Fighting the Ventilator: Introduction
A patient is connected to the ventilator. His eyes are closed and he appears calm. The ventilator is making soft rhythmic noises, and the patient’s chest is expanding and receding in unison with the ventilator. A sweep of signals is gently traversing the monitor screen.
This peaceful scene erupts suddenly. The patient bolts upright. His eyelids retract. His nostrils flare. Sweat drips from his brow. His skin turns blue. His sternocleidomastoids contract vigorously. His rib spaces retract. One or more alarms sound loudly.
A patient fighting (or bucking) the ventilator is frightening not only to the patient but to staff. If the physician cannot find the source of the problem and fix it, the patient may die in minutes. The physician must immediately diagnose and manage the problem, and do both concurrently. The physician quickly scans the monitors for clues. Sometimes the problem is immediately spotted and solved, such as disconnected ventilator tubing.
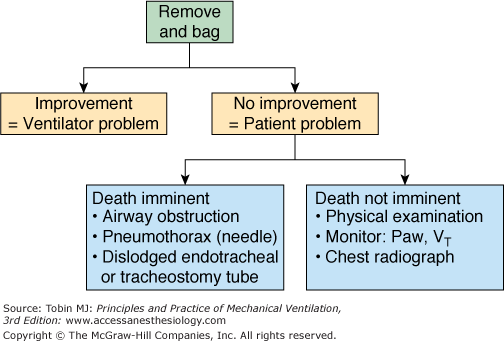
If the cause is not immediately obvious, the physician’s primary responsibility is to ensure adequate ventilation. This requirement takes precedence over diagnosis. After disconnecting the patient from the ventilator, the physician (or staff) starts to ventilate the patient manually with a self-inflating bag containing 100% oxygen. This step is both therapeutic and diagnostic (Fig. 53-1).1,2 If the distress resolves, it indicates that the problem originated in the ventilator. If the distress continues, it indicates the problem is within the patient.
Figure 53-1
Removing the patient from the ventilator and providing manual ventilation with a bag (100% oxygen) is both therapeutic and diagnostic. Patient improvement indicates that the ventilator is the cause of the distress. Lack of improvement indicates that the problem is within the patient. If death appears imminent, the physician rapidly checks for airway obstruction (by passing a suction catheter), a dislodged endotracheal or tracheostomy tube, and a pneumothorax (if deemed likely, a small-gauge needle is inserted); depending on the clinical picture, other life-threading problems, such as aortic dissection, should be considered. If death is not imminent, the physician undertakes a more detailed physical examination and assessment of monitored variables. A chest radiograph may also be obtained. Paw, airway pressure; VT, tidal volume.
Initial Assessment
Where a physician begins the assessment varies with the particulars of the patient’s presentation. The following sequence is not appropriate for all patients.
Because hypoxia can be rapidly lethal, the pulse oximeter reading is noted. Although several factors can give rise to erroneous readings, the displayed saturation generally bears a close relationship to the oxygen saturation on an arterial blood-gas test.3
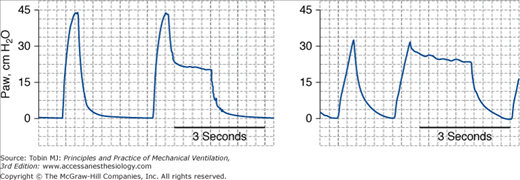
If the high-airway-pressure alarm is sounding, the physician should, if possible, perform an end-inspiratory occlusion maneuver to measure the plateau pressure (Fig. 53-2). An increase in peak airway pressure without a proportional increase in plateau pressure indicates narrowing of the airway.4 Narrowing may arise from bronchoconstriction, an increase in secretions, a collection of fluid in the inspiratory limb of the ventilator circuit, kinking of the tubing, a foreign body, or herniation of the cuff of the endotracheal tube over its distal tip.
Figure 53-2
End-inspiratory occlusion maneuver during passive ventilation in two patients. In each patient, the second displayed breath was occluded for 2 seconds or less—a period that was not sufficient to achieve a complete plateau in either patient. Left panel. Airway pressure (Paw) drops by 24 cm H2O during the end-inspiratory occlusion maneuver, indicating the presence of increased airflow resistance. Right panel. Paw drops by 5 cm H2O during the end-inspiratory occlusion maneuver, indicating decreased respiratory compliance consequent to noncardiogenic pulmonary edema secondary to sepsis.
An increase in peak airway pressure accompanied by a proportional increase in plateau pressure indicates a decrease in thoracic compliance, which may arise within the lung or from extrinsic compression. A decrease in lung compliance may arise from pulmonary edema, early stages of the acute respiratory distress syndrome, dynamic hyperinflation associated with intrinsic positive end-expiratory pressure (PEEPi), and atelectasis. Extrinsic compression may be caused by rapid collection of pleural fluid (hemothorax, parapneumonic effusion, or transudative effusion) or a pneumothorax. If a tension pneumothorax is suspected and death appears imminent, the physician should insert a 14- to 16-gauge needle into the second intercostal space. If a gush of air is heard, a thoracostomy tube is inserted. Gastric distension, ileus, ascites, or extensive eschar formation can also cause a decrease in thoracic compliance.
A major obstruction of the airway will commonly cause oxygen desaturation and an increase in peak airway pressure. If obstruction is suspected, the clinician should pass a suction catheter through the airway to determine if the airway is patent and to remove secretions or other material that is causing a blockage.
The alarm may signal a low exhaled tidal volume (VT). This can arise because of movement of the endotracheal tube into the hypopharynx or esophagus. It can also arise because of a disconnection in the ventilator circuit or a leak in the system. Many conditions can cause an increase in respiratory rate. Accordingly, a high rate is a very sensitive marker of an important change in a patient’s condition. Because it is influenced by so many factors, it is not helpful in directing the clinician to the cause of the new change.
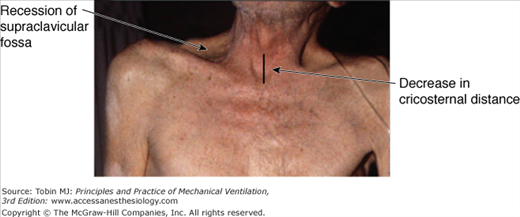
The patient’s pattern of breathing may signal a marked increase in work of breathing. Important signs include tracheal tug (how much the larynx descends with each inspiration); palpable activity of the sternocleidomastoid muscles; recession in the suprasternal notch, supraclavicular space, and intercostal spaces; and paradoxical movement of the rib cage and abdomen (Fig. 53-3).
Figure 53-3
Examination of the neck in a patient with acute respiratory distress. Inspection reveals recession of the supraclavicular fossa, recession of the suprasternal notch, increased tracheal tug (not visible on the photograph), and prominent sternocleidomastoid muscles. Palpation confirms phasic activity of the sternocleidomastoid muscles, tracheal tug, and decreased cricosternal distance, indicative of hyperinflation.
Auscultation of the chest may suggest a pneumothorax or movement of the endotracheal tube into the right main-stem bronchus. Cardiac auscultation may reveal the Hamman crunch, signaling the presence of pneumomediastinum. A new murmur may signal the development of a ventricular septal defect or other cardiac source of acute distress.
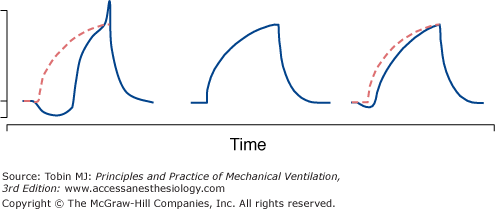
The pressure and flow tracings on the ventilator screen may provide clues to the sudden change in patient status. The contour of the airway pressure tracing may exhibit excessive scooping, indicating increased inspiratory effort; this pattern is seen when the delivered inspiratory flow no longer meets patient demand (Fig. 53-4).5,6 The flow tracing may not return to the zero-flow line in the period immediately before the next inspiration, indicating PEEPi.4 The flow tracing may exhibit a sawtooth pattern, indicating excessive secretions.7
Figure 53-4
Airway pressure waveforms recorded in a patient shortly after the initiation of mechanical ventilation (left), in a patient making no respiratory effort (controlled mechanical ventilation, middle), and in a patient receiving an appropriate level of assist-control mechanical ventilation (right). The dashed lines on the left and right panels reproduce the tracing achieved by passive, controlled mechanical ventilation as occurs in a patient receiving neuromuscular blocking agents. The left waveform depicts a patient in respiratory distress who has excessive work of breathing; this can be inferred from the initial concavity, which results from vigorous inspiratory effort, and the spike at the end of ventilator assistance, which is the result of expiratory muscle recruitment. The middle waveform depicts a patient making no respiratory effort. The right waveform depicts a patient performing an appropriate amount of respiratory work: The small downward dip at the start of the breath indicates the small inspiratory effort required to trigger the ventilator, and the distance between the solid line (actual airway pressure) and dashed line (expected tracing during controlled ventilation, as in the middle panel) is proportional to the amount of work performed by the patient’s inspiratory muscles while the ventilator is providing assistance. The patient in the right panel is performing much more respiratory work than the patient in the middle panel and much less work than the patient in the left panel. (Used, with permission, from Tobin et al.6)
A decrease in end-tidal carbon dioxide tension (PCO2) can be helpful. A sudden decrease in end-tidal PCO2 may arise with pulmonary embolism or air embolism. Esophageal intubation also causes end-tidal PCO2 to fall to zero. Unfortunately, end-tidal CO2 readings do not reliably reflect arterial CO2 tension (PCO2) in patients with underlying lung disease.
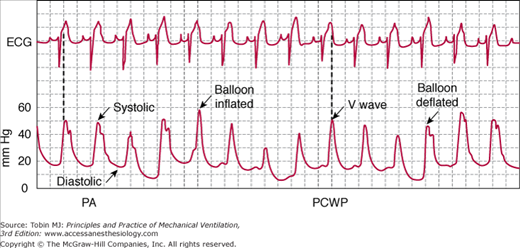
The blood pressure reading may signal a hypertensive crisis. The differential diagnosis of a fall in arterial pressure is wide, and influenced by clinical context. In a patient with a myocardial infarction, a fall in arterial pressure may signal the onset of cardiogenic shock. A pulmonary artery catheter can provide important clues. A new onset of large V (ventricular) waves in the pulmonary artery wedge tracing suggests the development of acute mitral regurgitation secondary to ruptured chordae tendineae cordis (Fig. 53-5).8 The electrocardiogram (ECG) may reveal arrhythmias or ST-segment elevation. In contrast to the average patient, communication with an intubated patient is very difficult. A nonintubated patient who develops acute distress consequent to an acute myocardial infarction is able to relay the onset of crushing chest pain to staff. The presence of a tracheal tube prohibits speech. Nevertheless, clinicians can obtain a considerable amount of information about the nature of symptoms. Nurses are frequently more patient and skilled than physicians in achieving an ongoing dialogue with a patient.
Figure 53-5
Simultaneous recordings of electrocardiogram (ECG, top) and pulmonary artery (PA) pressure (bottom) in a patient with acute mitral insufficiency. Following inflation of the balloon, giant V waves are seen in the pulmonary capillary wedge pressure (PCWP) tracing. Before balloon inflation and after balloon deflation, the PA tracing has a characteristic bifid appearance secondary to both a PA systolic wave and a V wave. Note that the V wave occurs later in the cardiac cycle than does the PA systolic wave, which is synchronous with the T wave on the electrocardiogram.
The sequence of the above steps will vary with the clinical context. The approach is also incomplete. For example, sudden distress may have no cardiopulmonary cause. Instead, it may originate in a blocked Foley catheter leading to an overdistended bladder.
Based on the initial assessment, a physician diagnoses the most likely cause of a change in a patient’s condition. Table 53-1 lists the most common causes of sudden respiratory distress in a ventilated patient.
|
Artificial Airway Problems
Several different problems with artificial airways can produce acute respiratory distress in the ventilated patient.
An endotracheal or tracheostomy tube that is initially properly positioned may subsequently move up or down in a patient’s airway. Downward movement into a main-stem bronchus (endobronchial intubation) is estimated to occur in approximately 10% of ventilated patients.9–11 It is usually caused by inadequate external fixation of the endotracheal tube, excessive neck movement, or both.12
Main-stem intubation occurs more often on the right side, because the right main-stem bronchus forms a less-acute angle with the trachea than does the left main-stem bronchus11 and because the bevel of most endotracheal tubes is directed rightward (Fig. 53-6). The lack of ventilation to the contralateral lung produces atelectasis, which, in turn, causes shunting of blood and hypoxemia. The delivery of a high volume to the intubated lung predisposes to the development of a pneumothorax.4
Clues to the development of endobronchial intubation include an increase in peak airway pressure accompanied by a proportional increase in plateau pressure, asymmetric expansion of each hemithorax, and a decrease in breath sounds over the contralateral lung. In a prospective study, however, Brunel et al10 found that clinicians considered breath sounds equal over both lung fields in 60% of patients with an endobronchial intubation.
If endobronchial intubation is suspected, the endotracheal tube should be pulled back a few centimeters, the chest auscultated again, and the position of the tube confirmed by radiography. The problem can be prevented by securing the tip of the tube at least 3 to 4 cm above the carina, and obtaining a radiograph at the time of intubation.12 In addition, centimeter markings on an endotracheal tube are helpful as a reference, although not completely reliable.10 In general, an endotracheal tube should be secured at reference markings of 23 cm in men and 21 cm in women.10 On the chest radiograph, the tip of the endotracheal tube should be located at a level corresponding to the top of the aortic knob when the head is in a neutral position.13
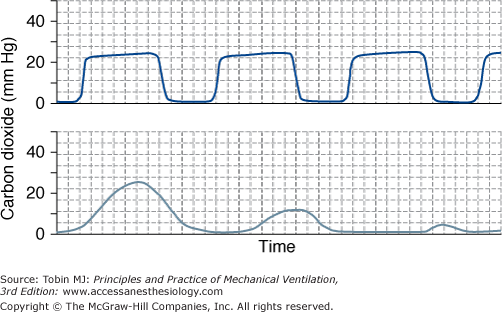
An endotracheal tube may also migrate above the vocal cords, or get dislodged into the esophagus.11 Esophageal intubation may arise as a result of attempts to blindly reposition an endotracheal tube. Malpositioning causes sudden distress accompanied by phonation, audible escape of air through the nose and mouth, absence of tube condensation, decrease in tidal volume (VT), decrease (or increase) in peak airway pressure, and abdominal distension. Chest radiography can be misleading because the trachea overlays the esophagus, making it difficult to tell whether the tube is located in the esophagus or trachea. A reading of zero on an end-tidal CO2 monitor helps in detecting esophageal intubation (Fig. 53-7). Upward movement of an endotracheal tube usually results from excessive neck movement and/or inadequate tube fixation. Conrardy et al12 found that neck extension caused an endotracheal tube to move an average of 1.9 cm away from the carina, but movement was as much as 5.2 cm in some patients. This observation may explain why patients who are carefully restrained can still self-extubate.
Figure 53-7
End-tidal CO2 tracing and esophageal intubation. Upper panel. End-tidal CO2 tracing in a patient in whom an endotracheal tube was inserted correctly in the trachea. During inhalation, end-tidal CO2 is 0 mm Hg. During exhalation, the CO2 tracing rises steeply, attains a near plateau, and then quickly returns to the baseline. The end tidal CO2 in this patient, 25 mm Hg, is below the normal range of 35 to 45 mm Hg. Lower panel. End-tidal CO2 tracing in a patient in whom an endotracheal tube was erroneously inserted in the esophagus. The expected shape of the CO2 tracing has been replaced by small transient rises in the CO2 tracing, which progressively decrease with each squeeze of the Ambu bag.
Herniation of the cuff of an endotracheal tube over its distal tip can cause complete airway obstruction.11 This problem most commonly occurs after changes in position of the tube or changes in the posture of the head and neck. Clues to cuff herniation include an increase in peak airway pressure, resistance during manual ventilation, a decrease in VT, difficulty in passing a suction catheter, and an abnormal musical sound during inspiration.2 Deflation of the cuff produces immediate relief. In a stable patient with an unclear source of airway obstruction, the diagnosis may not be made without bronchoscopy.
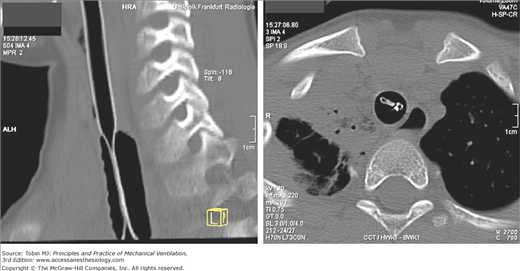
External compression of an endotracheal tube as a result of an inflated cuff is a rare cause of airway obstruction (Fig. 53-8).14
Figure 53-8
External compression of endotracheal tube. Computed tomography images of the sagittal plane of the cervical spine and trachea (left panel) and transversal plane at the level of the first thoracic vertebra (right panel) in an 8-year-old boy who required endotracheal intubation as a result of multiple injuries received in a traffic accident. The inflated cuff produced critical obstruction of the endotracheal tube. The white dot on the scan in the right panel indicates the obstructed lumen of the tracheal tube. (Used, with permission, from Hofstetter et al.14)
The failure of the cuff on an endotracheal tube to form a seal with the wall of the trachea leads to gas leakage, which may or may not be of clinical consequence.11 A large leak may cause the ventilator alarms to sound, signaling a low peak airway pressure and low exhaled VT, and cause alveolar hypoventilation. Other clues include ability to phonate, frothy secretions in the patient’s mouth, hearing a gurgle over the trachea or larynx on auscultation, a discrepancy between expired and inspired VT, and failure to maintain a set level of PEEP.11
The most common cause of cuff leak is malposition of the endotracheal tube. Consequently, the development of a cuff leak should cause the physician to reevaluate tube position.
The cuff may be deflated not only because of a leak in the cuff itself, but also because of a leak in the pilot balloon or in the external valve assembly.13 A clamp placed on the cuff inflation tubing proximal to the valve and pilot balloon can temporarily correct this problem (without having to immediately replace the endotracheal tube).
Complete rupture of a cuff can lead to aspiration of saliva, vomit, or food; the entire endotracheal tube needs to be replaced.
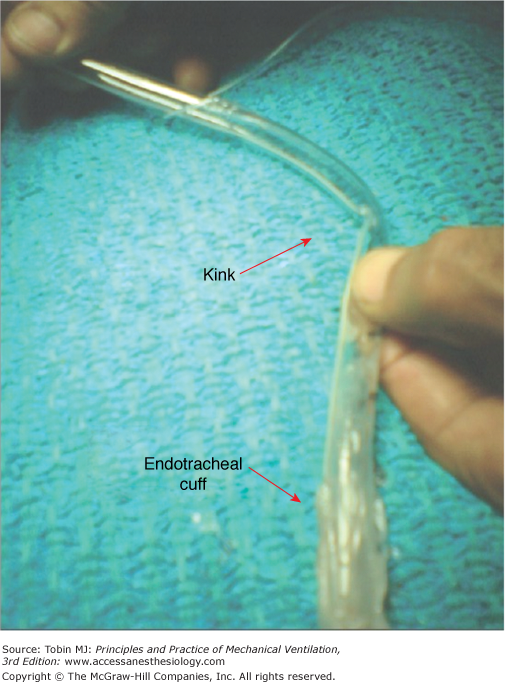
The endotracheal tube can become kinked as a result of changes in the position of the head and neck.10 Some authors have recommended immersing an endotracheal tube in warm saline as a means of decreasing the incidence of mucosal trauma and epistaxis following nasal intubation,15 but this practice has been reported to give rise to subsequent kinking.16 Kinking may also occur if the tube softens through being warmed by heated humidified gas,13 or when the patient bites the tube. Figure 53-9 shows an instance of endotracheal-tube kinking that was attributed to earlier movement of the tube from one angle of the mouth to the other.17 We speculate that this shift in position of the tube might have led to a force acting in a direction perpendicular to the torque of the natural curve of an endotracheal tube, and thus induced kinking. Kinking leads to an increase in peak airway pressure and difficulty in passing a suction catheter. When this occurs, the endotracheal tube should be withdrawn and replaced.
Figure 53-9
A kink found approximately 10 cm above the cuff of an endotracheal tube in a direction opposite to the natural curvature of the tube (toward the convex side) that resulted approximately an hour after the tube had been moved from the right angle of mouth and fixed on the left angle. (Used, with permission, from Hariharan et al.17)
The endotracheal tube can become occluded with a variety of foreign bodies, including dried lubricant,18 surgical tape,18 a broken stylet sheath,19 plastic debris retained from the time of manufacturing the Murphy eye in the tube,20 and nasal turbinates avulsed during nasotracheal intubation.21
Tracheoesophageal fistula is a rare complication that is primarily caused by ischemia of the tracheal wall secondary to pressure from an overinflated cuff or by movement of a tracheal tube or its tip.22 Patients with this complication typically have had both a tracheal tube and a nasogastric tube in place for a long time.11,23
Clues to the presence of a tracheoesophageal fistula include the inability to deliver a preset VT despite a functional cuff, gastric distension, audible leak of air through the mouth, copious airway secretions, the suctioning of gastric contents from the lower respiratory tract, and clinical deterioration and coughing on deflation of the cuff.13 Diagnosis needs to be verified by bronchoscopy with direct visualization of the esophagus or by cine-esophagography or cinetracheography.13 Most experts believe that closure of the fistula demands operative repair; opinions vary as to optimal timing.13
Erosion of the innominate artery has been reported to occur in 0.4% to 4.5% of tracheostomies.24 Most ruptures occur at some point during the first three weeks after a tracheotomy,24 with peak incidence 7 to 14 days after the procedure.25 The underside of the tracheal tube, the tracheal cuff, or the tip of the tube erode through the anterolateral tracheal wall, at the point where the innominate artery crosses the trachea. Risk factors include excessive movement of the tube, excessive movement of the neck, overinflation of the cuff, or radiotherapy. Several authors emphasize that making a tracheotomy incision below the third tracheal ring increases the risk of fistula formation, but the complication can occur even when the incision is placed between the second and third tracheal rings, as recommended.25
Any peristomal bleeding or hemoptysis in a patient with a recent tracheotomy should lead to detailed assessment to ascertain the underlying cause. Bleeding within 48 hours is usually associated with local factors such as traumatic puncture of anterior jugular or inferior thyroid veins, mucosal abrasion from a suction catheter, or systemic coagulopathy. Bleeding occurring 3 days to 6 weeks after a tracheotomy should be judged to be the result of innominate artery rupture until proven otherwise.25 The clinical presentation may be quite dramatic, with blood gushing from the tracheal tube. It may be heralded by pulsation of the tracheal cannula or a “sentinel bleed,” which is reported to occur in more than 50% of patients.25
Adequate oxygenation is the mainstay of immediate management with simultaneous identification and termination of bleeding.25 The tracheostomy cuff should be overinflated in an attempt to achieve tamponade, and the cuffed tube advanced so that the balloon lies distal to the tracheostomy stoma. An attempt should be made to digitally compress the artery by inserting an index finger into the stoma and compress the artery against the posterior surface of the manubrium.24 If this is successful, a blood transfusion should be administered while the patient is being transported to the operating room for sternotomy and ligation of the vessel. Time should not be wasted in performing imaging studies. Mortality is extremely high: greater than 85% in a review of the world literature.24
Other Problems
A stiff nasogastric tube can easily bypass the cuff of an endotracheal tube and enter the airway. If the nasogastric tube is connected to suction, the continuous negative pressure will create a constant flow of gas out of the airways.26 Consequently, the low VT alarm will sound. The negative pressure can also cause triggering of the ventilator. In particular, ventilator triggering in a patient who is receiving a paralytic agent suggests that a nasogastric tube is misplaced in the airway. Other complications of nasogastric tubes include passage of the tube through the tracheobronchial tree into the pleural space, causing the formation of a bronchopleural fistula; infusion of nutritional solutions through the misplaced nasogastric tube can cause an empyema. A nasogastric tube can also cause esophageal perforation leading to pneumomediastinum, pneumothorax, and tracheoesophageal fistula.
Airway secretions can cause problems by being too copious or too dry. Excessive secretions can lead to mucus plugging and atelectasis. To avoid this problem, careful bronchial toilet and frequent suctioning are necessary in patients with copious secretions. If atelectasis occurs and fails to resolve with conservative measures, bronchoscopy should be undertaken.
A helpful clue to the presence of excessive secretions is the presence of a sawtooth pattern on the flow tracing.7 In a study of fifty intubated patients, Jubran and Tobin7 found that the presence of a sawtooth pattern was approximately six to eight times more likely in patients who had secretions than in patients without secretions. Conversely, a smooth flow-volume curve was about one-quarter as likely to be found in patients with secretions as in patients without secretions. Clinical examination had much higher false-positive and false-negative rates (42% and 43%, respectively) than the flow-volume curves (12% and 14%, respectively). The usefulness of a sawtooth pattern for detecting secretions was confirmed by Guglielminotti et al27 in a study of sixty-two patients who were receiving pressure-support or assist-control ventilation.
A tracheal tube bypasses the upper airway, which normally heats and humidifies inspired gas. Consequently, secretions may become excessively dry and encrusted. Inspissated secretions can cause significant blockage of the tracheal tube over a relatively short period.23 Clues to this problem include an increase in peak airway pressure without an associated increase in plateau pressure, and difficulty in passing a suction catheter. Obstruction appears to be a greater problem when heat and moisture exchangers are employed, as opposed to hot water humidifiers (see Chapter 51).
When secretions dry and accumulate, they can cause complete obstruction or ball-valve obstruction of the endotracheal tube. When this occurs at the distal tip of the tube, positive pressure during inspiration will cause the mass to move away from the tip, permitting the flow of gas. During expiration, expiratory pressure will push the mass of dried secretions into the distal port producing occlusion and cessation of airflow. The ball-valve obstruction causes pulmonary hyperinflation and may produce a tension pneumothorax.28
Bronchoconstriction is a common cause of sudden respiratory distress. Clues to its development include an increase in peak airway pressure with little or no change in plateau pressure. Clinical manifestations include wheezes, pulsus paradoxus, and evidence of increased work of breathing (recession of the suprasternal space, supraclavicular fossae, and intercostal spaces; heightened activity of the accessory muscles; tracheal tug; and paradoxic motion of the rib cage and abdomen). Management includes bronchodilator therapy.
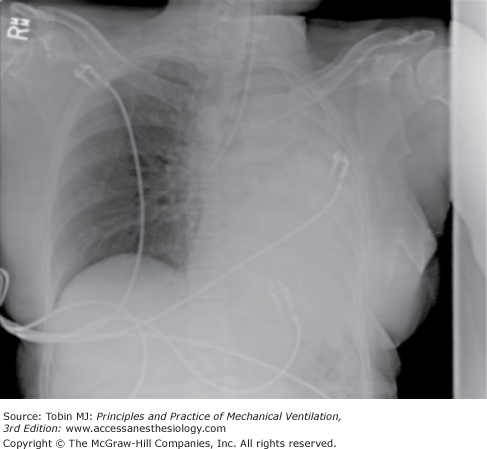
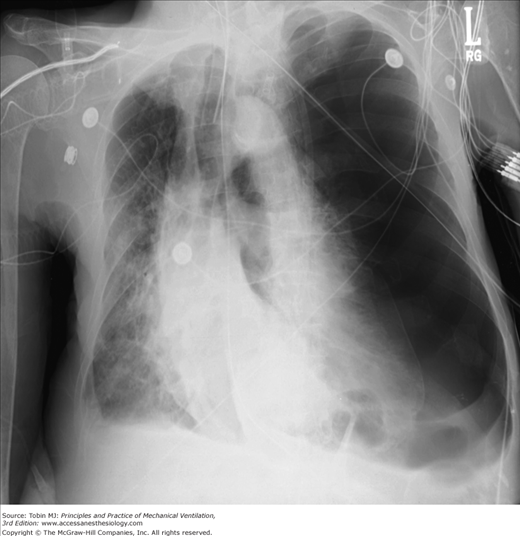
Sudden respiratory distress in a ventilator-supported patient should always arouse suspicion of a pneumothorax, because 60% to 90% of such pneumothoraces are reported to be under tension (Fig. 53-10).29Chapter 44 provides a full discussion of barotrauma and its pathogenesis.
Figure 53-10
Development of a large left pneumothorax in a mechanically ventilated patient. Portable anteroposterior chest radiograph showing large (tensionlike) pneumothorax with significant displacement of heart and mediastinum to the opposite side. Vascular congestion of the right lung also noted.
A pneumothorax is typically associated with an increase in peak airway pressure and a proportional increase in plateau pressure. Other manifestations include hyperresonance, tracheal deviation to the contralateral side, decreased breath sounds, and cardiovascular collapse. If a pneumothorax is suspected and death is imminent, a 14- to 16-gauge needle should be inserted into a second intercostal space. If a gush of air is heard, a thoracostomy tube is inserted. If the patient is stable, however, a chest radiograph should first be performed to verify the diagnosis before inserting a chest tube.
Pulmonary edema can cause sudden distress associated with an increase in plateau pressure and a smaller increase in the gradient between the peak and plateau pressures. When present, pink, frothy secretions and an increase in pulmonary artery wedge pressure are particularly helpful.
Acute pulmonary embolism is an uncommon cause of sudden respiratory distress in a ventilated patient. Typical manifestations include dyspnea, tachypnea, chest pain, fever, hemoptysis, pleural rub, and features of deep vein thrombosis. An important clue, when it occurs, is sudden hypoxemia with no change in peak airway pressure—a unique combination. The risks associated with transporting a critically ill patient alter the approach to diagnosis. Bedside duplex ultrasonography of the leg veins is a reasonable initial test.30 Although ventilation–perfusion scans are difficult to perform in a ventilated patient, they have reasonable diagnostic reliability.31 Helical computed tomographic scanning is worthwhile, although doubts have been raised about its accuracy in this setting.32
Some ventilated patients present with acute hypoxemia, which may or may not be accompanied by acute distress. The differential diagnosis for new onset of hypoxemia can be considered in terms of ventilator-related problems, progression of the underlying disease, onset of a new medical problem, effects of interventions and procedures, and medications.33Table 53-2 lists the causes of worsening oxygenation in the ventilated patient.
|