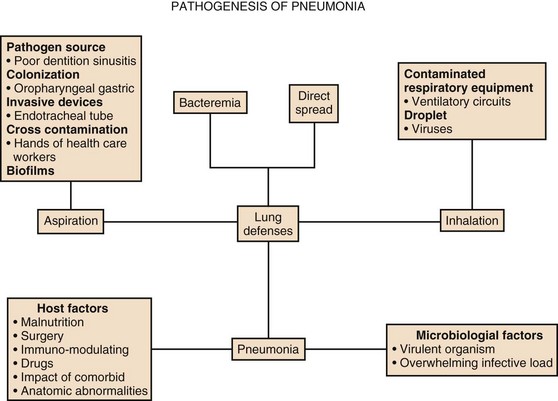
42 Colonization of the Upper Respiratory System and Digestive Tract The Role of Respiratory Therapy Equipment and Endotracheal Tubes EVALUATION OF NONRESPONDING PATIENTS Severe pneumonia with respiratory failure and sepsis is one of the commonest indications necessitating intensive care unit (ICU) admission. Pneumonia along with influenza was the eighth leading cause of death in 2009 alone. Pneumonia patients are managed in the ICU when severe forms of community-acquired pneumonia (CAP) are present or when a hospitalized patient develops life-threatening nosocomial pneumonia. Pneumonia patients admitted to the ICU usually have more severe disease, other comorbid medical conditions, and much higher mortality rate compared to the patients admitted to the wards.1 The incidence of pneumonia in hospitalized patients is directly related to the degree of underlying systemic illness and number of days on the ventilator.2 The elderly account for a disproportionate number of critically ill patients with all forms of pneumonia, often because they commonly have comorbid illness, which predisposes them to more severe forms of infection, and their short- and long-term mortality rates are higher than those of younger patients.3 Also, hospitalized patients receive therapeutic interventions that predispose them to pneumonia such as endotracheal intubation, nasogastric feeding, antibiotic therapy, and use of immunomodulatory medications. In 2005, there were more than 60,000 deaths in persons aged 15 years or older related to CAP in the United States.4 The mortality rate from severe CAP is over 30% in patients admitted to the ICU.5 Critically ill patients who develop ventilator-associated pneumonia (VAP) have a higher mortality rate compared with similar patients without VAP and incur USD $10,019 or more in additional hospital costs.6 With newer guidelines, there has been an emphasis on identifying patients with severe pneumonia early in the clinical course, facilitating early ICU admission, because this has been shown to decrease the mortality rate.7 Different scoring systems such as the ATS/IDSA guidelines (developed by American Thoracic Society and Infectious Diseases Society of America), PIRO (Predisposition, Insult, Response and Organ dysfunction) scores for CAP, and Acute Physiology and Chronic Health Evaluation score (APACHE II) and VAP PIRO score for VAP are used for assessing the severity of illness in pneumonia patients. It is still uncertain whether the use of any scoring system can lead to decreased mortality rates and more favorable outcomes in the management of patients with pneumonia. There is also interest in using biomarkers, such as procalcitonin (PCT), to diagnose the severity and guide the use and duration of antimicrobial therapy in patients with severe CAP and VAP.8–10 The emergence of multidrug-resistant (MDR) microorganisms is an alarming problem in the ICU and considerable forethought is necessary in choosing the right antibiotic to be used in this setting. In the ICU, almost 90% of episodes of nosocomial pneumonia occur in patients who are being mechanically ventilated for other reasons. Recent directives by the Centers for Medicare and Medicaid Services (CMS) and the Institute for Healthcare Improvement have led to the belief that VAP is preventable and should be a “never event” during the hospital stay. This may have led to underreporting in many instances.11 Many ICUs have incorporated ventilator bundles as a preventive strategy to minimize VAP, which may be an effective strategy, even if not being able to completely eliminate the problem. The incidence of pneumonia in nursing homes varies from 0.3 to 2.3 episodes per 1000 resident care days with a 30-day mortality rate of 14.7%.12 In the 2005 ATS guidelines on nosocomial pneumonia, patients with exposure to MDR organisms by virtue of being in contact with the health care environment prior to admission were defined as having health care–associated pneumonia (HCAP). This group included residents of a nursing home or long-term care facility; recipients of recent intravenous antibiotic therapy, chemotherapy, or wound care within the past 30 days; and chronic hemodialysis patients.2 The focus in managing these patients was to empirically treat for MDR pathogens. However, recent studies have shown that the HCAP population has significant heterogeneity, and that treatment of all patients does not always need to include coverage for methicillin-resistant Staphylococcus aureus (MRSA) and drug-resistant gram-negative organisms.13 Patients who are unable to mount an effective immune response are at increased risk of developing serious pneumonia. If the host defense mechanism is overwhelmed by the size of the inoculum, the virulence of the infecting microorganisms, or an excessive inflammatory response to infection, the patient can develop severe respiratory failure or sepsis. Depending on the time of onset and setting in which the infection developed, pneumonia is classified as community-acquired or nosocomial pneumonia, with specific definitions for VAP and HCAP among patients with nosocomial pneumonia (Box 42.1). Several prognostic scoring systems were developed to predict the mortality risk and guide the site of care decision. The pneumonia severity index (PSI) and the British Thoracic Society’s CURB-65 score help with determining mortality risk for patients with CAP.14 The PSI was developed to identify patients with a low risk of dying, placing all patients into five classes, based on the demographic characteristics, coexisting illnesses, physical examination findings, laboratory measurements, and radiographic findings. The ICU is most often used for patients in classes IV and V, who have a 30-day mortality risk of 4% to 10% and 27%, respectively.15 However, the PSI score may overestimate severity of illness in patients with older age, while underestimating severity in young patients without comorbid conditions.16 The complexity of the PSI contrasts with the simpler bedside scoring system, CURB-65. With this tool, 1 point is assigned each for the presence of confusion, blood urea nitrogen (BUN) greater than 7.0 mol/L (19.6 mg/dL), respiratory rate of 30 or more breaths/minute, systolic blood pressure 90 mm Hg or less, or diastolic blood pressure 60 mm Hg or less, and age 65 years or more.17 Mortality rate increases proportionately with scores higher than 3 (score 3, 17%; score 4, 41.5%; and score 5, 57%). Severe pneumonia and the host inflammatory response to infection are determined by several variables, including the identity of the pathogen, the timeliness and appropriateness of therapy, and patient variables. The latter include the genetic makeup of the individual, age, nutritional status, sex, and comorbid medical conditions. Although the PSI and CURB-65 score performed well to determine mortality risk, there is still a need to identify patients who will need ICU level of care at the earliest possible time point.18,19 According to the 2007 ATS/IDSA guidelines, severe CAP is present if a patient requires invasive mechanical ventilation or vasopressors or has any three of the nine minor criteria (Box 42.2).19 The PIRO score has also been used and is calculated within 24 hours of ICU admission with 1 point assigned for each of comorbid conditions (chronic obstructive pulmonary disease [COPD], immunosuppressed state), age older than 70 years, multilobar opacities on chest radiograph, shock, severe hypoxemia, acute renal failure, bacteremia, and acute respiratory distress syndrome (ARDS). Patients can be stratified into four classes: (1) low, 0 to 2 points; (2) mild, 3 points; (3) high, 4 points; (4) very high, 5 to 8 points. In patients admitted to the ICU, the PIRO score had a better performance than APACHE II and ATS/IDSA criteria to predict 28-day mortality rate.18 Severe pneumonia is a dynamic inflammatory process, and predicting which stable-appearing patients will require ICU care later is difficult. Hence, critically ill patients who do not have obvious need for ICU care, such as invasive respiratory/vasopressor support (IRVS), must satisfy certain minor criteria in order to be recognized. However, the predictive value of using just three minor criteria alone for making a decision to admit to the ICU is uncertain, and in one study, the use of four minor criteria improved the accuracy of predicting subsequent need for ICU care.20,21 A recent prospective study from Scotland excluded patients with the two major ATS/IDSA criteria present on admission, and demonstrated that each of the nine minor criteria was associated with an increased risk of need for mechanical ventilation, need for vasopressor support, and 30-day mortality.22 Another system developed in Australia—SMART-COP—can assess the need for intensive respiratory or vasopressor support.23 The SMART-COP was developed primarily to identify the need for IRVS. Different point scores were assigned to various parameters: low Systolic blood pressure less than 90 mm Hg (2 points), Multilobar pneumonia (1 point), low Albumin level less than 3.5 g/dL (1 point), high Respiratory rate 25 to 30 breaths/minute (1 point), Tachycardia higher than 125 beats/minute (1 point), Confusion (1 point), poor Oxygenation (2 points), and low arterial pH less than 7.35 (2 points). A score of more than 3 points identified 92% of patients requiring IRVS, with a specificity of 62.3%, outperforming PSI and CURB-65 scores to identify this specific end point.23 The SMART-COP as well as the ATS/IDSA 2007 guidelines are the most accurate for predicting need for ICU care.24 Renaud and coworkers examined risk factors for early admission to the ICU among 6560 patients, who presented to the emergency department and did not require immediate respiratory or circulatory support.25 They identified 11 criteria independently associated with ICU admission: male gender, age younger than 80 years, comorbid conditions, respiratory rate of 30 breaths/minute or higher, heart rate of 125 beats/minute or higher, multilobar infiltrate or pleural effusion, white blood cell count less than 3 or more than 20 g/L, hypoxemia (oxygen saturation <90% or arterial partial pressure of oxygen [PaO2] <60 mm Hg), BUN of 11 mmol/L or higher, pH less than 7.35, and sodium less than 130 mEq/L. They used these criteria to develop the risk of early admission to ICU index (REA-ICU index), which stratified patients into four risk classes, with the risk of ICU admission on days 1 to 3 ranging from 0.7% to 31%.25 Recent data from the German Competence Network for the Study of CAP (CAPNETZ) Study Group demonstrated that serum biomarkers (PCT) can be used as good predictors for short- and long-term all-cause mortality rates in patients admitted with CAP.26 A low level of PCT value in patients classified as high risk by PSI and CURB-65 scores predicted a low risk of dying, and probably a low need for ICU admission.27 Ramirez and colleagues showed that the ATS/IDSA 2007 minor criteria had comparable predictive value with other scores, such as CURB-65 and SMART-COP, but that the use of biomarkers like PCT and C-reactive protein (CRP), along with minor criteria, helped in predicting the need for delayed ICU admission.28 Pathophysiologic severity markers of severe sepsis, like soluble receptor for advanced glycation end products (SRAGE) along with severity scores have been examined in patients with CAP and are encouraging but need further studies for validation.29 Most patients with severe CAP (45-65%) have coexisting illnesses, and patients who are chronically ill have an increased likelihood of developing a complicated pneumonary illness (Box 42.3).30 The most common chronic illnesses in these patients are respiratory disease such as COPD, cardiovascular disease, and diabetes mellitus. In addition, certain habits such as cigarette smoking and alcohol abuse are also quite common in those with severe CAP, and cigarette smoking has been identified as a risk factor for bacteremic pneumococcal infection.31 Other common illnesses in those with CAP include malignancy and neurologic illness (including seizures). Milder forms of pneumonia may be more severe on presentation, if patients have not received antibiotic therapy prior to hospital admission. In addition, genetic differences in the immune response may predispose certain individuals to more severe forms of infection and adverse outcomes and may be reflected by a family history of severe pneumonia or adverse outcomes from infection.32 Also, genetic variability of the pulmonary surfactant proteins A and D may affect clearance of microorganisms and the extent of the inflammatory response, influencing the severity and outcomes for patients with CAP, notably missense single nucleotide polymorphisms and haplotypes of SFTPA1, SFTPA2, and SFTPD.33 Recent evidence also provides insights on the risk of developing pneumonia in patients taking inhaled corticosteroids. In a meta analysis of 18 randomized controlled trials with a total of 16,996 patients, inhaled corticosteroids were associated with a significantly increased risk of serious pneumonia when compared with placebo (relative risk [RR] 1.81; P < 0.001) or when the combination of inhaled corticosteroids and long-acting β-agonists was compared with long-acting β-agonists (RR 1.68; P < 0.002).34 However, the mortality rate of patients developing CAP while on inhaled corticosteroids may actually be lower than in patients not receiving this therapy. In a meta analysis of 33,148 patients with CAP, the overall mortality rate was 13.7%, but those admitted to the ICU had a mortality rate of 36.5%.5 Eleven prognostic factors were significantly associated with different odds ratios (OR) for mortality rate: male sex (OR = 1.3), pleuritic chest pain (OR = 0.5), hypothermia (OR = 5.0), systolic hypotension (OR = 4.8), tachypnea (OR = 2.9), diabetes mellitus (OR = 1.3), neoplastic disease (OR = 2.8), neurologic disease (OR = 4.6), bacteremia (OR = 2.8), leukopenia (OR = 2.5), and multilobar infiltrates (OR = 3.1). In other studies, the clinical features that predict a poor outcome (Box 42.4) include advanced age (>65 years), preexisting chronic illness of any type, the absence of fever on admission, respiratory rate more than 30 breaths/minute, diastolic or systolic hypotension, elevated BUN (>19.6 mg/dL), profound leukopenia or leukocytosis, inadequate antibiotic therapy, need for mechanical ventilation, hypoalbuminemia, and the presence of certain “high-risk” organisms (type III pneumococcus, S. aureus, gram-negative bacilli, aspiration organisms, or postobstructive pneumonia). Timing of initial appropriate antibiotics is also important, with a delay in the initiation of appropriate antibiotic therapy of more than 4 hours being associated with increased mortality risk.35–37 Other clinical features associated with an increased mortality risk include rapid radiographic progression during therapy, nonrespiratory clinical presenting symptoms, and the presence of HCAP risk factors. Many studies suggest that early ICU admission confers a better outcome in patients with severe CAP than delayed admission. In a study comparing patients directly admitted to the ICU from the emergency department to those moved to the ICU within 3 days after admission, delayed-transfer patients had a higher 28-day mortality rate (23.4% vs. 11.7%; p = 0.02) and a longer median hospital length of stay (13 days vs. 7 days; p < 0.001).38 In a study of 17,869 cases of CAP in the United Kingdom, only 5.9% needed ICU care, but early admission (within 2 days of hospitalization) appeared to be preferable and was associated with a lower mortality rate (46.3%) than was late admission (>7 days in the hospital, 57.6% mortality rate).7 In patients with severe CAP, the expected mortality rate for those admitted to the ICU is 35% to 40%, but higher rates have been observed if the percentage of patients who are mechanically ventilated is higher than 60%, implying that the prognosis is worse if ICU care is first provided late in the course of illness, after the onset of overt respiratory failure.7 Restrepo and colleagues noted that late admission to the ICU, more than 24 hours after presentation in patients with severe CAP, resulted in a higher mortality rate than did early admission (47.4% vs. 23.2%, p = 0.02; hazard ratio 2.6).39 Hospital-acquired pneumonia (HAP) is the second most common nosocomial infection in the United States,40 and current guidelines have emphasized the importance of the time of onset of the disease and the presence of risk factors for infection due to MDR pathogens in defining the approach to therapy. The major goals are early, appropriate antibiotic therapy in adequate doses and appropriate de-escalation of initial antibiotic therapy, based on microbiologic cultures and the clinical response of the patient.2 Unlike CAP, there are no well-studied scoring systems to assess disease severity in patients with HAP and VAP. Clinical studies have used surrogate markers such as the APACHE II, or the Sequential Organ Failure Assessment (SOFA) score as a measure of organ dysfunction and severity assessment.41 The Clinical Pulmonary Infection Score (CPIS) was originally introduced for diagnoses in patients with VAP (described later in the text), but it has also been used to follow the response of VAP patients to therapy.42 The value of APACHE II, SOFA score, and CPIS in the prediction of mortality risk during VAP episodes was assessed in a prospective observational study, which found that an APACHE II score greater than 16 (determined at the time of VAP diagnosis) was the only independent predictor of the mortality risk (OR 5; p = 0.019) in a logistic regression analysis.43 In a prospective study by Lisboa and colleagues from Spain, including 441 patients with VAP in three multidisciplinary ICUs, the mortality risk in patients with VAP was assessed by a simple four-variable VAP PIRO score: comorbid conditions (COPD, immunocompromised state, heart failure, cirrhosis, or chronic renal failure); bacteremia; systolic blood pressure less than 90 mm Hg; and ARDS.44 Patients were stratified into three levels of risk: (1) mild, 0 to 1 points; (2) high, 2 points (hazard ratio, 2.14); and (3) very high, 3 to 4 points (hazard ratio, 4.63). More recently, a newer and easier scoring system, the IBMP-10, was proposed and is based on the presence of immunodeficiency; (2) blood pressure less than 90 mm Hg (systolic) or less than 60 mm Hg (diastolic); multilobar infiltrates noted on a chest radiograph; platelet count less than 100,000/µL; and duration of hospitalization before the onset of VAP of more than 10 days.45 IBMP-10 score was comparable to the APACHE II score in its ability to predict mortality risk in patients with VAP but needs further validation in prospective studies. The 2005 ATS/IDSA guidelines characterized the predisposing factors for developing nosocomial pneumonia as modifiable and nonmodifiable. Mechanical ventilation (for > 2 days) is the most important risk factor for nosocomial pneumonia, but other identified risks include age older than 60, malnutrition (serum albumin < 2.2 g/dL), acute lung injury (ARDS), coma, burns, recent abdominal or thoracic surgery, multiple organ failure, transfusion of more than 4 units of blood, transport from the ICU, prior antibiotic therapy, elevation of gastric pH (by antacids or histamine-type 2 blocking agents), large volume aspiration, use of a nasogastric tube (rather than a tube placed in the jejunum or a tube inserted through the mouth), use of inadequate endotracheal tube cuff pressure, prolonged sedation and paralysis, maintaining patients in the supine position in bed, use of total parenteral nutrition (TPN) feeding rather than enteral feeding, and repeated reintubation.2 When a patient is mechanically ventilated, the risk of pneumonia is greatest in the first 5 days (3% per day), and declines thereafter to a risk of 2% per day for days 6 to 10, and to a rate of 1% per day or lower after this.46 Noninvasive ventilation for respiratory failure is associated with a much lower risk of pneumonia than endotracheal intubation. The relation between pneumonia and ARDS is particularly interesting, because up to one third of all cases of ARDS may be the result of pneumonia, and in some series, pneumonia is the most common cause of acute lung injury. In addition, secondary nosocomial pneumonia is the most common infection complicating the course of established ARDS.47,48 Seidenfeld and coworkers reported better survival in patients with ARDS in the absence of infection, but a subanalysis for pneumonia was not available.48 In a study by Chastre and colleagues of 243 consecutive patients who required mechanical ventilation 48 or more hours, 55% of the ARDS patients developed VAP compared to 28% without ARDS (p = 0.0005).49 Most patients who developed VAP had been treated with prior broad-spectrum antibiotics, and were infected with MDR pathogens (MRSA, nonfermenting gram-negative bacilli, and Enterobacteriaceae). Mortality rates from VAP can be as high as 50% to 70%, and case-control studies have documented death directly attributable to the presence of pneumonia.50 The patients with HAP often have other associated comorbid illnesses that predispose them to a high risk of dying, independent of the presence of pneumonia. Attributable mortality rate, defined as death directly related to infection and not due to underlying conditions, is challenging to measure, but older studies reported a higher mortality rate for patients with VAP than for similarly ill ventilated patients without VAP (52.4% with VAP, compared to 22.4% for patients without).51 Recently, however, Bekaert and colleagues assessed the population-attributable risk of ICU VAP by taking into account the confounding that is caused by time-dependent severity-of-illness indicators.52 They estimated that 4.4% (95% confidence interval, 1.6-7.0%) of the deaths in the ICU on day 30 and 5.9% (95% confidence interval, 2.5-9.1%) on day 60 were attributable to VAP. This corresponds to an ICU attributable mortality rate for VAP of about 1% on day 30 and 1.5% on day 60. Antibiotic-resistant organisms may add to the mortality risk of VAP, not because of increased virulence, but rather because these organisms are often not anticipated, and when present, are often initially treated with ineffective antibiotic regimens.53 The factor associated with the greatest impact on attributable mortality rate is the accuracy and timeliness of initial antibiotic therapy. Use of the wrong therapy, or delays in the initiation of therapy, are the most important predictors of VAP mortality rate.54 Initial appropriate therapy (using an agent to which the etiologic pathogen is sensitive) can reduce mortality rate, but administration of correct therapy at a later date, after initially incorrect therapy, may not effectively reduce mortality rate.54 The benefit of accurate empiric therapy may not apply to all patients, but may be greatest for those infected with P. aeruginosa or S. aureus55 and for those without the most severe degree of multiple organ dysfunction at the time of therapy.56 For some patients, even using the correct therapy does not reduce the risk of death if it is not given in adequate doses and if the therapy does not reach the site of infection. Although a number of host and bacteriologic factors enhance the mortality risk of nosocomial pneumonia, development of a superinfection, as opposed to primary nosocomial pneumonia, is a particularly ominous finding. Rello and associates observed that pulmonary superinfection had a 67% mortality rate, whereas primary nosocomial pneumonia had a 38% mortality rate.57 In earlier studies, Graybill and coworkers observed a 62% mortality rate with superinfection pneumonia, compared to a 40% mortality rate for primary nosocomial lung infection.58 These data, as well as information from Fagon and colleagues, emphasize the important role of prior antibiotics in enhancing mortality risk, an outcome that is likely the result of secondary infection by more virulent pathogens.59,60 As a result, antibiotic use has two pivotal roles in prognosticating outcome from nosocomial pneumonia: outcome is improved if the correct therapy is chosen, but if this therapy is followed by superinfection, then mortality risk is increased, generally because these infections involve difficult-to-treat drug-resistant organisms. Closely related to antibiotic resistance is the presence of bacteremia in patients with VAP, which is associated with increased mortality risk.61 When therapy is given, it is important to decrease the number and spectrum of antimicrobial therapy once culture data become available, referred to as “de-escalation.” Several recent studies have demonstrated that the use of de-escalation is associated with lower mortality risk compared to escalation or compared to a strategy of making no effort to reduce antibiotic therapy.62–64 The choice of how to administer a specific agent can also affect outcome, and one study of MRSA VAP found that the mortality risk with intermittent infusion of vancomycin was twice as high as when this agent was administered by continuous infusion.65 Other risk factors for mortality (Box 42.5) include prolonged duration of ventilation, coma on admission, creatinine greater than 1.5, transfer from another ward to the ICU, the presence of certain high-risk pathogens (particularly an antibiotic-resistant organism such as P. aeruginosa, Acinetobacter spp., or S. aureus), bilateral radiographic abnormalities, age older than 60 years, ultimately fatal underlying condition, shock, prior antibiotic therapy, multiple system organ failure, nonsurgical primary diagnosis, and a rising APACHE II score during pneumonia therapy.2,66 The ATS/IDSA guidelines included HCAP as a form of nosocomial pneumonia because these patients were at risk for infection with MDR pathogens because of recent contact with the health care environment. HCAP includes patients who were hospitalized in an acute care hospital for 2 or more days within 90 days of the infection; those who reside in a nursing home or long-term care facility; individuals who received recent intravenous antibiotic therapy, chemotherapy, or wound care within the past 30 days; and those attending a hospital or hemodialysis clinic. Recent clinical investigations have shown that HCAP includes a diverse group of patients, with some at risk for MDR organisms and others not. In fact, many HCAP patients have been treated successfully with a monotherapy regimen or with regimens used for CAP.13 Patients in the HCAP population who are at risk for MDR pathogens were those with severe illness or those with other risk factors including hospitalization in the past 90 days, antibiotic therapy in the past 6 months, poor functional status as defined by activities of daily living score, and immune suppression. The respiratory system has both innate and adaptive immunity, which helps prevent potentially harmful pathogens from adhering to the respiratory mucosa and proliferating. Multiple defense barriers in the conducting and gas exchange surfaces of the respiratory tract filter out invading pathogens. The combined effects of the physical, innate, and acquired host defense systems serve to recognize, localize, kill, and remove pathogens. The pathogens that reach the conducting airways are exposed to the soluble constituents in airway fluids. Multiple antimicrobial peptides, such as lysozyme (lytic to bacterial membranes); lactoferrin (which excludes iron from bacterial metabolism); IgA and IgG; and defensins are present in the airway mucosa and pathogens are expelled by the coordinated mucociliary system and cough reflex. Particles 1 µm and smaller reach the alveolar surface and interact with the alveolar macrophages and other components such as IgG, complement, surfactant, and surfactant-associated proteins. Bacteria are opsonized by IgG, complement, or surfactant proteins SP-A and SP-D and are ingested by alveolar macrophages. Pathogen recognition is mediated by toll-like receptors. They initiate local inflammatory response by releasing interleukins and cytokines and carry microbial antigens into the interstitium and to regional lymph nodes where they are taken up by specialized dendritic cells and presented to responding lymphocytes to initiate adaptive immune responses.67 Pneumonia develops when these host defenses are overwhelmed by the invading microorganisms (Fig. 42.1). This may occur because the patient has an inadequate immune response, often as the result of underlying comorbid illness, which can lead to anatomic abnormalities (endobronchial obstruction, bronchiectasis), disease-associated immune impairment, or therapy-induced dysfunction of the immune system (corticosteroids, endotracheal intubation).2,68,69 Pneumonia can also occur in patients who have an adequate immune system if the host defense system is overwhelmed by a large inoculum of bacteria (massive aspiration) or by a particularly virulent organism to which the patient has no preexisting immunity (such as an endemic virus) or to which the patient has an inability to form an adequate immune response. With this paradigm in mind, it is easy to understand why previously healthy individuals develop infection with virulent pathogens such as viruses (influenza), Legionella pneumophila, Mycoplasma pneumoniae, Chlamydophila pneumoniae, and S. pneumoniae. However, for chronically ill patients, it is possible for them to be infected not only by these virulent organisms but also by organisms that are not highly virulent. Owing to host defense impairments, organisms that commonly colonize these patients can cause infection as a result of an inadequate immune response. These organisms include enteric gram-negative bacteria (E. coli, Klebsiella pneumoniae, P. aeruginosa, Acinetobacter spp.) and fungi (Aspergillus and Candida spp.). There can also be genetic variations in the immune response, making some patients prone to overwhelming infection, due to an inadequate response, and others prone to acute lung injury, due to an excessive immune response.32 In fact, the failure to localize the immune response to the respiratory site of initial infection may explain why some patients develop acute lung injury and sepsis as the inflammatory response extends to the entire lung and systemic circulation.70 Bacteria can enter the lung via several routes, but aspiration from a previously colonized oropharynx is the most common way that patients develop pneumonia. Although most pneumonias result from microaspiration, patients can also aspirate large volumes of bacteria if they have impaired neurologic protection of the upper airway (stroke, seizure), or if they have gastrointestinal illnesses that predispose to vomiting. Other routes of entry include inhalation, which applies primarily to viruses, L. pneumophila, and Mycobacterium tuberculosis; hematogenous dissemination from extrapulmonary sites of infection (right-sided endocarditis); and direct extension from contiguous sites of infection. In critically ill hospitalized patients, bacteria can also enter the lung from a colonized stomach (spreading retrogradely to the oropharynx, followed by aspiration), from a colonized or infected maxillary sinus, from colonization of dental plaque, or directly via the endotracheal tube (from the hands of staff members). Studies have also shown that the use of nasal tubes (into the stomach or trachea), can predispose to sinusitis and pneumonia, but that a gastric source of pneumonia pathogens in ventilated patients is not common.71,72 Colonization of the upper respiratory and digestive tracts with pathogenic microorganisms is a major risk factor for the development of pneumonia.73 Factors enhancing airway colonization include antibiotic therapy, endotracheal intubation, smoking, malnutrition, general surgery, dental plaque, and therapies that elevate the gastric pH.74 The stomach can be the source of 30% of the enteric gram-negative bacteria that colonize the trachea of intubated patients, but it is difficult to decide if such colonization leads to pneumonia. In a recent multicenter randomized controlled study by Lacherade and colleagues, the use of intermittent subglottic secretion drainage resulted in a significant reduction in microbiologically confirmed VAP compared to control subjects (14.8% vs. 25.6%; P = 0.02),75 without a significant difference in the duration of mechanical ventilation and hospital mortality rate. The findings suggest that interruption of gastric to oral to tracheal transmission of bacteria can help to prevent VAP. The importance of oral colonization in VAP pathogenesis was shown in a study by DeRiso and coworkers, which demonstrated that the use of the oral antiseptic chlorhexidine in 353 patients undergoing coronary artery bypass surgery significantly reduced the incidence of respiratory tract infections by 69%.76 It has become difficult to separate colonization from infection in intubated patients, particularly with the recognition of ventilator-associated tracheobronchitis (VAT).77 VAT patients have an infection, with clinical signs (fever, leukocytosis, and purulent sputum) and microbiologic findings (Gram stain with bacteria and leukocytes, with either a positive semiquantitative or a quantitative sputum culture) but the absence of a new infiltrate on chest radiograph. It remains uncertain if VAT can progress to VAP, or if the two events are independent of one another. If VAT is a precursor of VAP, then serial surveillance cultures of endotracheal aspirates could identify MDR pathogens for targeted antibiotic treatment when VAT is present, in an effort to prevent VAP.78 The endotracheal tube bypasses the filtration and host defense functions of the upper airway and can act as a conduit for direct inoculation of bacteria into the lung. This route may be particularly important if bacteria form a biofilm and colonize the inside of the endotracheal tube itself.79,80 This can occur if tracheobronchial organisms reach the endotracheal tube, a site where they are able to proliferate free from any impediment by the host defense system. Bacteria commonly do grow at this location in a biofilm, which promotes the growth of MDR organisms.80 The biofilm represents a “sequestered nidus” of infection on the inside of the endotracheal tube, and particles can be dislodged every time the patient is suctioned. This is one of the mechanisms explaining the strong association between endotracheal intubation and pneumonia. Given the presence of biofilm in endotracheal tubes, it may be tempting to regularly reintubate patients and use a fresh tube, but this approach is not recommended because reintubation is itself a risk factor for VAP.81 Afessa and colleagues reported that the use of the silver-coated tube reduced the mortality rate of patients who developed VAP despite having the silver tube in place, compared with the mortality rate of patients who developed VAP with a standard tube in place (14% vs. 36%; P = 0.03).82 However, the overall mortality rate was high in patients with the silver tube, and the rate of death from respiratory failure was higher in the silver tube–treated patients than those with the standard tube (19% vs. 11%, P = 0.02), raising doubts on the impact of using the silver-coated tube on VAP patients. Other interventions, such as use of devices to remove biofilm from the tube interior and the coating of tubes with mimics of antimicrobial peptides (ceragenins), are in development to interrupt the pathogenesis of VAP.83 Just as a patient’s own tracheobronchial flora can spread to the endotracheal tube and amplify to large numbers, a similar phenomenon can occur in respiratory therapy equipment and in ventilator circuits.84,85 Ventilator circuit colonization has been studied and the greatest bacterial numbers are found at sites nearest to the patient, not the ventilator, suggesting that circuit contamination originates from the patient.84 One highly contaminated site is the condensate in the tubing, and this material can inadvertently be inoculated into patients if the tubing is not handled carefully. Because condensate colonization occurs in 80% of tubings within 24 hours, it does not appear that frequent ventilator circuit changes are useful or even able to reduce the risk of pneumonia; in one study, tubing changes every 24 hours (rather than every 48 hours) served as a risk factor for pneumonia.86 Although most patients have ventilator tubing changed every 48 hours, several studies have shown no increased risk of infection if tubing is never changed or changed infrequently.87,88 The use of heat moisture exchangers may be one way to avoid this problem, but they have had an inconsistent effect on preventing VAP. In addition, frequent changes of heat moisture exchangers (i.e., every 24 hours) have not been shown to have an impact on the incidence of VAP, and heat moisture exchangers should be changed no more frequently than every 48 hours.89 Pneumonia is generally characterized by symptoms of fever, cough, purulent sputum production, and dyspnea in a patient with a new or progressive lung infiltrate, with or without an associated pleural effusion. In nonventilated patients, cough is the most common finding and is present in up to 80% of all CAP patients, but is less common in those who are elderly, those with serious comorbid conditions, and individuals coming from nursing homes. Patients with CAP and an intact immune system generally have classic pneumonia symptoms, but the elderly patient can have a nonrespiratory presentation with symptoms of confusion, falling, failure to thrive, altered functional capacity, or deterioration in a preexisting medical illness, such as congestive heart failure.90 The absence of clear-cut respiratory symptoms and an afebrile status have themselves been identified as predictors of an increased risk of death. Pleuritic chest pain is also commonly seen in patients with CAP, and in one study, its absence was also identified as a poor prognostic finding.91 A recent study pointed out that nonsteroidal anti-inflammatory drugs prior to hospitalization were associated with a higher chance of developing pleuropulmonary complications (OR = 8.1).92 Certain clinical conditions are associated with specific pathogens in patients with CAP, and these associations should be evaluated when obtaining a history (Box 42.6).19 For example, if the presentation is subacute, following contact with birds, rats, or rabbits, then the possibility of psittacosis, leptospirosis, tularemia, or plague, respectively, should be considered. Coxiella burnetti (Q fever) is a concern with exposure to parturient cats, cattle, sheep, or goats; hantavirus with exposure to mice droppings in endemic areas; and Legionella with exposure to contaminated water sources (saunas). Following influenza, superinfection with pneumococcus, S. aureus (including MRSA), and H. influenzae should be considered. With travel to endemic areas in Asia, the onset of respiratory failure after a preceding viral illness should lead to suspicion of a viral pneumonia, which could be severe acute respiratory syndrome (SARS) or avian influenza.93 Endemic fungi (coccidioidomycosis, histoplasmosis, and blastomycosis) occur in well-defined geographic areas and may present acutely with symptoms that overlap with acute bacterial pneumonia. Clinicians should also be cognizant of the risk of bioterrorism and be able to detect clinical features of Bacillus anthracis, Yersinia pestis, and Francisella tularensis. Nosocomial pneumonia often presents with less definitive clinical findings, particularly in those who are mechanically ventilated, in which the clinical diagnosis is made in patients with a new or progressive radiographic infiltrate, along with some indication that infection is present (fever, purulent sputum, or leukocytosis). In addition, some patients can have purulent sputum and fever, without a new infiltrate, and be diagnosed as having purulent tracheobronchitis, an infectious complication of mechanical ventilation (VAT) that may also require antibiotic therapy, but is not pneumonia.2 In taking a history from a patient with nosocomial pneumonia, it is important to identify if there are risk factors present for drug-resistant organisms. For ventilated patients, these factors include prolonged ICU stay (≥5 days), recent antibiotic therapy, and the presence of HCAP risks.2,60 In CAP patients, risk factors for drug-resistant pneumococcus include recent β-lactam therapy, exposure to a child in day care, alcoholism, immune suppression, and multiple medical comorbid conditions.94,95
Pneumonia
Considerations for the Critically Ill Patient
Definitions and Risk Factors
Severe Community-Acquired Pneumonia
Risk Factors for Severe Forms of Community-Acquired Pneumonia
Mortality Risk from Community-Acquired Pneumonia
Pneumonia Acquired in the Hospital
Risk Factors Associated with Nosocomial Pneumonia
Mortality Risk from Ventilator-Associated Pneumonia
Health Care–Associated Pneumonia
Pathogenesis
General Overview
Route of Entry
Colonization of the Upper Respiratory System and Digestive Tract
The Role of Respiratory Therapy Equipment and Endotracheal Tubes
Clinical Features
Historical Information
Pneumonia: Considerations for the Critically Ill Patient