Fig. 16.1
Prevertebral region of the neck . The target site for needle insertion in classical approach is marked as asterisk. The breadth of the transverse process is marked as A. Reproduced with permission from Philip Peng Educational Series
The most common approach to the stellate ganglion is an anterior paratracheal approach at the level of the cricoid cartilage (C6) with or without fluoroscopy guidance. This approach is essentially a blockade of the cervical sympathetic chain in proximity to the middle cervical ganglion instead of the stellate ganglion. Thus, the classical approach is better termed cervical sympathetic block. The sympathetic outflow to the head and neck region (cervical trunk) can be blocked independently of the fibers to the upper limb [5]. Thus, development of Horner’s syndrome does not guarantee successful sympathetic blockade of the upper limb.
Also described are an anterior C7 paratracheal approach, medial approach to uncinate process [6], and a posterior T2 paravertebral approach [7]. The posterior approach aims to interrupt sympathetic outflow to the upper extremity with less chance of Horner’s syndrome. Thus, it may be indicated for neurolytic blockade when long-term side effects are undesirable.
The overall complication rate for stellate ganglion block is estimated to be approximately 0.17 % [8]. These complications range from moderate to severe and can mostly be attributed to incorrect placement of the needle and anomalous spread of local anesthetic.
Complications
Needle Trauma
Structures that lie close to the path of needle insertion are either vessels or organs in the vicinity. Moore documented puncture of pharynx , trachea , and esophagus [9]. Literature confirms that the esophagus is displayed lateral to the cricoid in approximately 50 % of the individual (mostly left side) and the prevalence is even higher at C7 level (Fig. 16.2) [10]. When the esophagus is punctured, mediastinitis is a legitimate concern [11]. Pneumothorax is another recognized risk, especially with the anterior C7 approach, as the dome of the pleura may extend 2.5 cm above the level of the first rib, especially on the right side. The risk of pneumothorax is increased further in tall, thin persons. The incidence of pneumothorax is up to 4 % with the posterior approach, which shares many of the risks of the thoracic paravertebral sympathetic block.
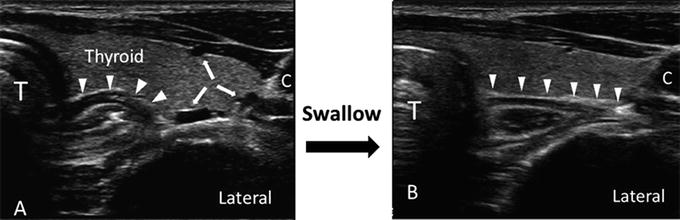

Fig. 16.2
Ultrasonographic image of neck at C7 level showing the variation of position of esophagus with swallowing. (a) Before swallowing, the esophagus (arrow heads) was seen covering half of the distance between trachea (T) and carotid artery (C); (b) during swallowing, the esophagus moved laterally toward the carotid artery, virtually covering the whole area between trachea and carotid artery. Note that the bold arrows showed the presence of three vessels in the preswallow scan. Swallowing action was evident by the increased in hyperechogenic shadow in the trachea. Reproduced with permission from Philip Peng Educational Series
Retropharyngeal hematomas are another potential complication due to puncture or passage of the needle through local vessels, which include vertebral artery (aberrant course), inferior thyroidal artery, ascending and deep cervical arteries (Fig. 16.3) [3, 10, 11]. Such hematomas have been reported with symptoms ranging from minimal patient discomfort to complete loss of the patient’s airway [12]. The frequency of catastrophic retropharyngeal hematoma after stellate ganglion block is approximately 1 in 100,000 cases [13]. However, the incidence of asymptomatic hematoma is much higher [14].
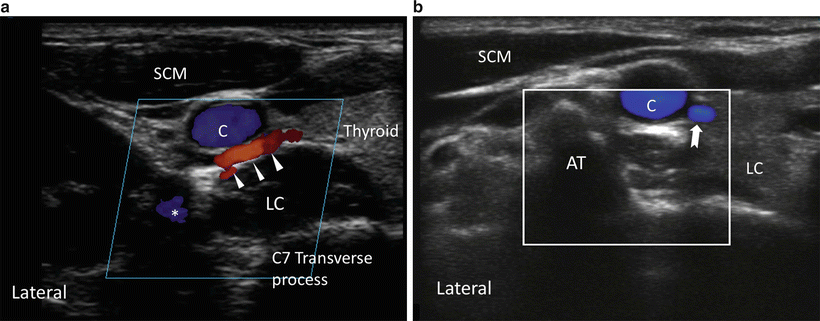

Fig. 16.3
(a) Ultrasonographic image of neck at C7 level on the right side showing the inferior thyroidal artery (arrow heads) in the long axis, crossing ventral to the paravertebral fascia. (b) Ultrasonographic image of neck at C6 level on the right side showing a vessel in the short axis (bold arrows). LC longus colli muscle, C carotid artery, SCM sternocleidomastoid muscle, AT anterior tubercle, * vertebral artery. Reproduced with permission from Philip Peng Educational Series
Intravascular Injection
The vessel most at risk is the vertebral artery. At the level of C7, the vertebral artery lies anterior to the stellate ganglion, before it swings posterior to enter the foramen transversarium of the sixth cervical transverse process. Thus, the anterior C7 paratracheal approach has a greater risk of vertebral artery puncture. However, the literature suggests that the vertebral artery enters the foramen transversarium at a level above C6 in 7–10 % (Fig. 16.4) [10, 15]. Kozody et al. have shown that as little as 2.5 mg of bupivacaine (a test dose) can cause major CNS effects when accidentally injected into the vertebral artery [16]. A smaller 1-mL test dose is recommended. Intravertebral artery local anesthetic injection may produce dizziness, nausea, light-headedness, and hypotension with low dosage and can result in coma, convulsion, and respiratory depression when higher doses are used [17]. These side effects are due to the direct effect of the local anesthetic on medullary and pontine centers. The duration and nature of the toxic effects depend on the dose injected and global and regional cerebral blood flow, as well as the precise neurovascular anatomy. Local anesthetic-induced neurologic symptoms, which appear after a low-dose injection, are often short-lived (minutes).
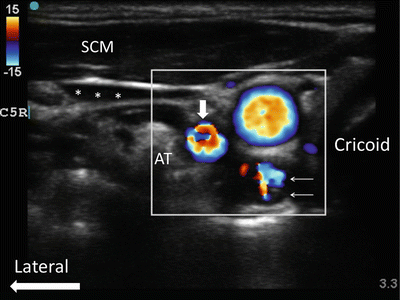

Fig. 16.4
Ultrasound image of the neck at the seventh cervical vertebral level (C7) with Doppler imaging. Va vertebral artery, Ca carotid artery, LC longus colli muscle, SCM sternocleidomastoid muscle, * internal jugular vein (compressed), solid bold arrow vertebral artery, line arrows artifact secondary to the shadowing of the carotid artery. Reproduced with permission from Philip Peng Educational Series
Accidental injection of air into the vertebral artery, with subsequent cerebral air embolism was reported by Adelman [18]. This complication represents two errors, not just one. Other vascular structures at risk are the carotid and jugular vessels, which lie lateral to the needle path, but there are no recent reports of puncture of these blood vessels .
Intraspinal Injection
Nerve roots of the brachial plexus exiting from intervertebral foramen may have an accompanying dural cuff. The vertebral canal and its contents lie posteromedial to the stellate ganglion. Thus, dural puncture may occur, either as a result of needle placement too medial or injection into a lateral extension of the perineural dural cuff of the cervical somatic nerve root [19]. Intrathecal injection of local anesthetic will produce a high spinal block, characterized by loss of consciousness, high motor block, hypotension, and apnea. This serious complication necessitates ventilatory and hemodynamic support until it wears off. Transient locked-in syndrome has been reported, as has subdural injection [20–22]. Wulf and Maier, in a survey of approximately 45,000 stellate ganglion blocks performed in Germany, reported six subarachnoid blocks and three high epidural injections [23]. Most important of all, care should be taken to avoid inadvertent injection of neurolytic agents into the epidural, subdural, or subarachnoid spaces, as this may lead to long-term neurologic deficit such as spinal cord infarction [24].
Anomalous Spread of Drug
Even when the drug is injected into the correct anatomical plane, anomalous spread may cause complications. Both bilateral recurrent laryngeal nerve palsy and contralateral Horner’s Syndrome have been reported [25]. Bilateral block causes unopposed vocal cord adduction and airway obstruction. Local anesthetic spread posteriorly and anterolaterally can produce brachial plexus blockade in up to 10 % of patients, and phrenic nerve block, respectively [6]. Because of the possibility of somatic spread, it is necessary to check for normal sensory and motor function in the blocked limb when evaluating the success of the sympathetic block .
Drug Effects
Extensive blockade of the cardiac sympathetic nerves has been reported following a properly performed stellate ganglion block. This resulted in bradycardia, secondary to unopposed vagal tone [26]. This has resulted in at least one case of cardiac arrest [27]. Schlack et al. demonstrated in a canine model that left stellate ganglion blockade caused impairment of left ventricular function. The mechanism was asymmetric cardiac contraction and asynchrony, caused by loss of sympathetic tone in the antero-apical segment of the left ventricle, supplied by the left sympathetic chain [28]. Although it is difficult to extrapolate these animal data to humans, who may have different patterns of myocardial innervation, the authors suggest that it may remain a risk in patients with already compromised cardiac function. Data to confirm this are lacking.
One case of migraine has been reported following a stellate ganglion block, presumably due to an idiosyncratic reaction and a loss of unilateral sympathetic tone in the cerebral vasculature [29]. Although absorption of correctly injected local anesthetics to toxic levels would be considered unlikely in stellate ganglion blockade, Wulf et al. reported toxic plasma levels in 30 % of patients after injecting 10 mL of 0.5 % bupivacaine [23, 30]. There have been no recent reports of injection of the wrong drug, but it remains a theoretical possibility.
Ultrasound emerges as a popular method for the guidance of pain intervention [5]. The advantages of ultrasound over fluoroscopy are that it allows the visualization of the soft tissues and vessels, and precise delivery of the local anesthetic to the CST , which is defined by the fascia, not the bony structure. Therefore, ultrasound potentially minimizes the risk of direct trauma to the vessel and organs and reduces the total dose of local anesthetic injected. Typically, 3–5 mL of local anesthetic is required .
Thoracic and Lumbar Sympathetic Blockade
The sympathetic chain lies in the paravertebral region, receiving fibers from somatic nerve roots via the rami communicantes. In the thoracic region it lies adjacent to the neck of the ribs, relatively close to the somatic nerve roots and the parietal pleura, with pneumothorax being a possible complication. For this reason, transcutaneous approach to the thoracic sympathetic chain without radiologic imaging support is not commonly performed. Long-lasting thoracic sympathectomy is usually achieved by surgical ablation, using either thoracotomy or, more recently, thoracoscopy .
In the lumbar region , the sympathetic chain and its ganglia lie on the anterolateral border of the vertebral bodies, separated from the somatic nerve roots by the psoas muscle and fascia. The ganglia are found in variable locations but most consistently found at the L3 level (Fig. 16-5) [31]. The popular technique is fluoroscopy-guided needle insertion to the anterolateral border of the L2, L3, or L4 vertebrae. It requires the insertion of a needle 5–6 cm from the posterior midline with the patient in the prone position. The needle passes through the paravertebral muscles, “walks off” the transverse process, and passes through the psoas muscle and fascia to reach the lumbar sympathetic chain in the anterolateral aspect of the vertebra. The volume of local anesthetic injection varies, from high volume (e.g., 20 mL) at a single level to low volume at multiple levels .
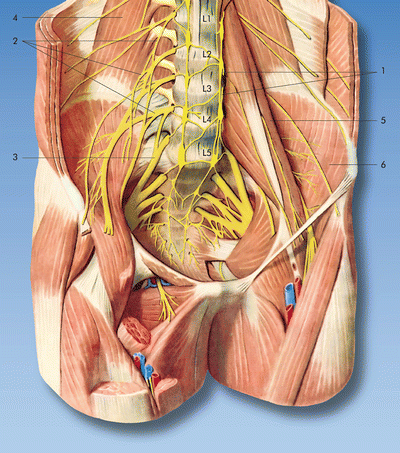

Fig. 16.5
Anatomy (anterior view): (1) sympathetic trunk with communicating branches, (2) lumbar plexus, (3) lumbosacral trunk, (4) quadratus lumborum muscle, (5) psoas major muscle, (6) iliac muscle Reproduced with permission from Dr. Danilo Jankovic
Complications
Intraspinal and Intravascular Injection
The vertebral column and the spinal canal lie posteromedial to the sympathetic chain. Injection of local anesthetic in the spinal canal is rare, but theoretically possible. Intraspinal injection (intrathecal, epidural, or subdural) and postdural puncture headache can follow puncture of either an extended dural cuff or the intraspinal dura [32, 33]. Intravascular injection is a possible complication, as both the aorta and inferior vena cava lie anterior to the sympathetic chain. Puncture of these structures is rarely reported , but it can occur in the clinical setting. The vertebral venous plexus is also at risk, as it is close to the path of the needle. The risk of intravascular injection into either a perivertebral vein or a major vessel is minimized by appropriate use of fluoroscopy and contrast medium before the injection of local anesthetics or neurolytic agents. Negative aspiration before injection yields false reassurance, as the sensitivity of this test is only 40.7 %. This low sensitivity can be explained by the fact that the veins of the vertebral plexus constitute a thin-walled, low-pressure system and collapse under aspiration [34].
Needle Trauma
Confirmation of needle position with fluoroscopy is necessary when performing neurolytic blockade of the lumbar sympathetic ganglia. The risks of ‘blind’ technique are needle trauma to the kidney, ureter, and bowel. In a cadaver study, three out of 80 “blind” needle attempts resulted in needle insertion into grossly osteoporotic vertebral bodies or the hilum of the kidney [35]. These incidents can be prevented with the utilization of fluoroscopic guidance .
Drug Effects
Complications can occur from the use of either local anesthetics or neurolytic agents. Significant sympathetic blockade and postural hypotension may occur as a result of the physiologic response to injection. Another possible undesirable effect is sexual dysfunction in male patients, although this may also be caused by vascular insufficiency, an indication for lumbar sympathetic block in the first place. There remains a possibility that sympathetic blockade of a limb where there is critical fixed stenosis of the arterial supply to one region may vasodilate only the normal vasculature. This will give rise to a “steal” syndrome—deterioration of perfusion to the ischemic area, if there is a fixed inflow .
The most common complication associated with lumbar chemical sympathectomy is genitofemoral neuralgia [36]. The genitofemoral nerve arises from the lumbar plexus at the first lumbar segmental level and passes on the ventral surface of the psoas muscle. It emerges from the anterior aspect to supply the groin and upper thigh. The incidence varies between 5 and 40 % and most cases are transient, lasting less than 6 weeks [37, 38]. A transdiscal approach to lumbar sympathetic block has been advocated to avoid genitofemoral neuralgia because the needle does not pass through the psoas muscle [39].
Ureteric injury is uncommon but can occur following chemical sympathectomy [40]. Whether injury is related to needle trauma or ureterolysis from the neurolytic agents is unclear. Most case reports claimed fluoroscopic confirmation of needle location and delayed presentation of urological symptoms, suggesting that injury is more likely related to the neurolytic agent. This highlights the importance of limiting the amount of neurolytic agents applied .
Intravenous Regional Sympathetic Block
The technique of intravenous sympathetic blockade has been used for treatment of sympathetically mediated pain in the upper limb . The technique is essentially one of perfusion of the isolated limb with a sympatholytic solution. After an interval of 20–30 min, when a significant portion of the drug is assumed to have become fixed to the tissues, the tourniquet is deflated. The block is repeated, often weekly, for three to six times. This method of sympathetic block is becoming unpopular due to the lack of support of efficacy from the literature [41].
Sympatholytic agents used for intravenous regional sympathetic block are guanethidine (not available for this use in the United States), bretylium, reserpine, phentolamine, and ketanserin. Guanethidine is an agent that blocks reuptake of noradrenaline in sympathetic nerve endings for up to 3 days, thus depleting the stores. It should not be used in patients on monoamine oxidase inhibitors for this reason, as there is an initial release of amine from the stores. Guanethidine is usually used in a dose of 10–20 mg in up to 40 mL of saline or dilute local anesthetic for the upper limb. The dose and the volume are generally higher for the lower limb. The rationale for using local anesthetic in the mixture is that there is less pain at the initiation of the injection. However, local anesthetics can reduce the sympatholytic actions of guanethidine [42].
Complications
Drug Effects
Despite the relative simplicity of the technique, there is a risk of unwanted systemic absorption if the drug bypasses the inflated tourniquet or following deflation. Transient decrease in blood pressure on tourniquet release is common [43], although Sharpe et al. reported prolonged hypotension (80 mmHg for 1 week) can occur after repeated blocks [44]. Autonomic denervation due to drug accumulation may be responsible for the prolonged hypotension.
Other adverse events following cuff deflation were transient apnea and syncope during an intravenous regional anesthesia using guanethidine and lidocaine [45]. Whether this neurologic event was due to hypotension or drug toxic reaction is unclear. Seizures following cuff deflation have been reported with a tourniquet time of as long as 60 min with a lidocaine dose as low as 1.5 mg/kg. Compartment syndrome has also been reported [46].
Visceral Nerve Blocks
Celiac Plexus Block
The celiac plexus innervates the upper abdominal viscera, including pancreas, diaphragm, liver, spleen, stomach, small bowel, ascending and proximal transverse colon, adrenal glands, kidneys, abdominal aorta, and mesentery. It contains preganglionic splanchnic afferent, postganglionic sympathetic fibers, and parasympathetic fibers (Fig. 16.6). Celiac plexus blockade may therefore be indicated in chronic or cancer pain involving one of these organs, the pancreas and stomach being the most common.
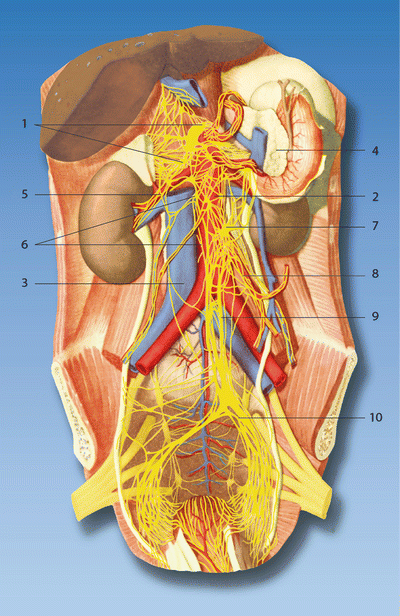

Fig. 16.6
Anatomy of the neural plexus to the visceral organ . (1) Celiac plexus, (2) aorta, (3) inferior vena cava, (4) pancreas, (5) renal plexus, (6) abdominal aortic plexus, (7) inferior mesenteric ganglion, (8) inferior mesenteric plexus, (9) superior hypogastric plexus, (10) inferior hypogastric plexus. Reproduced with permission from Dr. Danilo Jankovic
The greater (T5–10), lesser (T10–11), and least (T12) splanchnic nerves form the preganglionic sympathetic supply for celiac ganglia. These nerves lie on the thoracic paravertebral border, pierce the diaphragmatic crura, and form the plexus lying on the anterior and lateral aspects of the abdominal aorta, between the origins of the celiac arterial axis and the renal arteries. The celiac ganglia number between one and five and may be up to 4.5 cm in diameter .
Four techniques of blocking the splanchnic nerve and celiac plexus are commonly used. The first is the retrocrural splanchnic nerve block technique. The needles, one on each side, are placed posteriorly and paravertebrally below the 12th rib and advanced medially to make contact with the L1 vertebral body. With this approach, the aim is to position the needle tip close to the splanchnic nerves behind the aorta and the diaphragm. A modification of this classical retrocrural technique is to direct the needle more cephalad at the level of the anterolateral margin of T12 vertebra. Theoretical advantage of this modification is to block the visceral sympathetic pathway more effectively with a smaller volume of neurolytic solution. The second approach is the transcrural technique, which aims to block the celiac plexus proper by positioning the needles (one from each side) farther anterior and through the diaphragmatic crura. Under radiologic guidance, injectate is deposited anterior and caudal to the crura and posterior to the aorta. A smaller volume of drug is required, thus minimizing the risk of somatic block. The third approach is the transaortic approach developed by Ischia using a single needle from the left side of the back [47]. The advantages of this technique are a single needle insertion and a smaller dose requirement of local anesthetic or neurolytic agent, resulting in a lower risk of retrocrural somatic spread. However, there is a slightly higher risk of hematoma formation. The fourth approach is a percutaneous anterior approach. Fine needles guided by ultrasound may be used [48]. Visceral or vascular perforation can occur, but the sequelae of perforation may be minimized by antibiotic coverage and avoidance of the technique in “coagulopathic” patients. Celiac plexus block can also be performed under direct vision following a laparotomy. Alternatively, endoscopic ultrasound-guided (EUS) injection is a safe and cost-effective approach [49]. With an ultrasound transducer mounted in front of the viewing lens of the endoscope, the aorta and celiac artery can be easily identified as reference landmarks prior to injection .
Celiac plexus block is considered to be a relatively safe procedure; however, the possibility for serious complications exists. In many clinical contexts in which this block is offered (e.g., intra-abdominal malignancy), the analgesic benefit is considered to outweigh these risks .
Complications
Hypotension
Because of the sympathetic blockade of splanchnic vasculature, the most common complication of celiac plexus blockade is hypotension. Without adequate prehydration or vasopressor drugs, this may occur in 30–60 % of patients. There is some evidence that the incidence of hypotension is higher with a retrocrural approach [50]. In a meta-analysis of neurolytic celiac plexus blocks, Eisenberg et al. report 10 studies covering 571 patients of whom 217 (38 %) had hypotension [51]. Splanchnic vasodilatation and visceral blood pooling contribute to orthostatic hypotension. Providing an intravenous fluid bolus prior to the procedure can minimize the incidence of hypotension. It is recommended that blood pressure and an electrocardiogram (ECG) be monitored for 2 h after a block. Patients should remain supine or in the lateral position for at least 1 h postprocedure, or until they can stand unaided. In approximately 3 % of patients, orthostatic hypotension may persist for up to 5 days [52].
Diarrhea
Unopposed parasympathetic activity following celiac plexus block can lead to gastrointestinal hypermotility [53]. Additionally, after a successful celiac plexus block the patient will need smaller doses of opiate analgesics. Diarrhea is usually transient, but may sometimes develop a chronic pattern. The incidence of transient diarrhea is approximately 40 % [54]. When diarrhea occurs in the presence of preexisting dehydration and pooling of blood in the splanchnic circulation, life-threatening hypovolemia may appear if massive intestinal fluid loss is not replaced. Somatostatin has been suggested as therapy in this situation, and octreotide may have a role in treatment of persistent diarrhea.
Needle Trauma
Needle puncture and drug injection into the aorta, vena cava, renal vessels, and various viscera have been reported [55]. The anatomy may be distorted by tumor or other mass in the retroperitoneum or abdomen. One expects the risk of hematoma formation to be highest with Ischia’s transaortic approach . Aortic puncture is more likely with needle placement on the left side than on the right side. A large retroperitoneal hematoma following vascular puncture may cause hypovolemia and must be differentiated from hypotension due to splanchnic vasodilatation. Limiting the size of the needle and ensuring normal patient coagulation status will reduce the risk of bleeding.
Aortic dissection after formation of an infected pseudo aneurysm has been reported after celiac plexus block, possibly related to the effect of neurolytic agent on the aortic wall [56]. Kaplan et al. report fatal aortic dissection, which extended to the superior mesenteric and hepatic artery, resulting in extensive liver and bowel infarction [57]. Other vascular complications include phlebitis, vessel thrombosis, and vasospasm.
Unintentional injection between vertebrae producing an incidental discogram was reported by Wilson [58]. Pneumothorax is another theoretical complication, even though the point of needle insertion is below the 12th rib. Chylothorax has been reported in association with tumor and after puncture of the cisterna chyli during celiac plexus block [59]. The cisterna chyli classically lies anterior to the first two lumbar vertebrae to the right of the aorta, but this is variable. The transdiaphragmatic movement of the retroperitoneal lymph collection is via lymphatic. Retroperitoneal fibrosis after multiple blocks has been reported [60].
Infection
Because of the proximity of the needle path to the bowel, especially with the anterior and EUS approach, infection is a concern. In a series of 90 patients, only 1 patient developed an infectious complication, a peripancreatic abscess, which resolved with a short course of antibiotics [61]. Retroperitoneal abscess has also been reported [62].
Neurologic and Neurovascular Sequelae
The most serious complications of celiac plexus block are neurologic; however, the overall incidence of major neurological adverse events is low [63, 64]. There are several mechanisms of injury. Drug misplacement and anomalous or excessive retrocrural spread can affect epidural and lumbar somatic nerve roots. Direct accidental intrathecal injection can also occur, which could lead to permanent paraplegia. Permanent and extensive autonomic blockade may cause male sexual dysfunction.
The arterial supply to the spinal cord may be damaged during celiac plexus block. The anatomy of the blood supply is variable, and the major radicular artery of Adamkiewicz may arise from T7 to L4. In 80 % of patients, this vessel lies on the left. It enters via a single intervertebral foramen to supply the anterior spinal artery of the lower two-thirds of the cord. Damage to this artery (either mechanical by a needle or chemical by neurolytic drug) may lead to paraplegia. Although radiologically guided techniques minimize the incidence of direct intravascular injection, neurolytic drugs deposited perivascularly may alter arterial reactivity and cause vasospasm. This has been demonstrated in isolated canine lumbar arteries in vitro [65]. Injury to artery of Adamkiewicz due to compression, spasm, or both can lead to anterior spinal artery syndrome [66]. There is a possibility that using only a right-sided approach might lessen the incidence, but it might also diminish the effectiveness.
Drug Effects
Phenol-induced cardiotoxicity may account for a report of cardiac arrest in a patient undergoing intraoperative splanchnic nerve block during laparotomy [67]. Ventricular fibrillation occurred 3 min after injection of 30 mL of 6.66 % phenol, after negative aspiration under direct vision. The authors cite other reports of cardiac toxicity of phenol, mostly arising from transdermal absorption in dermatologic and plastic surgical practice, where much higher doses are used. In the case of celiac plexus block, cephalic spread of the neurolytic agent may result in involvement of the cardiac nerves and plexus, which may in turn affect the heart and surrounding thoracic structures [68].
Systemic effects have been reported as a result of absorption of a large volume of alcohol administered for retrocrural celiac plexus block. Measured serum ethanol concentration was up to 39 mg/dL after injection of 25 mL of 50 % ethanol bilaterally and 29 mg/dL after 15 mL of 99.5 % ethanol [69, 70]. Although this will not cause any serious impairment and is below the legally defined limit for intoxication, the authors noted that all patients reported a feeling of mild euphoria. However, toxic alcohol levels may appear in patients who have a genetic deficiency of aldehyde dehydrogenase, which is relatively common in the Japanese population. There is also a possibility of interaction with drugs such as disulfiram or metronidazole, although this has not so far been reported.
In summary, the retrocrural technique has the lowest risk of visceral or vascular puncture, but a higher risk of somatic nerve block due to a larger volume of drug. Transcrural injection requires smaller volumes but has a slightly increased risk of perforation of vital visceral structures. Transaortic celiac plexus block, a single-needle technique, uses the least amount of drug but most likely causes vascular damage and hematoma formation even with a fine needle .
Pelvic Visceral Nerve Blocks
The superior hypogastric nerve and the ganglion impar are two sites amenable to blockade for chronic or cancer pain of the lower abdominal or pelvic organs [71]. The superior hypogastric plexus is found on the anterior aspect of the sacrum, in the midline (Fig. 16.6). Approach to the superior hypogastric plexus is percutaneous, from a point between the sacral ala and the interspace between the L5 and S1. The needle passes anteromedially to the anteriolateral aspect of the L5–S1 area. A transdiscal technique has been described (Fig. 16.7) [72]. The ganglion impar (or ganglion of Walther) lies on the concavity of the sacrum and is blocked percutaneously using a specially bent needle inserted toward the sacrococcygeal junction. The transdiscal technique is gaining popularity because of easier access to the target site without bending the needle (Fig. 16.8) [73].
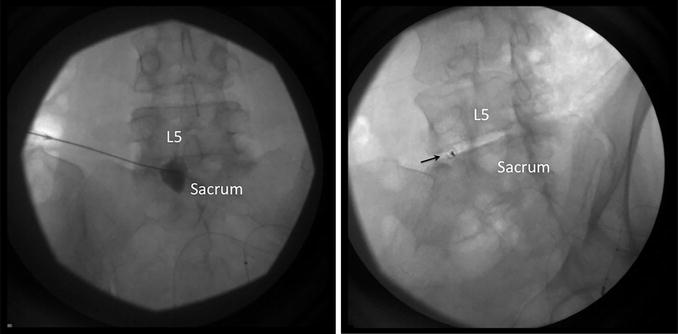
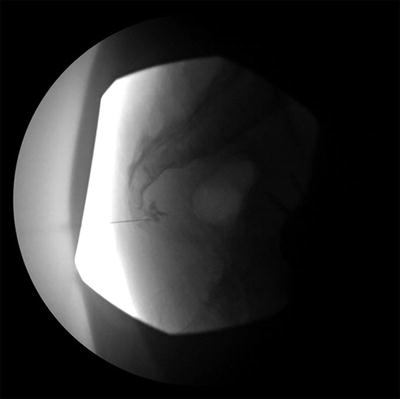

Fig. 16.7
(a) The image on the left is an anteroposterior radiograph of the lumbosacral junction. The needle was seen inserted to the anterior aspect of the lumbosacral junction with contrast confirmation. (b) The image on the right is an oblique radiograph of the lumbosacral junction. The spinal needle (indicated by the arrow) was inserted with transdiscal technique evident with the end-on view. Thus, only one needle inserted was required as this technique allowed the needle to reach the anterior aspect of lumbosacral junction. Reproduced with permission from Philip Peng Educational Series

Fig. 16.8
Ganglion impar injection . The needle was inserted via the sacrococcygeal disc space. The puncture needle was initiated by the introducer needle through the skin and then a 25 G spinal (Whitacre) needle was inserted through the introduced needle to minimize the risk of discitis. The needle position was further confirmed, with contrast (with permission from Philip Peng)
Despite the presence of a number of case series, there are no recent reports of complications from the superior hypogastric plexus block [74]. Intravascular injection can easily occur due to the close proximity of the iliac vessels. Although there are limited reports of complications with the transdiscal technique, careful technique should be used to minimize the risk of discitis, disc herniation, or disc rupture [75, 76]. Strict sterile technique should be used, and some practitioners advocate for the use of periprocedural antibiotic prophylaxis to minimize the risk of infectious complications.
Regarding the ganglion impar block, Plancarte et al. published a case report in which epidural spread of contrast material was demonstrated within the caudal canal [71]. However, no adverse effects resulted from this. Overall, the ganglion impar block is considered to be a safe procedure with no long-term complications being reported.
Somatic Nerve Blocks
Facet Joint Block
The lumbar facet (zygapophyseal) joint has long been considered by some to be a significant source of low back pain whereas cervical facet joint disease is linked to chronic neck pain [77]. The facet or zygapophyseal joints are true synovial joints with considerable sensory innervation and overlap. The medial branch of the posterior ramus supplies the lower pole of one facet joint and the upper pole of the adjoining facet joint .
A diagnostic facet joint injection or a medial branch block may be considered in patients with back or neck pain. Real-time fluoroscopic- or ultrasound-guidance is recommended to ensure accurate needle placement because off-target injection by a few millimeters can result in aberrant drug spread to intervertebral neural foramen or the epidural space, yielding false positive result of pain relief [78]. Injection of contrast material (0.3 mL) can enhance accuracy and injection of a small volume of local anesthetic (0.5–1 mL) will decrease the risk of spread to the epidural space or somatic nerves. In the neck, the vertebral artery lies just lateral to the facet joint, thus intravascular injection or damage is known risk .
Complications
Increased Pain
Transient increased pain is the most common side effect (2–20 %), which may last from 6 weeks to 8 months [79].
Infection
Intraspinal Injection
Configuration of the facet joints in the lumbar spine (oblique orientation with a curved shape) largely prevents needles from entering the vertebral canal; however, spinal anesthesia following attempted lumbar facet block has been reported [86]. These cases may be due to erroneous needle placement, possibly through a nerve root dural cuff.
Thomson et al. report chemical meningism after attempted facet joint block with local anesthetic and steroids, and this was presumably caused by inadvertent intrathecal injection, since there are very few reports of meningism associated with epidural injection of steroids [87]. Spinal cord injury during attempted cervical facet joint injection has also been reported [1].
Other Complications
Excessive local anesthetic injection and spread to the somatic roots can cause ipsilateral weakness, although we have found no recent reports of this obvious complication. This may be caused by needle placement too anterior or excess volume causing joint rupture. It should be remembered that the maximum volume of the facet joint is 1.0 mL. Pneumothorax during attempted thoracic facet joint injection has been reported [1]. Overall, the facet nerve block is a very safe procedure if one follows the recommended technique and aseptic protocol [80].
Facet Joint Radiofrequency Denervation
Radiofrequency (RF) neurotomy interrupts nociceptive pathways by applying heat (75–800 °C) from the tip of an electrode to denervate nerves. This technique is used for treatment of trigeminal neuralgia , dorsal rhizotomy , and dorsal root entry zone (DREZ) interruption for deafferentation syndromes [88]. Radiofrequency procedure can also be used for facet joint denervation in the lumbar and cervical regions. The complications are commonly secondary to direct needle trauma, needle misplacement during lesioning, and to low-level heat injury to the nerve .
Complications
Various side effects have been reported for cervical radiofrequency neurotomy, including postoperative pain, ataxia, denervation sensitivity, and vasovagal syncope [89, 90]. Ataxia is most common when the third occipital nerve is treated. The third occipital nerve carries a large proportion of fibers that provide cutaneous, innervation, and as a result postprocedural numbness can occur, which often leads to temporary dysesthesia and pruritus after 1–3 weeks. These side effects are relatively common and predictable outcomes that are attributed to destruction of the target nerve, hence these outcomes are not commonly referred to as complications [91].
More serious complications that arise from cervical RF neurotomy are rare but have been reported. Many of these complications have occurred during RF techniques that were performed under general anesthesia when the patient was unable to report adverse symptoms. These complications can be devastating and range from misplacing electrodes adjacent to the spinal cord during lesioning (leading to direct spinal cord injury) to thermocoagulation of reinforcing radicular vessels leading to spinal cord infarction [91].
Fluoroscopically guided percutaneous radiofrequency denervation of the lumbar facets is commonly used as a treatment for chronic low back pain. Adverse effects in this region of the spine are less common compared to cervical RF neurotomy. Complications occur in approximately 1 % of patients and present mostly as localized pain or neuritis pain in the first 2 weeks after treatment [92]. Cutaneous numbness and dysesthesia can also occur but usually resolve within 3 weeks [88]. Severe nerve injury, such as injury to the spinal nerve or ventral ramus, is rare but has been reported [91, 93]. This complication results in denervation of the entire associated dermatome and myotome of that segment in the lower limb. Other complications that have been reported include superficial burns due to insulation breaks in the electrodes, as well as burns at the site of the grounding pad adhesion due to generator malfunction [94, 95].
Pulsed radiofrequency is emerging as a popular technique in the management of neuropathic pain. This technique does not appear to be neurodestructive [96], and there is little published evidence demonstrating its efficacy [88]. Consideration for complications due to pulsed radiofrequency will not be addressed in this review .
Epidural Blockade
The epidural space may be approached in the cervical, thoracic, lumbar, or sacral regions (via the sacral hiatus). The most frequently injected agents are steroids and dilute local anesthetics, although opioid has been used in some circumstances [97, 98]. These injections are used with increasing frequency in the management of chronic spinal pain and radiculitis [99]. The transforminal approach to the epidural space has become popular in recent years because it has proven clinical efficacy over conventional techniques [100]. The major advantage of this approach is drug delivery directly to the site of nerve root impingement as opposed to only a fraction of the injected dose reaching target with the conventional interlaminar approach [101].
In 2004, the American Society of Anesthesiologists Closed Claims Study identified major complications associated with epidural steroid injections that resulted in malpractice claims [1]. Complications specific to steroid injections can relate to local or systemic drug effects. The mechanical and traumatic complications can also occur; however, these complications are similar to any epidural injection and will be discussed elsewhere. The Closed Claims Study did not specify complications that may occur according to the levels and techniques of needle insertion, and there has been a growing concern of neurological complications following the use of the transforaminal approach and injections performed at the cervical level [102]. The former will be discussed separately later, and the complications associated with cervical epidural steroid injection have been reviewed elsewhere [103]. A recent retrospective study examined 4265 ESIs performed in 1857 patients, over a 7-year period which included 161 cervical IL injections, 123 lumbar IL injections, 17 caudal injections, and 3964 lumbar TF injections [99]. While there were no major complications identified, there were 103 minor complications that resulted in an overall complication per injection rate of 2.4 %. In this review, the most common complications were increased pain (1.1 %), pain at the injection site (0.33 %), persistent numbness (0.14 %), and “other” (0.80 %). Complications were less common with a transforaminal technique (2.1 %) compared to an interlaminar approach (6 %). This section will provide an update regarding complications associated with epidural steroid injections .
Complications
Neurotoxicity
Arachnoiditis and aseptic meningitis are direct complications that may result from unintentional intrathecal (not epidural) injection of steroid. The symptoms of arachnoiditis can overlap with the symptoms for which many patients are receiving epidural steroid injections , as it most commonly occurs among patients who have had multiple spinal procedures. Recently, Lima et al. performed a randomized, double-blind controlled trial on dogs that demonstrated intrathecal administration of methylprednisolone was responsible for causing histological changes in the spinal cord and meninges [104]. Findings included meningeal thickening, adhesion of the pia, arachnoid, and dura mater, and nerve roots surrounded by fibrosis. Furthermore, a review by Abram and O’Connor identified 65 published series and 18 case reports in 6947 patients who received one or more epidural steroid injections and 368 patients who received one or more subarachnoid steroid injections [105]. There were no reports of arachnoiditis after epidural injection of steroids when intrathecal injection was excluded, highlighting the importance of using a local anesthetic test dose, and/or fluoroscopy with radiocontrast dye to help minimize the risk of inadvertent intrathecal injection of steroid. Nelson suggested that polyethylene glycol may be the offending agent [106]. However, Benzon et al. found that nerve conduction was affected by polyethylene glycol at concentrations seven times higher than clinically relevant concentrations [107]. Even at higher concentrations, the conduction defects were reversible. There is no definitive treatment for arachnoiditis or aseptic meningitis [108].
Neurologic Injury
Severe neurological injury following cervical, thoracic, or lumbar epidural steroid injections can occur due to direct needle trauma to the spinal cord. This type of injury can occur with any epidural injection and will be discussed elsewhere. Another mechanism of injury relates to the injection of a steroid suspension that results in embolization of end arterioles supplying the spinal cord [109]. The blood supply to the spinal cord comes from a single anterior spinal artery and two posterior spinal arteries. At each vertebral level, radicular arteries from the aorta travel along with the segmental nerve roots into the neural foramen and supply the corresponding nerve roots. Some of these radicular branches contribute to the perfusion of anterior spinal cord by joining the anterior spinal artery. The most important radicular artery supplying the lumbar region is the artery of Adamkiewicz. At the cervical level, the important contributing radicular artery originates between C3 and C8. This type of embolic injury appears to be most commonly associated with injections performed at the cervical level. Following cervical transforaminal injections, fatal anterior spinal artery syndrome [110], massive cerebellar infarct [111], and bilateral complete cortical blindness [112] have all been reported. Transforminal injections performed in the lumbar region carry a lower risk; however, it has been determined that previous surgery at this level may increase the risk of spinal cord infarction [113]. All of the particulate-containing corticosteroid preparations available for use have been found to contain large enough particles to occlude capillaries and arterioles. Dexamethasone is a nonparticular steroid solution that minimizes this risk, however, theoretically may result in a shorter duration of effect due to increased solubility [114].
The rate of unintentional intravascular injection using the transforaminal approach is estimated to be 11 % [115]. It is important to note that the sensitivity of a positive blood aspirate in detecting intravascular injections is only 45 %. According to the Closed Claims Study, spinal cord injury due to infarction appears to be less common than injury due to direct spinal cord trauma [1].
Infection
Until recently, infectious complications following epidural steroid injections were considered to be extremely rare; however, the risk has been highlighted by recent catastrophic events [108]. In 2012 in the United States, an outbreak of fungal meningitis occurred among patients who received an injection containing contaminated preservative-free methylprednisolone acetate [116, 117]. In total, there were 751 cases of fungal infection, resulting in 64 deaths. The majority of these patients received the injection through an epidural or paraspinal route (89 %), with the remainder receiving peripheral joint or other nerve injections. Furthermore, in 2012 there was an outbreak of methicillin-sensitive Staphylococcus aureus (MSSA) infections in Toronto, Ontario, Canada following epidural steroid injections . An investigation concluded that nine patients developed serious infections (meningitis and/or abscesses) over a 4-month period [118].
In theory, the risks of neuraxial infection are increased when faulty aseptic technique or bacteremia is present, as for any spinal injection. However, as per the fungal outbreak in the United States in late 2012, contaminated medication at the time of injection can also be a contributing factor. In theory, the immunosuppressive effects of steroids may increase the risk of an infection. However, epidural abscesses can also occur on an idiopathic basis, in the absence of an intervention, and certain risk factors can increase this incidence. Tang et al. reviewed 46 cases of spontaneous epidural abscess and found that 46 % of these patients were diabetic [119]. In addition to fungal pathogens, common bacterial culprits would include Staphylococcus aureus (likely skin contaminant) [120, 121].
It would seem that, despite the theoretically increased risk of infection, clinical reports do not indicate that there is any greater incidence associated with epidural steroids than with local anesthetic agents alone, provided the same precautions and contraindications are noted. Even allowing for underreporting, the incidence just from published series and reports appears to be less than 0.01 %. With such a low incidence, routine prophylactic antibiotic use cannot be justified as this could lead to development of resistant antibiotic strains [108].
Dural Puncture
The frequency of inadvertent dural puncture in the laboring population ranges from 0.04 to 6 % [122]. However, the incidence of a headache is lower among patient undergoing ESI, likely due to the use of smaller gauge needles, an older patient population, and the use of contrast dye during fluoroscopic guidance. McGrath et al. analyzed 284 IL epidural injections and reported only 1 postdural puncture headache (incidence of 0.004 %) [99]. Proceduralists performing these techniques must be able to identify and recognize various patterns of contrast dye following administration to avoid the direct injection of medication into the intrathecal or subdural space .
Systemic Side Effects of Steroids
Suppression of adrenal cortical response has been reported after oral, nasal, inhaled, and parenteral as well as epidural steroid administration. Cushingoid side effects , including fluid retention, electrolyte imbalance, and fat redistribution, have been reported after epidural steroid injection . Stambough et al. [123] reported a case of hypercorticism after two injections a week apart totaling 160 mg of methylprednisolone acetate while Tuel et al. [124] reported one case following a single cervical epidural administration of 60 mg methylprednisolone acetate. In both cases, return of normal clinical and biochemical functions took weeks to months. Exogenous steroid replacement should be considered for patients undergoing surgery who have had epidural steroids administered within the previous 3 months [125].
Steroid-induced myopathy is characterized by progressive proximal muscle weakness, increased levels of creatinine kinase, and myopathic evidence on electromyography. Iatrogenically induced steroid myopathy (proximal limb ) was reported by Boonen et al. after epidural administration of triamcinolone diacetate [126].
The effects of steroids on glucose levels must also be considered. Steroid administration is known to reduce the hypoglycemic effect of interfere, leading to increased blood glucose levels in diabetic patients [127]. Diabetic patients can be warned about experiencing elevated blood glucose levels (and insulin requirements) for several days after administration of corticosteroids. Even et al. [128] assessed 30 diabetic patients who received an ESI and found elevated blood glucose levels which normalized within 2 days.
Although there is no consensus for the frequency or dose of steroid administration to prevent systemic side effects, it is prudent not to repeat injections within a 4-week interval and to limit the number of epidural steroid injections to three in 6 months, based on human and animal data .
Systemic Side Effects of Epidural Opioid
A review from 2005 found that only 2–10 % of anesthesiologists in North America add opioid to epidural steroid [97]. The addition of epidural morphine to steroid may further relieve low back pain but the associated benefits vary [129, 130]. Most of these early studies added 8 mg of epidural morphine to steroid. However, life-threatening ventilatory depression was noted in 3 of 14 patients who received an admixture of steroid and morphine (8 mg) [131]. While lower dose epidural opioid (e.g., morphine 5 mg) has been used, the effect produces analgesia up to 24 h [132]. The common side effects are pruritus (57–90 %), nausea and vomiting (40–64 %), and urinary retention (20–43 %) [129–132]. Clinicians must carefully weigh the limited benefit of epidural opioid against potential serious risks .
Other Complications
Various minor complications have been reported in different case series (Table 16.1). McGrath et al. identified the most common complications as being increased pain (1.1 %), pain at injection site (0.33 %), and persistent numbness (0.14 %) [99]. Overall, these complications were more common with the TF technique when compared to IL. Other complications identified include a decrease in bone marrow density in postmenopausal women who received a cumulative ESI dose of greater than 120 mg methylprednisolone [133]. A follow-up analysis did not identify an increased incidence of pathological fractures in this population [134]. Case reports exist describing delayed allergic reactions to epidural steroid/local anesthetic [135], persistent hiccup presumably due to systemic effect of steroid [136], and vision loss secondary to retinal hemorrhage [137].
Table 16.1
Complications and side effects of epidural steroid injection (interlaminar and caudal approach): aggregate data from published series
Injection type | Number reported | Complications or side effects | |
|---|---|---|---|
Cervical epidural injections | 1788 | 40 (2.2 %) | |
24 (1.3 %) | |||
Headache [138] | 16 (0.9 %) | ||
10 (0.6 %) | |||
9 (0.5 %) | |||
7 (0.4 %) | |||
Other (fever, insomnia) [138] | 7 (0.4 %) | ||
Cervical subtotal | 123 (6.9 %) | ||
Lumbar, thoracic and caudal epidural injections | 13,233 | 45 (0.34 %) | |
35 (0.26 %) | |||
7 (0.13 %) | |||
6 (0.05 %) | |||
Facial flushing [143]
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

| |||


