whom may have borderline hepatic or renal dysfunction, emphasizing the need for maintaining adequate circulating blood volume. Despite the many challenges presented by the bariatric surgery patients, anesthesia and surgery can be performed safely in these patients particularly with normal cardiac, pulmonary, renal, and hepatic function. In the patients with significant associated morbidity, outcome can still be very good if careful attention is directed at preoperative assessment and perioperative care based on appropriate preparation and monitoring. Obesity is an epidemic that is replacing more traditional public health concerns such as under-a nutrition and infectious diseases.1 Since 1980, the worldwide prevalence of obesity has doubled.13 In North America, the prevalence of obesity is alarmingly high with 24.1% of Canadians and 34.4% of Americans having a body mass index (BMI) over 30 kg/m2.13,14 With the obesity epidemic, there has been a rise in the prevalence of obesity-related co-morbidities such as type 2 diabetes, coronary heart disease, hyperlipidemia, and hypertension.15 In addition to increasing morbidity, obesity in and of itself leads to increased mortality by markedly lessening life expectancy.16–18 Although diet, exercise, and medical therapy have been attempted, bariatric surgery appears to be the only effective and durable treatment for obesity and its weight-related comorbidities.19,20 In the Swedish Obese Subjects (SOS) trial, Sjostrom and colleagues have demonstrated that when compared to conventional therapy, bariatric surgery results in long-term weight loss, improved lifestyle, and amelioration of obesity-related comorbidities.21 Follow-up to this prospective cohort study demonstrates that bariatric surgery not only leads to long-term weight loss and improvement of obesity-related comorbidities but also leads to decreased overall mortality.22 BMI is an index of weight-for-height that is used to classify obesity. It is defined as weight in kilograms divided by the square of height in meters (kg/m2). Obese individuals have a BMI over 30 kg/m2. Obesity is further classified into three categories; Class I obesity is defined as a BMI between 30.00 kg/m2 and 34.99 kg/m2, Class II obesity is defined as a BMI between 35.00 kg/m2 and 39.99 kg/m2, and Class III obesity is defined as a BMI over 40.00 kg/m2. Eligibility for bariatric surgery continues to be determined by the 1991 National Institutes of Health (NIH) consensus conference on gastrointestinal surgery for severe obesity. According to the NIH consensus, individuals are candidates for bariatric surgery if they are morbidly obese (BMI >40 kg/m2 or BMI >35 kg/m2 with obesity-related comorbidities), have failed numerous attempts at diet and exercise, are motivated and well-informed, and are free of significant psychological disease.12 Operations to alter the gastrointestinal tract to induce weight loss have been in practice for over 60 years. Bariatric procedures are classified as purely malabsorptive, purely restrictive, or a combination of the two. Following early animal studies by Kremen in 1954, jejunoileal bypass was introduced as one of the first procedures to address the surgical management of obesity.13 This purely malabsorptive bariatric procedure fell out of favor due to severe complications related to bacterial overgrowth and hepatic failure.14 In an effort to enhance the malabsorptive benefits of bariatric surgery while minimizing its complications, biliopancreatic diversion (BPD), was introduced by Scopinaro in 1979 as an alternative to jejunoileal bypass.15 Several modifications ensued including the addition of the duodenal switch (DS) by Hess and Hess and the replacement of a distal gastrectomy with the creation of a sleeve gastrectomy by Marceau and colleagues.16 Selected bariatric centers continue to employ BPD–DS in the surgical management of morbid obesity. In the early 1970’s, Printen and Mason introduced the gastroplasty.17 This purely restrictive bariatric procedure was later modified to include a vertical banded gastroplasty (VBG)18 and then ultimately developed into the laparoscopic VBG in the 1990’s.19 Due to long-term failures and high rates of revisional surgery, procedures involving gastroplasty have fallen out of favor. As an alternative to gastroplasty, Wilkinson described the use of a non-adjustable prosthetic material wrapped around the proximal stomach over a Nissen fundoplication in the early 1980’s.20 This purely restrictive procedure was later modified by Kuzmak and colleagues in the early 1990’s to include the placement of an inflatable silicone gastric band.21 In 1993, Belachew and colleagues were the first to place this silicone gastric band laparoscopically thus championing the laparoscopic adjustable gastric band (LAGB).22 LAGB continues to be employed by selected centers in the surgical management of morbid obesity. Mason and Ito are credited as the first to employ the gastric bypass as a surgical procedure for morbid obesity.23 While initially utilizing a loop gastrojejunostomy and a significantly larger gastric pouch, these two surgeons were able to demonstrate the effectiveness of this bariatric procedure. Two critical modifications to the Roux-en-Y gastric bypass (RYGB) during the subsequent four decades include the replacement of a loop gastrojejunostomy with a Roux-en-Y by Griffen and colleagues24 and the laparoscopic approach (LRYGB) championed by Wittgrove and colleagues.25 With over 90% of bariatric procedures being performed laparoscopically, the LRYGB has emerged as the gold standard in the treatment of morbid obesity in North America.26 A more recent addition to the surgical management of morbid obesity is the laparoscopic sleeve gastrectomy (LSG). LSG was first developed by Marceau and colleagues as a component of BPD–DS.16 Due to challenges associated with higher morbidity, mortality, and long-term weight loss failure in the super obese, Regan and colleagues proposed LSG as a first stage procedure in a two-stage approach to LRYGB.27 Given the favorable outcomes of this first stage procedure, Baltasar and colleagues suggested that LSG is reasonable as a stand-alone procedure.28 Although long-term outcomes of this bariatric procedure are not fully known, LSG remains a viable option in the surgical management of morbid obesity. LAGB involves placing an inflatable silicone band around the proximal portion of the stomach to achieve weight loss via a purely restrictive mechanism (Fig. 1). This bariatric procedure is accomplished with minimal dissection. A retrogastric tunnel is created between the pars flaccida and right crus medially, and the phrenoesophageal ligament laterally. By placing a blunt instrument through this passage, the band is grasped and passed through the retrogastric tunnel. Once the band is in position, the tubing is passed through the buckle of the band and fastened. An anterior gastric tunnel is created so as to prevent band prolapse. This is achieved by placing anterior gastrogastric sutures from the greater curvature of the stomach to the lesser curvature of the stomach. Finally, the tubing is externalized and connected to an external port that is placed in the subcutaneous tissues. Fig. 1. LAGB. A — Inflatable silicone band; B — Tube connecting band to port; C — External port.
The Bariatric Surgical Patient
Andrew Smith, Jameel Ali and Timothy D. Jackson
Chapter Overview
General considerations in the morbidly obese that could impact ICU care
Airway management
Ventilatory management
Cardiovascular factors
Pharmacokinetics
Miscellaneous factors
Bariatric Surgery Procedures
Classification of Obesity and Criteria for Surgery
Evolution of Bariatric Surgery
Bariatric Procedures
Laparoscopic Adjustable Gastric Band (LAGB)
Laparoscopic Sleeve Gastrectomy (LSG)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree