Chapter 18
Head Injury
Jameel Ali, Lyne Noël de Tilly and R. Loch Macdonald
Chapter Overview
Head injuries account for a large percentage of worldwide trauma related mortality and morbidity. In North America over 90% of pre-hospital trauma deaths have an associated head injury and of those head injuries which are admitted to hospital, roughly 75% sustain mild injuries, 15% moderate, and 10% severe head trauma.1,2 The outcome in patients with mild injuries is generally good but mortality and morbidity increase progressively in the moderate and severe categories.1 Head injury is among the leading causes of death worldwide and is the leading cause of death among adults under 45-years in the United States. Thus, an important impact on survival could be achieved by aggressive management of moderate and severe head injuries. Ideally, such care is frequently provided in the intensive care environment. Proper management of these injuries requires a clear understanding of the unique anatomical and physiologic features of the brain as they are affected by trauma. Generally, medical/surgical intervention is not effective in reversing the primary tissue damage resulting from the direct brain trauma itself. The main focus of management is therefore prevention of secondary brain injury.
In this chapter, we will discuss the unique features of intracranial anatomy and physiology, how they affect the presentation of head injuries and how they may be manipulated to preserve brain function in the head injured patient. Classifications and investigation of head injuries, their clinical presentation as well as principles of medical and surgical management will also be discussed.
Anatomic Considerations
The brain is confined within a fixed space by a rigid non-yielding bony skull. Addition of mass to the intracranial contents as would occur from hemorrhage in trauma will eventually lead to increased intracranial pressure (ICP), which will reduce cerebral blood flow and affect brain function. The unyielding rigid surface of the skull also predisposes to direct (coup) as well as contra coup tissue injury on movement of intracranial contents resulting from force applied to the skull and shearing of brain tissue.
To understand intracranial changes following trauma, the intracranial contents may be considered to consist of brain, cerebrospinal fluid (CSF) and blood. The meninges which cover the brain consist of three layers; the duramater, arachnoid mater and pia mater
The dura mater is a two-layered tough fibrous layer of tissue closely applied to the inner surface of the skull. The potential space between the skull and dura (epidural or extradural space) houses the meningeal arteries and veins. Skull fractures can lacerate these vessels leading to rapidly fatal epidural hematomas. This commonly occurs, secondary to a temporal bone skull fracture overlying the middle meningeal artery. At specific sites the dura splits into two leaves to accommodate the venous sinuses. Massive hemorrhage can occur from injury involving these sinuses.
The arachnoid mater is a thin layer of translucent tissue deep to the dura. It is only loosely adherent to the dura by dural border cells which results in a potential space (the subdural space) between the dura and arachnoid within which hemorrhage from bridging veins can occur (acute or chronic subdural hematoma). Cerebral atrophy in elderly patients causes the brain to recede from the skull which increases the potential for a subdural space to develop and be filled with blood from veins bridging from the brain to the dura after minor trauma. Bridging veins are believed to tear at the point where they traverse the potential subdural space because their walls are thinnest in this segment. The arachnoid membrane encases the brain but does not by and large enter the sulci. The arachnoid is a water tight barrier between it and the third meningeal layer, the pia mater.
The pia mater is tightly attached to the entire brain and spinal cord surface. The space between these two (subarachnoid space) contains CSF which is formed in party by the choroid plexus in the cerebral ventricles at the rate of approximately 20 ml per hour. The CSF circulates over the entire brain and spinal cord surface before entering the systemic circulation through the arachnoid granulations. Bleeding into the subarachnoid space (subarachnoid hemorrhage) is frequently seen with brain contusions or major vessel injuries. When such bleeding obstructs the arachnoid granulations CSF flow can be impeded, giving rise to CSF accumulation in the ventricular system or traumatic hydrocephalus. This adds mass to the cranial cavity and can lead to increased ICP.
The tentorium cerebelli separates the brain into two main compartments — one above the tentorium or supratentorial compartment and the other below or the infratentorial compartment. The midbrain passes through an aperture in the tentorium, the incisura or hiatus which allows continuation of the supraratentorial with the infratentorial compartment. The oculomotor nerve with parasympathetic fibers on its surface courses along the edge of the tentorium.
If a supratentorial hematoma or hydrocephalus displaces the brain medially and inferiorly through the tentorialincisura, transtentorial herniation develops. There is a classic sequence of events starting with decreased level of consciousness due to bi-hemispheric cortical dysfunction from the brain shift. It is important to realize that ICP may not be increased early on. The parasympathetic fibers of the oculomotor nerve, which are responsible for pupillary constriction, are compressed leading to ipsilateral pupillary dilation. The motor fibers of the cortico spinal tract in the adjacent cerebral peduncle of the midbrain are also compressed giving rise to contralateral motor paralysis. Paralysis is contralateral because these fibers decussate lower in the medulla. Next there is occlusion of the cerebral aqueduct which obstructs CSF outflow and further aggravates the now increased intracranial pressure and compression of the posterior cerebral artery resulting in infarction of this vascular territory. Progressive rostral-caudal deterioration ensues with dilation of the other pupil, midbrain Duret hemorrhages, loss of corneal reflexes, lower brainstem reflexes and eventually brain death. The critical early signs from an intracranial mass lesion such as traumatic hemorrhage are therefore ipsilateral pupillary dilation and contralateral motor paralysis. In about 10% of cases, the hemiparesis may be on the same side due to the Kernohan notch syndrome.3
Another herniation syndrome from lateral displacement of the brain by a unilateral hematoma is subfalcine herniation where the cingulate gyrus and associated anterior cerebral arteries are pushed across under the falx cerebri.
Physiologic Considerations
The interplay of cerebral blood flow and metabolism, ICP, systemic hemodynamics, and arterial blood–gas changes is crucial in determining the patient’s response to head trauma and understanding the basic concepts involved provides a solid guide to therapeutic interventions. The brain consumes 25% of oxygen used by the body and is highly dependent on a continuous supply of oxygen and glucose-rich blood. Cerebral perfusion pressure (CPP) (mean blood pressure minus ICP) ensures this. As in any fixed rigid container such as the skull, the internal pressure is determined by the volume of its contents. The relevant contents under consideration are the brain, blood, and CSF; the pressure is the Intracranial Pressure (ICP). Normal ICP is approximately 10 mmHg and usually lower. While there is some controversy as to whether increased ICP after head injury is a cause of poor outcome or a consequence of severe irreversible injury, sustained ICP above 20 mmHg is associated with poor outcome and would be expected to reduce CPP and cause brain injury.
The pathophysiology of increased ICP and reduced CPP after Traumatic Brain Injury (TBI) includes brain swelling and cerebral edema due to the injury that reduces cerebral blood flow, causes ischemia and then further brain swelling, and edema. There are two approaches for treatment based on this. The most common is ICP- or CPP-based. The ICP based approach is to prevent and reduce increased intracranial pressure by elevating the head of the bed (with spine precaution), sedation, pharmacologic paralysis, treatment of hypertension, CSF drainage, osmotic diuretics, and occasionally barbiturates.4 A variation of this focuses on maintaining adequate CPP and uses flat position in bed, no sedation, hyperventilation or barbiturates, and elevating the blood pressure to maintain CPP if necessary. At the opposite end of the spectrum is the Lund concept, where the goal is to reduce cerebral edema by flat head position, sedation, aggressive blood pressure reduction, no pharmacologic paralysis and no hyperventilation, osmotic diuretics, or barbiturates.4
Autoregulation of cerebral blood flow: The cerebral vasculature dilates or constricts in response to variations in mean systemic arterial blood pressure, maintaining stable cerebral blood flow over a range of mean systemic arterial blood pressures from about 50–150 mmHg. Cerebral blood flow cannot easily be measured in the clinical setting except intermittently by techniques such as computed tomographic or magnetic resonance perfusion, xenon Computed Tomography (CT) or positron emission tomography or focally, and continuously by implanted thermodilution probes. Furthermore, the adequacy of cerebral blood flow depends on metabolic demand of the brain. Thus, in practice, CPP tends to be used as a surrogate for cerebral blood flow recognizing that the two are not linearly related. Autoregulation may be disrupted in severe brain injury.5 Other indirect methods of monitoring brain perfusion are discussed in the following paragraphs.
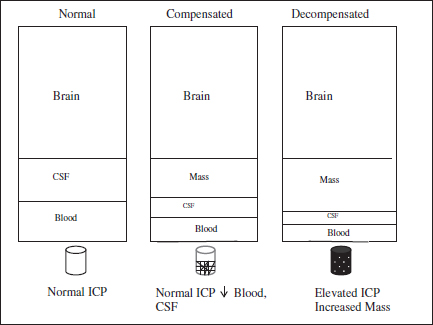
The Monro–Kellie doctrine depicts the relationship between ICP and volume (Fig. 1). Under normal conditions the ICP is low with normal intracranial volumes of blood, CSF, and brain tissue.6 The total intracranial volume is constant. If there is an increase in the volume of one component there initially is a compensatory decrease in the other volumes resulting in a homeostatic mechanism directed at maintaining normal ICP. With addition of a mass such as an intracranial hematoma, the compensatory mechanisms include decreased blood and CSF volume. Eventually, compensatory mechanisms are exhausted and ICP rises. It follows that to decrease ICP in patients with an intracranial hematoma:
Fig. 1. Monro–Kellie Doctrine.
2. CSF volume could be decreased by drainage from a ventricular catheter.
3. Brain mass could be diminished by agents such as mannitol or hypertonic saline.
4. The intracranial mass could be evacuated.
Each of these would decrease the overall intracranial volume to accommodate the added intracranial mass and maintain normal ICP and cerebral perfusion and function. However, when the added intracranial mass enlarges further or the effects of the interventions and compensatory mechanisms are exhausted there is a precipitous rise in ICP that leads to transtentorial herniation with increased ICP and death of the patient. The key to management is to intervene before this terminal event.
Another response to head injury is the Cushing reflex whereby an increase in ICP is associated with bradycardia. There is usually an increase in mean systemic arterial pressure which is less consistent than the bradycardia and which may be a compensatory mechanism to preserve cerebral perfusion pressure. In general, it should be recognized that this reflex is a compensatory response and attempts to lower the blood pressure should be avoided because this would lower the cerebral perfusion pressure. The best treatment for this systemic hypertension is to reduce the increased ICP. The combination of increased ICP from a head injury and systemic hypotension from inadequate resuscitation plus inadequate oxygenation is a well recognized situation associated with poor prognosis and can prove lethal for these patients. This is the basis for focusing on prevention of secondary brain injury by maintaining adequate perfusion and oxygenation.
Classification of Head Injuries
There are many methods of classifying head injuries. The first distinction is between primary and secondary injury. The primary injury can be classified based on mechanism. Primary injuries are shearing injuries (a spectrum from concussion on the mild end to diffuse axonal injury on the severe end) and each of the types of bleeding described below. Head injury also can be categorized by morphology and severity.
Classification based on mechanism
Primary injuries
As in other anatomic areas brain injuries may be classified into direct impact, acceleration–deceleration, blast and penetrating.
DIRECT IMPACT INJURY includes scalp laceration and contusion, skull fracture, epidural hematoma, and brain contusion. Scalp lesions and skull fractures are pathognomonic of something hitting the head or the head hitting something. Acceleration–deceleration injuries could arise from direct impact but the head does not have to hit anything to undergo this, for example, in a restrained occupant of a motor vehicle crash. The fundamental damaging process is rotational or angular acceleration–deceleration about the center of rotation of the head that causes shearing of axons and blood vessels. The role of contact of the intracranial contents against the irregular inner surface of the skull is likely less important. Shearing of blood vessels leads to contusions. Contusions are:
1. Coup contusions under the site of impact.
2. Contra coup, usually in the basal frontal lobes and anterior temporal lobes, since they are farthest from the brain center of rotation and experience the greatest shearing.
3. Gliding contusions in the superior parts of the hemispheres.
4. Contusions in the corpus callosum and superior cerebellar peduncle.
SHEARING MECHANISMS occur on a continuum from mild, causing transient loss of consciousness, to severe which is called diffuse axonal injury and is characterized by white matter lesions in the corpus callosum and brainstem, petechial contusions, axon retraction balls, coma, usually no increased ICP, and often poor outcome.
PENETRATING INJURIES arise from gunshots, knife or other penetrating missiles. Low velocity penetrating injuries such as from knives and such, will not cause shearing injury but will only cause injury by direct disruption of brain tissue or by hemorrhage from damaged blood vessels. Gunshots and shrapnel, however, carry kinetic energy that is dissipated in the brain. Kinetic energy is equal to ½ of the mass of the projectile multiplied by the velocity squared. Thus, velocity is more important than mass. Although cranial gunshot wounds are highly mortal overall, the dependency on velocity explains why high velocity military guns are even more lethal than civilian hand guns. Penetrating bullet injuries cause brain damage along the tract of the bullet plus by producing cavitation and shock waves that propagate away from the bullet trajectory and through the brain and further by hemorrhage from damaged blood vessels.
BLAST INJURIES are due to pressure waves from explosions.7 The pressure waves propagate through the head and body tissues, each of which has a different acoustic impedance. Mechanical disruption of tissues occurs at the interfaces between tissues with different impedances. In addition, the pressure waves cause sudden changes in ICP that cause cavitations in the brain damaging axons and blood vessels.
Classification Based on Morphology
1. The tissue plane of injury: Scalp lacerations, contusions and avulsions — these are obvious on examination and indicate the head hit something or something hit the head.
2. Skull fractures: They occur in 10% of severe head injuries and are classified as closed or open: linear, depressed, diastatic, or basal.
Closed, open and linear fractures are self-explanatory.
Depressed fractures mean the inner table of the skull is deeper inside the head than normal. Infants can have ‘ping–pong’ depressed fractures because their skulls are thin and soft, resulting in indentation as would occur when a ping–pong ball is pressed.
Diastatic fractures are traumatic separation of the skull along a suture line, usually in children and young adults because the sutures have not fused yet.
Basal skull fractures are most commonly through the temporal bone or the basal frontal bones. They may be difficult or almost impossible to image even on CT scan and diagnosis may rely on clinical features. Basal temporal bone fractures are characterized by retroauricular ecchymosis (Battle sign), hemotympanum, otorrhea, and facial or vestibulocochlear cranial nerve palsies. Anterior frontal fossa basal fractures may cause raccoon eyes and rhinorrhea. Basal skull fractures may traverse the carotid canal giving rise to carotid artery injuries which would require CT angiography or standard contrast angiography for diagnosis. Fractures also increase the likelihood of intracranial hematomas.
INTRACRANIAL LESIONS — may be diffuse, localized or both.
Diffuse lesions — these vary from concussion in which there is a transient loss of brain function as detected by altered mental status or transient loss of consciousness, normal CT scan and no major sequelae to diffuse axon injury characterized by coma, diffuse shearing injury to the brain, and poor prognosis.
Localized lesions are named according to the location of the hematoma — epidural hematoma, subdural hematoma, subarachnoid hemorrhage, contusions, and intracerebral hemorrhage.
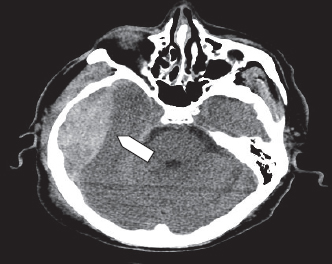
1. Epidural hematoma — On axial CT imaging these are biconvex hyper dense hematomas adjacent to the inner table of the skull, and if sizable, associated with compression and shift of the underlying brain (Fig. 2). Since the dura is more firmly adherent to the skull at the cranial sutures, they usually do not (although they can) extend across sutures. Epidural hematomas are almost always acute complications of head injury, only rarely do they increase in size days after the injury. They are uncommon and complicate less than 5% of head injuries.
Fig. 2. Right temporal epidural hematoma (arrow head) demonstrates a medial convexity.
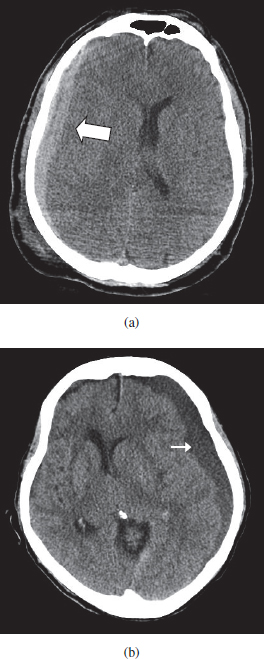
2. Subdural hematomas are more common than epidural hematomas and result from laceration by stretching and shearing forces of the bridging veins over the cerebral cortex. Acute subdural hematomas are hyperdense on CT scan and are diagnosed within 3 weeks of trauma. Chronic subdural hematomas are of variable density on CT scan and present more than 3 weeks after trauma or in patients who cannot recollect any traumatic event. Subdural hematomas are not confined by suture lines and on axial imaging have a concave inner margin (Figs. 3a and 3b). Acute subdural hematomas are common and occur in 10–20% of head injuries. They frequently occur in elderly patients with some degree of cerebral atrophy resulting in retraction of the brain from the skull thus stretching the bridging veins making them very vulnerable to minimal trauma. The resulting bleeding is not as rapid as in the epidural hematoma of arterial origin but in cases of acute subdural hematoma the prognosis is worse because of underlying brain injury. These patients also present with signs of increased ICP and require rapid evacuation of the hematoma in addition to other supportive measures which are discussed in the following paragraphs.
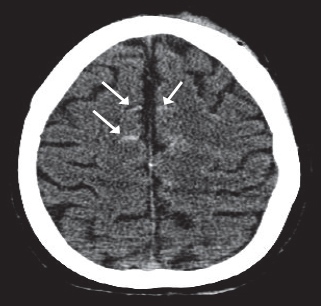
3. Subarachnoid hemorrhage — This is characterized by high density acute blood in the normally hypodense CSF-filled subarachnoid space. It may be focal on the surface of the brain over contusions, or more diffuse (Fig. 4). It occurs on CT scan in about 10% of head injuries although small amounts of subarachnoid bleeding can be detected in the CSF by lumbar puncture in a much higher percent of patients with head injuries. Traumatic subarachnoid hemorrhage is virtually always an acute complication of head injury.
Fig. 3. (a) Right Frontal parietal subdural hematoma (arrow) shows a crescentic configuration. (b) Large chronic left hemispheric subdural hematoma (arrow) causing compression of the left lateral ventricle and shift of the midline structures toward the right.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree