Fig. 40.1
Sagittal MRI STIR lumbar spine demonstrating decreased height and biconcave deformity of the L2 vertebral body (personal library)
The patient described the pain as a 10/10 at its worst, typically after 10–15 min of exercise. He experienced tenderness to palpation over the upper lumbar spinous processes. He also described radicular pain over the anterior thigh bilaterally, correlating with coexisting multilevel degenerative disk disease, worse at L3–L4, and mild disk protrusion at L5–S1, consistent with MRI findings. Physical therapy provided no relief of pain. A lumbar epidural steroid injection was performed 1 month after his first visit. It had no effect on his back pain but decreased his radicular pain to a 3–4/10 for approximately 2 weeks. A TENS unit and back brace brought minor back pain relief. The patient’s exercise regimen was limited because of continuous back pain, and he perceived his overall physical function as rapidly deteriorating.
With weight gain, the axial pain increased. Long-acting therapies including OxyContin and a fentanyl patch were discontinued because of nausea and constipation.
Given the characteristics of his pain and physical examination, we thought that the patient had an unhealed fracture, not radiographically visible at the L2 level, in the fibrous material. Multidisciplinary and contradictory discussions with the patient’s endocrinologist were conducted with disagreement between the pain physician and endocrinologist on the medical necessity of the performing kyphoplasty. Given the patient’s rapid deterioration in functional status and after carefully weighing the risks and benefits, kyphoplasty was eventually seen as a valid option. The patient underwent balloon kyphoplasty of the L2 vertebral body. Under monitored anesthesia care and after thorough vertebral body periosteal infiltration with an 18-gauge spinal needle, the kyphoplasty trocar was advanced under fluoroscopic guidance within the L2 vertebral body. The placement of the trocars and the balloons was limited to the lateral aspect of the vertebral body, given the near-complete apposition of the superior and inferior end plates in the mid third of the vertebral body. The balloon was inserted and inflated within the L2 vertebral body under low pressure. It was then deflated and methylmethacrylate was injected within the vertebral body. The distribution of cement within the L2 vertebral body followed the concavity seen in the MR image.
This technique was used to avoid disruption of the cortex after inflating the balloon and depositing methylmethacrylate. Posterior wall entry was strong, but there was no need for mallotting while passing the 2 mm posterior wall, suggesting replacement of osseous material with fibrous material consistent with known fibrous dysplasia (Fig. 40.2).
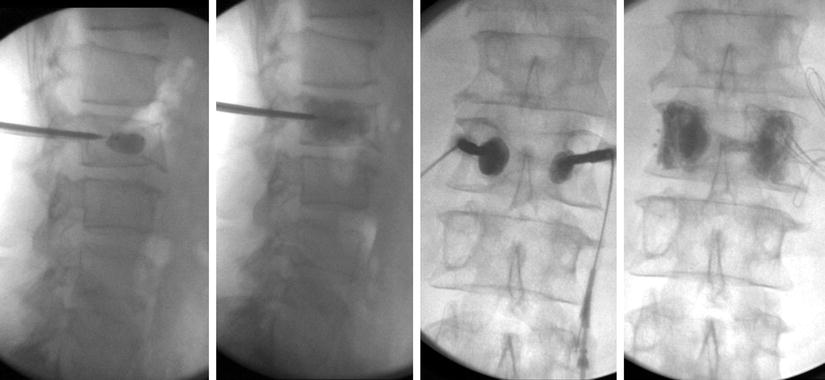

Fig. 40.2
Kyphoplasty at L2 demonstrating distribution of cement within the dysplastic lesion (personal library)
The patient experienced immediate and near-complete resolution of his acute and chronic pain. He was able to walk without pain in the recovery room, and 1 week after the procedure, he resumed an aggressive exercise regimen including 300 sit-ups, jogging several miles, and kickboxing three to four times per week. He lost weight, his functional status improved rapidly, and his medical comorbidities were alleviated. With weight loss, obstructive sleep apnea improved dramatically that he no longer required nocturnal CPAP. He continued to complain of a radicular pain in the left lower extremity from a disk protrusion at L5/S1, but this was mild overall and responded well to repeat steroid injections.
40.2 Discussion: The Syndrome of Axial Fibrous Dysplasia
40.2.1 Etiology and Pathogenesis
First described as a benign intramedullary fibro-osseous lesion by Lichtenstein and Jaffe [1, 2], fibrous dysplasia (FD) is currently defined as a genetic, non-inherited disease that affects men and women equally [3]. In this benign process, the normal bone is replaced with fibrous connective tissue. The result is ineffective remodeling of poorly mineralized, immature trabecular bone, widening of the affected bones, and subsequent cortical bone thinning. These changes cause a loss in mechanical strength and increased pain and fracture [4].
The process takes place in the medullary marrow cavity and is thought to result from a mutation of the guanine nucleotide stimulatory protein (GNAS1) gene, which encodes the alpha subunit of the stimulatory G protein (G1) located on chromosome 20q13.2–13.3.
This protein is responsible for stimulating guanine nucleotide-binding protein Gsα, leading to high levels of cyclic adenosine monophosphate and, in turn, overexpression of the c-fos proto-oncogene, which regulates expression and differentiation of osteoblasts and osteoclasts [5, 6].
The cell mutation seems to take place after fertilization in somatic cells; as a consequence all affected cells carry the same dysplastic feature from that moment on. Depending on where the affected cells are located during embryogenesis and numbers of cells affected, the clinical presentation of the disease is variable. The disease can manifest at any age, but the majority of lesions are detected around the third or fourth decade of life. Following the original genetic mutation, the primitive bone fails to remodel into mature trabecular lamellae. The resultant immature trabeculae enmeshed in dysplastic fibrous tissue, although turning over constantly, never achieve the complete remodeling process of mature bone that offers stability to the skeleton. The immature matrix also lacks effective and normal mineralization. As a consequence, the combination of insufficient mineralization coupled with lack of stress alignment from immature trabeculae results in significant loss of mechanical strength, subsequent pain, and pathological fractures [4]. The Office of Rare Diseases of the National Institutes of Health lists FD as a rare disease, meaning that fewer than 200,000 people in the USA are affected. The true prevalence is difficult to determine given the rarity of the disease and the likelihood of people being affected without experiencing symptoms. It is estimated that 1:15,000–1:30,000 Americans are affected with a lifetime incidence of 1:9000–1:18,000 [7].
FD comprises roughly 7% of all nonmalignant bone tumors and 2.5% of all bone lesions [8]. There are two forms: monostotic and polyostotic. The appendicular and axial skeletons have been implicated in each form [4] (Tables 40.1 and 40.2).
Table 40.1
Types of fibrous dysplasia
Characteristics | |
|---|---|
Monostotic | • The replacement of normal bone with fibrous connective tissue, causing a loss in mechanical strength and increased pain and fracture • Most prevalent form • Involves a single bone, may include multiple lesions on one bone • Rarely, can involve the spine—only 2% of monostotic FD cases • Lesions evolve in parallel with skeletal growth. Rarely progress past adolescence • Most often found incidentally |
Polyostotic | • Identical pathophysiology • Less prevalent form • Involves two or more bones • Higher incidence of spinal involvement than the monostotic form • Lesions continue to grow after bone maturity, leading to deformity and fracture |
Bone involvement | % of FD cases | Spinal involvement | Associated pathology | |
|---|---|---|---|---|
Monostotic | Single | 70 | Yes | |
Polyostotic | Multiple | 30 | Yes | |
McCune-Albright syndrome | Single (monostotic) or multiple (polyostotic) | 3 | Yes | Precocious puberty, café au lait skin pigmentation ± hyperthyroidism, acromegaly [10] |
Mazabraud syndrome | Single (monostotic) or multiple (polyostotic) | Unknown (80 total cases of Mazabraud syndrome described to date) | Yes | Soft tissue myxomas |
Of patients with FD, 3% have McCune-Albright syndrome [9] (a triad of precocious puberty, café au lait skin pigmentation, and fibrous dysplasia of the bone). FD also exists with Mazabraud syndrome (fibrous dysplasia with soft tissue myxomas), 80 cases of which have been described to date [10]. Mazabraud syndrome is seen in 70% of females, and the onset of fibrous dysplasia typically precedes myxomas.
The two main forms of FD differ by the location of bone involvement. The monostotic form (70% of cases) involves a single bone; the polyostotic form (30% of cases) involves two or more bones [9]. The appendicular skeleton is much more commonly affected, with only a few case reports involving the axial skeleton. In one survey of monostotic FD, the skull was affected in 20% of cases, the lower limbs in 34%, upper limbs in 10%, and the axial skeleton in just 2% [11]. Malignant transformation is rare, occurring less than 5% of the time [4].
The exact incidence and prevalence of spinal pathology in FD are unknown. In two studies of more than 100 FD patients, evidence suggests that spinal pathology is almost exclusively seen in the polyostotic form [12–14]. In 62 patients with polyostotic FD, 39 (63%) had 76 lesions in the spine, the overwhelming majority lumbar or thoracic [12, 13]. Of 56 patients with FD, the spine was affected in 6 of 24 patients with McCune-Albright syndrome. In polyostotic patients, spinal involvement was noted in 7 of 33 (21.2%) patients [13]. FD in the spine was also significantly more frequent in patients with FD in the skull or pelvis (Table 40.3).
40.2.2 Clinical Manifestations
In the monostotic type, the dysplastic lesions are incidental findings, with diagnosis made usually when radiographs are performed as part of an examination of the region affected [4]. When symptomatic, the monostotic lesions seem to enlarge parallel to skeletal growth [15].
The rarer polyostotic form has a different clinical presentation and natural history. Severe deformities by late adolescence accompany this form. Those lesions tend to enlarge even after skeletal maturity is achieved and commonly progress to fractures [4].
When symptomatic, by age 30 fibrous dysplasia causes localized bone pain and deformity frequently related to fatigue or stress fractures. Diagnosis is made by radiographic findings in typical FD image. Otherwise, biopsy is indicated [4]. FD is found in long bones of the legs, arms, pelvis, and craniofacial bones, with spinal involvement estimated in 1.4%–5.5% of lesions [16]. Sole spinal involvement is rare. Differential diagnosis includes simple bone cyst, fibroma, metastasis, osteoblastoma, multiple myeloma, chronic infectious spondylitis, hemangioma, giant cell tumor, and Paget’s disease [17]. Upon diagnosis, osteomalacia from vitamin D deficiency, hypophosphatemia, hypothyroidism, Cushing’s, and hyperparathyroidism must also be ruled out, given bone turnover with FD.
Localized pain is often the first symptom of FD, especially if the disease affects the femoral neck. In addition to pain, patients may also experience limping. Women affected may have increased pain during pregnancy. Pain during the menstrual cycle is also possible because of more estrogen receptors in the fibrous dysplastic bone [4].
Bone deformity is also common. Deformity depends on the site of the lesion, age of the patient, severity of disease, and the type of FD. In the appendicular skeleton, the classical deformity is at the level of the proximal femur. The pathognomonic, so-called shepherd crook that appears in the polyostotic form, occurs as a result of an abnormal remodeling process in the proximal femur that continues even after the skeleton reaches maturity. Large weight-bearing bones such as the femur appear as bowing deformities and are accompanied by a shortened ipsilateral leg and widened hip region as a result of fatigued fractures of the dysplastic bone. Sudden pathological fractures and pain result from underlying swelling and deformity. In addition to limb length discrepancies and pain, approximately 50% of patients with monostotic disease have pathological fractures (most commonly of the femur).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






