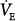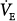Mechanical Ventilation for Severe Asthma: Introduction
It is estimated that 6000 to 10,000 patients require mechanical ventilation for acute asthma in the United States each year.1 Asthma exacerbations that lead to mechanical ventilation typically progress over 1 or more days, with viral infections being the most common etiology (Table 30-1).2 Airways typically show extensive mucous plugging and eosinophilic infiltration with edema, explaining the often limited immediate response to bronchodilators.3 Approximately 20% of patients experience a more explosive onset, with attacks occurring over minutes to hours.2–4 The precise cause of “sudden asphyxial” asthma is not always apparent, but recognized triggers include aeroallergens, nonsteroidal antiinflammatory agents, airway irritants, and emotional distress.2,4 Rapid-onset attacks are characterized by profound bronchospasm with minimal mucous plugging, explaining both their sudden onset and rapid resolution.3
| Slow-Onset | Rapid-Onset | |
|---|---|---|
| Time course (onset) | One or more days | Minutes to hours |
| Triggers | Virus, unknown | Aeroallergen, nonsteroid antiinflammatory drug, airway irritant, emotional stress, unknown |
| Frequency | ∼80% | ∼20% |
| Mechanisms of airflow obstruction | Mucous plugging and airway edema > bronchospasm | Bronchospasm (“dry airways”) |
| Airway inflammation | Eosinophils | Neutrophils |
| Response to treatment | Slow: minimal response to initial bronchodilators | Rapid: good response to initial bronchodilators |
| Prevention | Steroids early in exacerbation | Avoid triggers |
| Duration intubation | Often several days | Often < 24 hours |
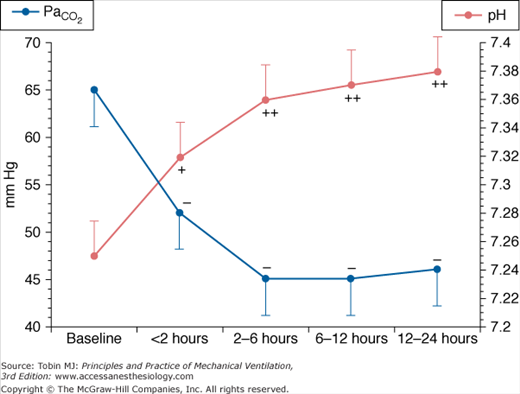
Regardless of the mode of onset, life-threatening asthma is associated with a markedly increased airway resistance, pulmonary hyperinflation, and high physiologic dead space, which together lead to hypercapnia and risk of respiratory arrest.5 Hypercapnia per se is not an indication for intubation, as most asthma exacerbations with hypercapnia do respond to bronchodilator therapy and others may be successfully managed with noninvasive ventilation (Fig. 30-1).6–11 Inhalation of a helium–oxygen gas mixture (heliox) may also reduce work of breathing and might decrease the likelihood of intubation.11,12
Figure 30-1
Change in partial pressure of arterial carbon dioxide (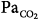 ) and pH associated with the use of noninvasive positive-pressure ventilation by face mask in patients with severe asthma. (Used, with permission, from Meduri GU, Cook TR, Turner RE, et al. Noninvasive positive pressure ventilation in status asthmaticus. Chest. 1996;110:767–774.)
) and pH associated with the use of noninvasive positive-pressure ventilation by face mask in patients with severe asthma. (Used, with permission, from Meduri GU, Cook TR, Turner RE, et al. Noninvasive positive pressure ventilation in status asthmaticus. Chest. 1996;110:767–774.)
Indications for intubation include respiratory arrest, depressed level of consciousness, or progressive fatigue and exhaustion. Rapid-sequence intubation has been advocated by some experts,13 but a high degree of confidence in being able to intubate is a prerequisite before use of a paralytic because it may be difficult to achieve effective bag-mask ventilation in patients with markedly increased airway resistance. Regardless of the technique used, intubation should be performed by the most skilled operator present. Prolonged airway manipulation and repeated failed intubations in the setting of fulminant asthma may prove catastrophic.
Various drugs have been used to facilitate intubation. Ketamine does not cause respiratory depression or hypotension, but may increase laryngeal reflexes and predispose to laryngospasm with excessive upper airway manipulation.14 Propofol decreases airways resistance after intubation as compared with etomidate or thiopental.15 Etomidate, however, causes less hypotension than propofol and may be a better alternative in hemodynamically unstable patients.14 Indeed, many of the drugs used to facilitate intubation reduce vascular tone, which, together with a sudden decrease in venous return secondary to pulmonary hyperinflation, may result in postintubation hypotension.16 The latter should be anticipated and managed by rapid fluid administration and manual ventilation via an Ambu bag at a slow rate to limit the severity of hyperinflation.17 Postintubation hypotension that is profound and refractory to these first-line interventions may be managed with a bolus dose of a rapidly acting vasopressor, such as phenylephrine, vasopressin, or epinephrine.
Ventilator Management
Controlled hypoventilation with permissive hypercapnia was first proposed as a ventilator strategy for severe asthma almost 30 years ago.18 In brief, the rationale for this approach is that hypercapnia poses less risk than does markedly increased lung volume.19,20 Although randomized studies of controlled hypoventilation in asthma are lacking, a retrospective analysis suggested that it is associated with better outcomes than a conventional ventilator strategy.19 To apply controlled hypoventilation rationally, it is essential to understand methods for monitoring pulmonary hyperinflation and how the latter is influenced by the choice of ventilator settings.21–25 Subsequent discussion of ventilator strategy and monitoring will be limited to use of volume-cycled ventilation, the preferred method for managing patients with severe airflow obstruction.23
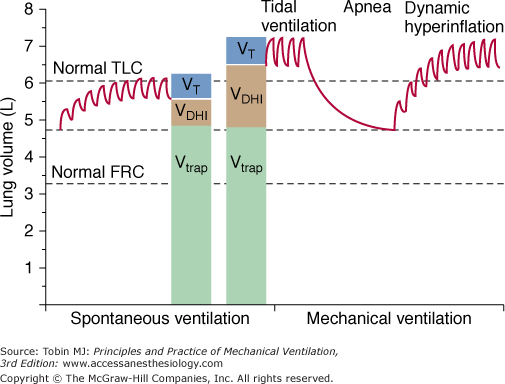
Patients with acute severe asthma breathe near their total lung capacity; with mechanical ventilation, lung volumes may increase further (Fig. 30-2).23 During mechanical ventilation, dynamic hyperinflation is initiated when there is insufficient time during expiration for complete exhalation of delivered tidal volume, resulting in an increase in end-expiratory lung volume and auto–positive end-expiratory alveolar pressure (auto-PEEP). With subsequent breaths, a progressive increase in lung volume leads to improved expiratory gas flow because of higher elastic recoil pressure and an increase in airways diameter, permitting the entire tidal volume to be exhaled (Fig. 30-3). Although dynamic hyperinflation may be an adaptive process that enhances expiratory flow, it can result in severe hypotension and barotrauma, life-threatening complications that are a consequence of extreme alveolar overdistension.
Figure 30-2
Lung volumes in severe asthma during spontaneous ventilation and with mechanical ventilation. Tidal breathing occurs near total lung capacity (TLC) during spontaneous ventilation and above TLC during mechanical ventilation. After delivery of tidal volume (VT) during mechanical ventilation, a prolonged apnea permits exhalation of volume produced by dynamic hyperinflation (VDHI); lung volume at the end of apnea, however, remains well above normal functional residual capacity (FRC) because of gas trapped behind occluded airways (Vtrap). (Adapted, with permission, from Tuxen et al.23)
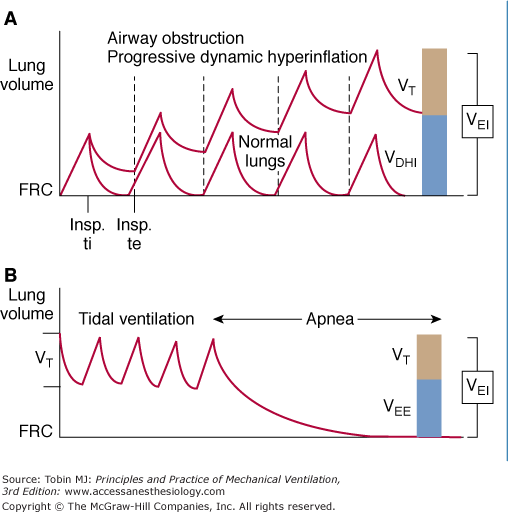
Figure 30-3
A. Dynamic hyperinflation. FRC, functional residual capacity; VDHI, volume (above FRC) caused by dynamic hyperinflation; VEI, volume (above FRC) at end inspiration; VT, tidal volume. (Adapted, with permission, from Tuxen. Am J Respir Crit Care Med. 1994;150:1722–1737.) B. Measurement of VEI and VEE (volume above FRC at end expiration) by use of a prolonged apnea. VEE (volume at end expiration) and VDHI (volume produced by dynamic hyperinflation) are equivalent. (Used, with permission, from Tuxen DV, Lane S. The effects of ventilatory pattern on hyperinflation, airway pressures, and circulation in mechanical ventilation of patients with severe air-flow obstruction. Am Rev Respir Dis. 1987;136:872–879.)
The severity of hyperinflation in severe asthma varies among patients and within individual patients over time. Serial monitoring of dynamic hyperinflation is important for identifying an increased risk of hypotension and barotrauma and to assess changes in the severity of airflow obstruction in response to therapy.
Two methods of monitoring the severity of airflow obstruction in status asthmaticus have been used: measurement of total exhaled volume during a prolonged apnea beginning at end inspiration and assessment of airway pressure. Tuxen et al21–24 used a prolonged apnea to assess dynamic hyperinflation in patients with severe asthma, with the total amount of gas exhaled during the apnea defined as the volume (above functional residual capacity) at end inspiration (VEI). The volume at end expiration (VEE) represents the increase in lung volume caused by dynamic hyperinflation and is calculated by subtracting tidal volume from VEI (see Figs. 30-3 and 30-4). In one study, hypotension and barotrauma were more reliably predicted by VEI than by airway pressures.24 Nonetheless, measurement of VEI and VEE has not gained widespread clinical acceptance, in part because of the need for paralysis.23
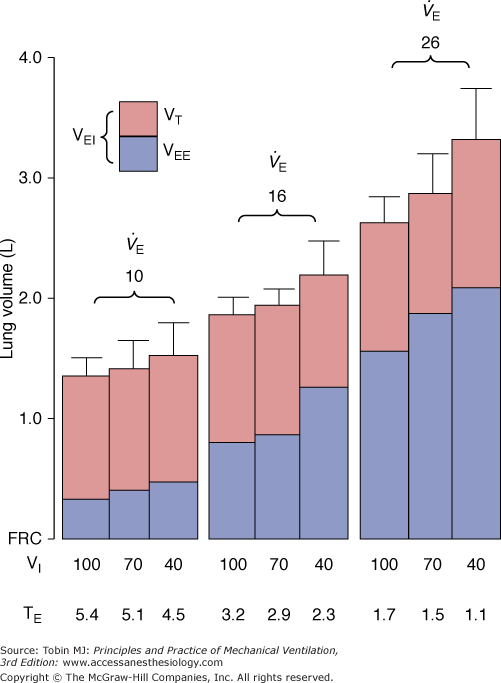
Figure 30-4
Effect of minute ventilation ( ) and inspiratory flow rate (VI) on dynamic hyperinflation during mechanical ventilation for severe asthma. VEE, volume at end expiration; VEI, volume at end inspiration; VT, tidal volume. (Used, with permission, from Tuxen DV, Lane S. The effects of ventilatory pattern on hyperinflation, airway pressures, and circulation in mechanical ventilation of patients with severe air-flow obstruction. Am Rev Respir Dis. 1987;136:872–879.)
