Nursing staff education and accreditation
Pain assessment using validated tools
Setup and change of pump and infusion circuit
Monitoring including sensorimotor block, pain, and sedation scores
Recognition of adverse effects
Discontinuation of epidural analgesia and catheter removal
Medical protocols and/or guidelines
Standardized prescribing
Frequency of patient assessment and handover
Mobilization and enhanced recovery
Identification and management of serious complications
Follow-up
Quick reference guide
Resuscitation team, oxygen, drugs, and equipment available
Documentation
Patient information (pre- and postprocedure)
Audit and quality assurance
Training in Epidural Techniques
Epidural block is a core skill for anesthesiologists and a clear understanding of the relevant anatomy, physiology, and pharmacology is required. As well as technical aspects of safe epidural blockade such as gentle technique and meticulous asepsis, it is essential that the importance of patient preassessment and case selection, consent and documentation is understood. Utilization of a test dose (e.g., lidocaine with adrenaline ) to detect inadvertent intrathecal or intravascular catheterization and awareness of warning (‘red flag’) symptoms and signs which may signal an evolving neurological complication are key teaching points [31, 32]. The Australian and New Zealand College of Anesthetists requires 50 labor epidurals, 20 other lumbar epidurals, and five thoracic epidurals to be performed during anesthesia training [33]. However, it has been shown using CUSUM analysis , a useful tool to assess proficiency in practical skills, that this number may not be sufficient for all trainees to achieve competency and training should be tailored to the individual [34]. Konrad and colleagues reported in a learning curve study that 90 epidural attempts are needed to achieve a success rate of 80 % [35]. In our experience, the use of peripheral nerve and trunk blocks as well as local infiltration techniques has reduced training in epidural analgesia. This trend should be monitored by training bodies as it could lead to deskilling of trainees although workshops and simulators can supplement in-theatre training. Ultrasound can be useful to confirm the position of the interspinous space (i.e., midline) and the depth of the ligamentum flavum, especially when attempts at conventional insertion have failed or the procedure is expected to be difficult [36]. The evidence for ultrasound-assisted epidural catheter placement is limited but utilization is likely to increase as image quality and needle visualization improves [37, 38].
Risks of Epidural Anesthes ia
Complications of epidural block may arise from physiological changes resulting from the block, adverse drug effects, problems associated with the needle or catheter, or wrong drug/route error. The adverse effects and nonneurological complications which should not result in permanent harm if treated appropriately are listed in Table 14.2. Intravenous access should be secured prior to commencing epidural blockade and maintained for the duration of the infusion. Death or major morbidity from wrong drug or route error is idiosyncratic and rare. In contrast, neurological complications of centroneuraxial block assume two distinct patterns. Temporary neuropathies are typically patchy sensory deficits whereas a serious neurological complication can be defined as having the potential to cause permanent functional impairment (usually from weakness or pain). Mechanisms and causes of damage to the neuraxis attributable to epidural blockade are listed in Table 14.3.
Table 14.2
Adverse effects and nonneurological complications of epidural blockade
Adverse effects |
Hypotension |
Urinary retention |
Pruritus |
Motor block |
High block |
Block failure |
Allergy |
Complications (excluding neurological) |
Local anesthetic toxicity |
Postdural puncture headache |
Table 14.3
Causes of neurologic damage attributable to epidural blockade.
Cord compression |
Hematoma |
Needle or catheter trauma |
Coagulopathy |
Abscess |
Exogenous infection via needle/catheter |
Hematogenous |
Local spread (e.g., paravertebral) |
Cord ischemia |
Anterior spinal artery syndrome |
Cord and nerve root trauma |
Needle/catheter/injectate |
Arachnoiditis (inflammation) |
Wrong drug or toxic injectate |
Infection |
Local anesthetic neurotoxicity |
Coincidental Causes of Neurologic Inju ry
Consideration of noniatrogenic causes is mandatory (see Table 14.4). Coincidental pathology such as spinal stenosis can lead to cord compression when triggered by the additional volume of local anesthetic injection. It is well known that temporary obstetric palsies occur with and without neuraxial blockade [39]. Surgical damage [40] and patient positioning [41, 42] are both likely to be more common causes of permanent nerve injury than regional anesthesia. In a recent large retrospective 10-year single-center study of 380,000 consecutive patients undergoing all types of procedures, Welch and colleagues reported that the use of general or epidural anesthesia increased the risk of postoperative peripheral nerve injury, but there was no difference with the use of peripheral nerve blockade or spinal anesthesia [43]. In this study, nerve injuries resulting from the surgical procedure were excluded. Proving that the regional technique was not responsible may be impossible, even with advanced diagnostic techniques, and there is significant potential for misclassification of these injuries.
Table 14.4
Coincidental conditions mimicking neurologic injury from epidural blockade
Spinal tumors |
Spinal vascular malformation |
Prolapsed intervertebral disc |
Guillain–Barré syndrome |
Multiple sclerosis |
Spinal hematoma |
Metastases |
Thalassemia |
Infections (e.g., viral) |
Embolic |
Iatrogenic (e.g., hypotension, surgery, positioning, drugs) |
Serious Neurological Complications of Epidural Anesthes ia
Whereas general anesthesia techniques have gradually become safer and less disruptive of normal physiology, the invasiveness of regional techniques is little changed; serious complications of centroneuraxial block still occur and they are often severe. Discussion of material risk remains topical [44, 45]. Although it is highly desirable to provide an estimate of the risk of a severe complication prior to a neuraxial procedure, the incidence of severe neurological complications quoted to the patient varies widely and the issue is reported to be a source of “confusion and concern” for anesthesiologists [46, 47]. Although a number of large studies have been published during the last 15 years, which help to provide contemporary data, it remains difficult to estimate incidence confidently for an individual patient.
The range of reported incidence figures reflects varied methodology—different patient populations have been studied and there is no consistent definition for a serious neurological complication. For example, some studies include patients who recovered, drug errors and cardiovascular complications [48, 49], or exclude major categories such as spinal hematoma and epidural abscess [50]. Sources of numerator and denominator data include voluntary reporting [49], postal questionnaire [25, 48], hotline reporting [51], pharmaceutical sales [25], and analysis of litigation or no-fault insurance systems [40, 48, 49]. Better studies corroborate their figures using multiple sources including referrals to neurology, radiology, and neurosurgery but this heterogeneity makes comparison difficult and the incidence figures quoted must be seen as estimates only.
Also, many studies are not powered to detect severe complications because very large numbers of patients are needed. Studies reporting no adverse events (i.e., zero numerators) in seemingly large series are easy to misinterpret. The “rule of 3/n” should be used; it states that for n observations with a zero numerator the upper 95 % confidence limit is 3/n [52]. For example, if a study reports that no neurological complications are observed in 4000 procedures, then according to the rule of 3/n which describes the upper 95 % CI for the actual incidence, the rate of permanent injury may be approximately 1:1400 [53, 54].
Third, an overall figure for incidence of serious complications of epidural analgesia should not be provided because the risk of a severe complication may differ up to 100-fold between patients at low risk [55] and those with multiple risk factors [25, 49]. The importance of considering risk: benefit on a patient-by-patient basis is highlighted in a retrospective study published in 2004 by Moen and colleagues [25]. This key study reported the incidence of serious neurologic complications associated with centroneuraxial blocks performed during the 1990s in Sweden and achieved the participation of 85 % of anesthesiology departments. The estimated denominator was 1,260,000 spinals and 250,000 epidurals (including CSE ). In the general population, severe neurologic complications occurred after 1 in 3600 epidural procedures but the rate was higher in women undergoing total knee arthroplasty (1 in 1800) and much lower in obstetric patients (1 in 25,000). Major neurologic complications most commonly occurred in patients undergoing orthopedic, general, vascular, and urologic surgery. The authors proposed this was related to important risk factors in this group of patients including disordered coagulation, osteoporosis, spinal stenosis, and immunosuppression. In this study, patients who recovered after treatment for a serious complication were included.
The 3rd National Audit Project of The Royal College of Anesthetists (NAP3) was a 1 year prospective audit published in 2009 which included adverse events relating to wrong drug/route error and cardiovascular collapse, as well as serious neurological complications of centroneuraxial blockade [49]. Participation of 100 % of public hospitals in the United Kingdom was achieved and the estimated denominator included 293,000 epidurals and 42,000 CSEs. The majority of severe complications occurred after epidural or CSE procedures in the perioperative setting where the incidence of permanent harm was approximately 1:5700 compared to 1 in 166,000 epidural blocks and 1 in 25,000 CSEs in obstetric patients. Although the authors concluded that the results were largely reassuring, 22 patients were excluded from analysis because complete recovery was documented by 6 months. These cases included epidural abscesses, spinal cord trauma, and an intravertebral hematoma as well as an unknown number of other cases with full resolution prior to reporting. When these 22 known serious complications with full recovery are included, the overall incidence in NAP3 was very similar to that found in the Swedish study by Moen and colleagues [25, 56].
It is important to note that the incidence of severe complications associated with spinal anesthesia is low and relatively consistent across subgroups—around 1 in 20,000 or less [25, 49, 57]—and all neuraxial block techniques (i.e., epidural, CSE , and spinal anesthesia) are associated with very low complication rates in obstetric patients [25, 49, 55].
Risk Factors for Neurologic Injury
The recognized patient- and technique-related factors which increase the chance of a serious neurological complication after neuraxial block are presented in Table 14.5 [58–60]. Older patients with multiple comorbidities undergoing epidural blockade in the perioperative setting are particularly at risk of serious neurological complications [48, 49]. Coagulation abnormalities and degenerative conditions of the spine, including spinal stenosis and osteoporosis, a common combination in older female patients, are also important risk factors [25]. Degenerative changes in the spine reduce the compliance of the intravertebral space and injection of a large fluid volume into the epidural space can cause transient paraplegia [61]. If the diagnosis is known in advance, other regional techniques such as plexus, trunk or peripheral nerve blockade, local infiltration, or spinal anesthesia can be selected, avoiding cord compression risk [62]. However, a serious complication can occur in the absence of any risk factors, [25, 63] and significant hemorrhagic complications of peripheral nerve and plexus block have also been reported [64].
Table 14.5
Risk factors for severe neurologic complications after epidural blockade
Patient factors |
Female sex |
Atherosclerosis |
Diabetes |
Advanced age |
Spinal disorders |
Osteoporosis |
Ankylosing spondylitis |
Spinal stenosis |
Osteoarthritis |
Other spinal deformity |
Neuropathy |
Coagulation abnormality (including liver disease; bleeding disorder) |
Technique factors |
Epidural catheter |
Traumatic puncture |
Dysesthesias during insertion |
Prolonged continuation of block |
Hypotension |
Diagnosis of Neurologic Inju ry
Because compressive causes such as spinal hematoma and abscess are reversible if treated promptly, dilute solutions of local anesthetic, which avoid motor block, should be used so that pathologic lower body neurological deficit can be identified as early as possible. Initial presentation as cauda equina syndrome is a feature of several etiologies and it should be readily recognized by all practitioners. The fine autonomic fibers of the cauda equina are often the first to be affected by compression, ischemia, or neurotoxicity [65]. Damage to S2-S4 roots produces an atonic bladder although continence may be preserved if intravesical pressure is low. Progression of the syndrome leads to weakness of muscles below the knee as well as the hamstrings and gluteal muscles, with loss of ankle jerks and preservation of the knee jerk. Sensory loss in the sacral roots produces the characteristic saddle-shaped anesthesia of the perineum, buttocks, and thighs, extending to foot and calf if L5 and S1 roots are involved. Even using the lowest possible effective concentration of local anesthetic for epidural infusion, detection of the onset of painless cauda equine syndrome may be difficult in an immobile postoperative patient with a catheterized bladder. Patient outcome depends on vigilance and a high index of suspicion, especially in at-risk cases. The cardinal signs of new or progressive sensorimotor deficit, bladder dysfunction, and back pain can usually be detected using serial neurological assessment every 4 h until 24 h after epidural catheter removal. If these ‘red flag’ features become apparent, the epidural infusion should be discontinued to exclude a local anesthetic or volume effect. Magnetic resonance imaging (MRI ) should be performed within 4 h after the infusion is stopped if the situation has not improved, as well as neurosurgical referral and early decompression if indicated. Should there be a delay in stopping the epidural infusion after onset of new ‘red flag’ signs, MRI scanning may need to proceed before the local anesthetic effect resolves [66]. In summary, the key management principles are (1) diligent serial clinical neurologic assessment; (2) early MRI ; and (3) consultation with colleagues in radiology, neurology, and neurosurgery.
Intravertebral Hematoma
Bleeding into a relatively enclosed bony space such as the intravertebral canal can result in permanent neurological sequelae with minimal volume by compressive effects (Fig. 14.1). Spinal cord compression from a hematoma developing in the subarachnoid, subdural, or extradural space can rapidly produce irreversible paraplegia but the deficit is potentially reversible if treated early [66].
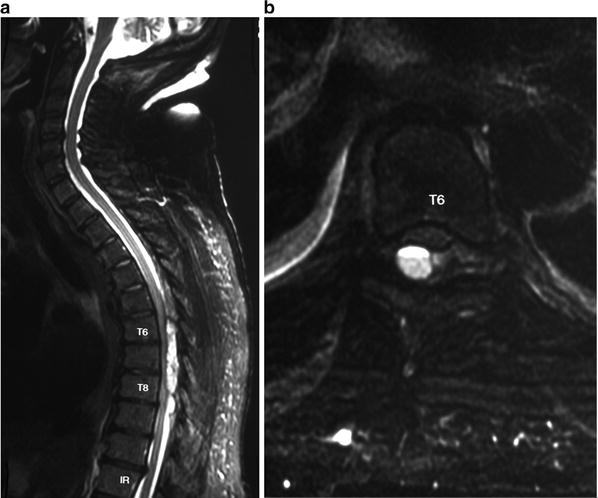

Fig. 14.1
Thoracic epidural hematoma demonstrated by MRI . Sagittal (a) and axial (b) T2 MR images of cervicothoracic spine showing acute epidural hematoma posterior to the cord at T5–T9 level. Note well-defined layering of blood in (b) (Courtesy Dr. Ayton Hope, Auckland City Hospital.)
Etiology
It is clear that an epidural Tuohy needle is more traumatic than a single-shot spinal anesthetic using a fine pencil point needle. The study by Moen and colleagues reported that the incidence of spinal hematoma in the nonobstetric population was 1:10,300 after epidural versus 1:480,000 after spinal blockade [25]. Intravertebral hematoma is more likely to occur in the presence of the ‘red flags’ listed in Table 14.5 but may occur in the absence of recognized risk factors [63]. In the study by Moen and colleagues [25], coagulation abnormality was documented in only one-third of spinal hematoma which underlines the importance of vigilance and postoperative neurologic monitoring. Some of these cases may be caused by puncture of epidural veins or Adamkiewicz’s artery , which usually lies close to the midline along the L3 spinal root or in some cases L4/5 [67].
Incidence
After low-molecular-weight heparins (LMWHs) were introduced for routine thromboprophylaxis in 1993, an unfortunate cluster of nearly 60 cases of spinal hematoma occurred in the United States that were associated with administration of relatively high doses of LMWH . Those affected were typically older female orthopedic patients undergoing joint replacement surgery with epidural analgesia, for whom the estimated reporting rate of spinal hematoma was 1 in 3100 during 1993–1997 [57]. But this is not a problem which is confined to the United States; the similar incidence in Sweden [25], Australia [13], Germany [21], and the United Kingdom [68] suggests that this is a worldwide phenomenon. In contrast, the incidence of spinal hematoma after obstetric epidurals is probably less than 1:100,000 [48, 55]. In Moen and colleagues’ study [25], there were two spinal hematomas reported among 255,000 obstetric blocks, but both occurring in patients with the syndrome of hemolysis, elevated liver enzymes, and low platelets (HELLP).
Diagnosis and Treatment
Because prognosis is better when preoperative neurologic deterioration is less severe, early diagnosis of spinal hematoma should be a central aim of postoperative surveillance. In a neurosurgical review of all 613 cases of spinal hematoma identified in the literature until 2003 [67], only one in ten reported cases was related to “a needle in the back,” the largest group being idiopathic/spontaneous (38 %). Of the cases related to neuraxial blockade, symptoms appeared within 24 h in two-thirds of evaluated cases (31/46). Overall, complete neurologic recovery from spinal hematoma was achieved in about 40 % of cases. Comparing prompt surgical intervention (laminectomy and clot evacuation within 12 h) with treatment delayed beyond 12 h, the rate of complete recovery was 66 % versus 29 %. Although recovery after conservative treatment occurred in 25 of 33 cases (76 %), these patients were carefully selected [67].
Prevention
Formalized guidelines for centroneuraxial blockade in the presence of anticoagulation have subsequently been developed and implemented in several jurisdictions including the United States [64], Scandinavia [69], and Europe [70]. The American Society of Regional Anesthesia (ASRA ) guidance for use and timing of neuraxial block in the context of drugs which affect coagulation is summarized in Table 14.6 [64]. These expert recommendations are not based on high-level evidence but on pharmacology of hemostasis-altering drugs or published case reports and case series. Partial or complete hemostatic failure from any cause (or combination of causes) produces a spectrum of risk, which may be negligible in the case of low-dose aspirin, and very high in fully heparinized patients [71], or in the presence of thrombolytic therapy [72]. Drug half-lives can be longer in the elderly and in patients with renal impairment and other comorbidities. The guidelines stress the importance of other precautions for safe practice, including frequent clinical neurological surveillance and avoidance of local anesthetic solutions which cause motor block [64].
Drug class | Recommendation |
|---|---|
Antiplatelet drugs | |
(a) Aspirin/dipyridamole/NSAIDsa | No contraindication; perform block at any timeb |
(b) Thienopyridine derivatives | Discontinue agent 7 days (clopidogrel) to 14 days (ticlopidine ) before CNBc |
(c) GP Ilb/IIIa receptor antagonists | CNB contraindicated within 8 h (eptifibatide, tirofiban) to 48 h (abciximab) of administration |
UH | |
(a) Subcutaneous | No contraindication with twice daily dosing (<10,000 U/day)d |
(b) Intravenous | Perform CNB or remove catheter 2–4 h after last dose and confirmed normal APTT ; delay heparin administration for 1 h after CNB (e.g., intraoperatively)e |
LMWH | |
(a) Prophylactic | Single daily dosing: CNB or catheter removal 10–12 h after LMWH; administer LMWH 4 h after CNB/catheter removalf |
Twice daily dosing: Delay LMWH for 24 h after surgery and remove epidural catheter 2 h before first dose | |
(b) Therapeutic | Delay CNB at least 24 h after LMWH ; otherwise as above |
Oral anticoagulants | |
(a) Warfarin | After stopping warfarin, document normal INR before CNB; when starting warfarin, remove catheter when INR still ≤ 1.5 |
(b) Factor Xa inhibitors | Rivaroxaban: CNB 48 h after last doseg |
(c) Direct thrombin inhibitors | Fondaparinux: CNB 72 h after last doseg |
Dabigatran: CNB 72 h after last doseg | |
Thrombolytics | Insufficient data; extreme riskh |
Herbal medicines | No contraindication; perform block at any timeb |
Data from a large retrospective audit from Finland during the period 2000–2009 [48], as well as the NAP3 audit from the United Kingdom [49], suggest that guidelines for neuraxial blockade in the setting of anticoagulation have reduced the incidence of spinal hematoma but not eliminated the risk [20]. In the Finnish study, timing of antithrombotic drug administration was not in accordance with current recommendations in six out of 13 cases of intravertebral hematoma following neuraxial puncture, reinforcing the fact that serious complications can be avoided when practice guidelines are followed [20, 48].
Acceptable Laboratory Values for Safe Institution of Epidural Blockade
The minimum platelet count below which it is safe to place an epidural is not known. It is generally accepted that isolated thrombocytopenia to a platelet count of 100 × 10 [9]/L does not pose a risk for spinal hematoma, in the absence of other risk factors, and there is some evidence that the safe level may be as low as 75 × 10 [9]/L in obstetrics [73, 74]. Use of bleeding time as a screening test is not recommended but in an individual patient with a history of bleeding or easy bruising, platelet function analysis can be useful to identify platelet function disorders [75].
It is probably inadvisable to perform epidural blockade if any abnormality in other coagulation parameters (e.g., prothrombin time (PT) or activated partial thromboplastin time (APTT )) is present. However, where the anticipated benefits are great or if general anesthesia is contraindicated, there is some evidence that minor abnormalities are acceptable for single-shot spinal anesthesia although a nonneuraxial technique would be preferable [63]. There are very limited data in the setting of congenital disorders such as hemophilia and von Willebrand’s disease [76]. Factor levels should be certainly be corrected to normal if centroneuraxial block is warranted although in our institution neuraxial blockade is avoided in these patients [76]. Coagulopathy associated with other conditions such as major trauma, sepsis, uremia, and liver failure is incompatible with centroneuraxial block until platelet abnormalities and clotting pathways are corrected [74]. For deeper block techniques such as paravertebral or lumbar plexus block, it is recommended that neuraxial precautions are followed but single-shot peripheral nerve blocks performed using ultrasound are relatively safe in the setting of abnormal coagulation [64, 70].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