) and inspiratory flow rate (VI) on dynamic hyperinflation during mechanical ventilation for severe asthma. VEE, volume at end expiration; VEI, volume at end inspiration; VT, tidal volume. (Used, with permission, from Tuxen DV, Lane S. The effects of ventilatory pattern on hyperinflation, airway pressures, and circulation in mechanical ventilation of patients with severe air-flow obstruction. Am Rev Respir Dis. 1987;136:872–879.)
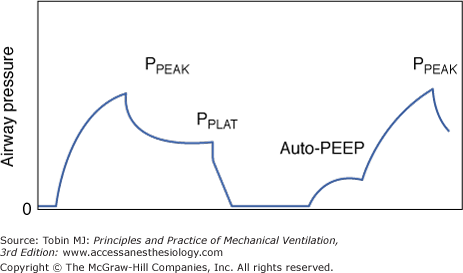
A more commonly used monitoring method is assessment of airway pressures (Fig. 30-5). As described initially,18 controlled hypoventilation in status asthmaticus targeted a peak airway pressure (PPEAK) below 50 cm H2O, a strategy used by others subsequently.26 PPEAK, however, is highly dependent on inspiratory flow-resistive properties and may not accurately reflect the degree of hyperinflation.23,27 Patients with asthma often have a PPEAK above 50 cm H2O when managed with high inspiratory flow rate (see below), but their risk of barotrauma may be negligible.27 Furthermore, PPEAK may not consistently reflect the reduction in dynamic hyperinflation after expiratory time is prolonged, presumably because of an increase in airways resistance as lung volume decreases (Fig. 30-6).25
Figure 30-5
Proximal airway pressure during an end-inspiratory and end-expiratory airway occlusion. End-inspiratory occlusion is followed by an initial rapid fall in airway pressure, secondary to intrinsic airways resistance and then a more gradual fall in pressure secondary to gas redistribution and tissue resistance. PPEAK, peak airway pressure; PPLAT, plateau airway pressure; auto-PEEP, auto–positive end-expiratory pressure.
Figure 30-6
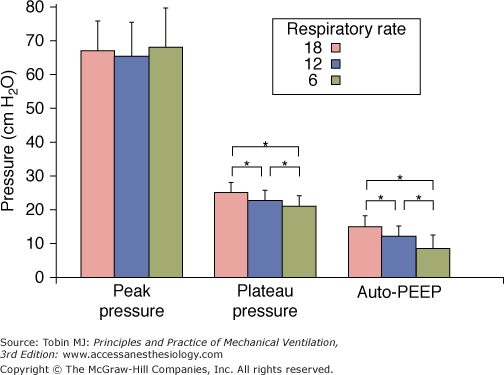
Peak airway pressure, plateau pressure, and auto-PEEP at respiratory rates of 18, 12, and 6 breaths/min during mechanical ventilation for severe asthma (n = 12). (Used, with permission, from Leatherman JW, McArthur C, Shapiro RS. Effect of prolongation of expiratory time on dynamic hyperinflation in mechanically ventilated patients with severe asthma. Crit Care Med. 2004;32:1542–1545.)
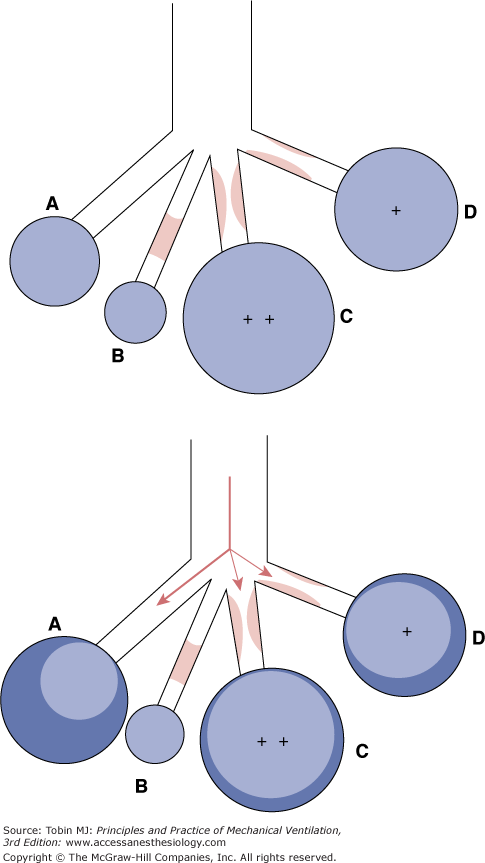
Plateau airway pressure (PPLAT) may be a better parameter for monitoring lung hyperinflation in status asthmaticus (see Fig. 30-5).23,27–31 Patients with severe airflow obstruction typically have near-normal respiratory system compliance and an increase in PPLAT results from dynamic hyperinflation. At constant tidal volume, changes in the degree of dynamic hyperinflation in response to bronchodilators or manipulation of expiratory time can be inferred from changes in PPLAT.25 Because PPLAT provides an estimate of maximal alveolar pressure, it might (in theory) help to predict risk of alveolar rupture. It should be noted, however, that PPLAT represents the average end-inspiratory alveolar pressure and the maximal alveolar pressure in some units will be higher (Fig. 30-7).31 The safe threshold for PPLAT is not well defined in status asthmaticus, but an acceptable upper limit of 25 to 30 cm H2O has been suggested most often (Table 30-2).23,27,30,31
Figure 30-7
Top. Effect of varying degrees of airway obstruction on end-expiratory alveolar volumes and pressures. Bottom. Expected distribution of the tidal volume on application of pressure during mechanical ventilation in the setting of inhomogeneous obstruction (With kind permission from Springer Science and Business Media: Oddo et al. Management of mechanical ventilation in acute severe asthma: practical aspects. Intensive Care Med. 2006;27:1–10.)
| Ventilator settings (initial) | |
| Mode | Assist-control |
| VT | 7 to 9 mL/kg |
| Rate | 10 to 14 breaths/min |
| VI | 60 to 70 L/min |
| Waveform | Decelerating or square |
| PEEP | ≤5 cm H2O |
 | SaO2 > 90% |
| Sedation/paralysis | |
| Propofol | 2 to 5 mg/kg/h infusion |
| Fentanyl | 50 to 200 μg/h infusion |
| Vecuronium | 0.1 mg/kg bolus PRN |
| Therapy of airflow obstruction | |
| Albuterol-ipratropium MDI | 6 puffs qh × 4, then q1-2h |
| Methylprednisolone | 2 mg/kg/day |
| Ventilator adjustments | |
Goals: PPLAT < 30 cm H2O (≤25 ideal) and pH ≥ 7.2
| |
When measured by end-expiratory airway occlusion, auto-PEEP provides an estimate of average end-expiratory alveolar pressure. The expected levels of auto-PEEP in status asthmaticus have been infrequently reported, but two studies found average values of 15 and 10 cm H2O, respectively.25,32 In general, changes in auto-PEEP track closely with changes in PPLAT, and either (or both) may be helpful for following the degree of dynamic hyperinflation. On occasion, patients with status asthmaticus who have radiographic evidence of hyperinflation and elevated PPLAT may have unexpectedly low values of measured auto-PEEP when ventilated at very low respiratory rates that encourage airway closure, preventing accurate assessment of end-expiratory alveolar pressure.33 Auto-PEEP should always be measured during passive expiration, because forceful exhalation often leads to gross overestimation of auto-PEEP and the severity of dynamic hyperinflation, potentially leading to unnecessary restriction of minute ventilation.34
In severe asthma, three key factors determine the degree to which VEE increases during mechanical ventilation: expiratory resistance, tidal volume, and expiratory time. The ventilator settings that most influence the severity of hyperinflation are tidal volume, respiratory rate, and inspiratory flow rate.
Tuxen and Lane21 examined various ventilator settings in severe asthma and found that minute ventilation was the most important determinant of dynamic hyperinflation (see Fig. 30-4). For a given minute ventilation, the extent of dynamic hyperinflation was similar regardless of the specific combination of respiratory rate and tidal volume.21 The degree of hyperinflation became quite marked when minute ventilation was increased from 10 to 16 and 26 L/min; at the highest minute ventilation, both hypotension and barotrauma were noted.21
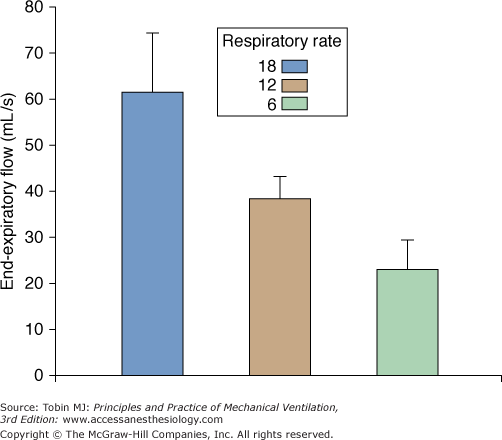
Although these very high levels of minute ventilation clearly pose a risk in the ventilated patient with asthma, the benefit of extreme limitation of minute ventilation, as advocated by some authors,35 is less certain. One study examined the effect of prolongation of expiratory time on dynamic hyperinflation—as assessed by PPLAT and auto-PEEP—when the baseline minute ventilation approximated 10 L/min.25 As expected, PPLAT and auto-PEEP decreased when expiratory time was prolonged, but the magnitude of the reduction was not profound (see Fig. 30-6). For example, when respiratory rate was reduced from 12 to 6 breaths/min (adding 5 seconds to expiratory time), PPLAT and auto-PEEP fell by only 2 to 3 cm H2O.25 The relatively modest effect on dynamic hyperinflation is understandable given the low expiratory flow rates after a few seconds of expiration (Fig. 30-8). Because flow decreases progressively throughout expiration, the reduction in hyperinflation that results from a given prolongation of expiratory time will depend on the baseline respiratory rate (i.e., less benefit at lower respiratory rates) (Fig. 30-8).
Figure 30-8
End-expiratory flow rates at respiratory rates of 18, 12, and 6 breaths/min during mechanical ventilation for severe asthma (n = 7). (Used, with permission, from Leatherman JW, McArthur C, Shapiro RS. Effect of prolongation of expiratory time on dynamic hyperinflation in mechanically ventilated patients with severe asthma. Crit Care Med. 2004;32:1542–1545.)
Another factor that may limit the degree to which prolongation of expiratory time lessens lung hyperinflation is the presence of gas trapped behind occluded airways. Total pulmonary hyperinflation has two components: a dynamic component that is amenable to ventilator manipulation and a second component caused by gas trapped behind occluded airways, with the latter being unaffected by expiratory time (see Fig. 30-2). One study suggested that trapped gas accounted for a greater percentage of total pulmonary hyperinflation than did dynamic hyperinflation.22
In essence, there is often little to be gained by reducing the respiratory rate below 10 to 14 breaths/min when a tidal volume of 7 to 9 mL/kg is used (see Table 30-2). An exception might be when hyperinflation is marked (e.g., PPLAT >30 cm H2O) or has resulted in complications, because even a small reduction in hyperinflation might have a meaningful clinical impact. Unfortunately, dramatic reductions in hyperinflation may not be easily achieved by ventilator manipulation alone and must await improvement in airflow obstruction.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree