Common Nerve Blocks
Nerves send sensory impulses to the brain for interpretation. These impulses travel in a regular pattern when the nervous system is working correctly. When a nerve is injured, this normally controlled transmission of neurologic impulses fails and the nerve fires aberrantly. This aberrant firing is interpreted by the brain as neuropathic pain. Injured nerves also develop an increased sensitivity to routine triggers such as mechanical, thermal, and chemical stimuli thus, not only are damaged nerves sending aberrant impulses interpreted as pain, but also there is an amplification of their activity in response to everyday triggers.
Many people mistakenly think of the term nerve block as a permanent block of a nerve’s function. In clinical practice destruction of the nerve rarely occurs—rather, it is bathed in a local anesthetic solution that eventually wears off. For unknown reasons, pain relief via a nerve block can last longer than the life of the anesthetic injected. Nerve fibers sending pathological painful impulses to the central nervous system (CNS) become “trained” at aberrant transmission, in the same way that an athlete’s muscles become highly trained at performing a task. The intensity and persistence of the pathologic signal is over and above what are appropriate functional levels. In addition, the CNS becomes trained at receiving these painful impulses; this is known as neuroplasticity. Neuroplasticity is the CNS ability to reorganize itself by forming new neural connections in response to new situations or to changes in their environment. Neuroplasticity takes place at both the transmitting and receiving ends, creating a powerful pathologic connection. This maladaptive pain signal can be broken with a nerve block. The block allows the continuous pain feedback loop to be broken, allowing the body to reset itself. The way that I explain this to my patients is that pain is transmitted along the neurologic system, similar to an electrical system. Like any electrical system, things can go haywire. So if an electrical gadget—such as your computer or phone—is not working properly, what is the first thing you do to try to fix it? You turn off and on the power. When the power comes back on, more often than not things return to normal. That is what we are trying to achieve with the nerve block; we are rebooting the system. Before the injection it is essential to discuss with patients; realistic expectations. Nerve blocks do not always provide prolonged pain relief and may need to be repeated to achieve maximal benefit.
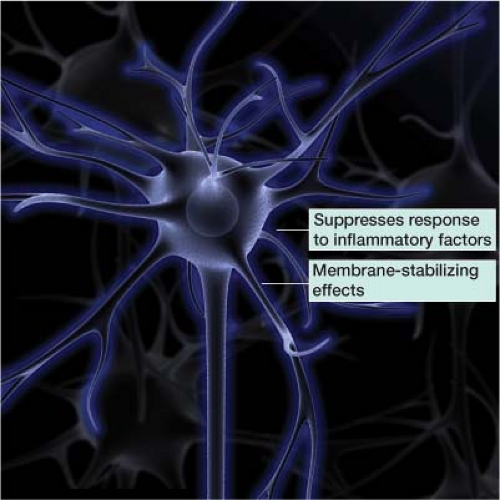
In clinical practice, practitioners often add a steroid to the local anesthetic. The steroid suppresses the targeted nerve’s response to inflammatory factors. This inflammatory response can aggravate a nerve, changing its normal firing pattern. Steroids also exert a membrane-stabilizing effect on injured nerve segments, reducing ectopic discharges from the affected nerve (Fig. 27-1).1,2 The concentrated steroid dose delivered by needle directly to the level of pathology is in much lower quantities than would be needed via oral or intravenous administration.
In most nerve blocks a local anesthetics and steroid are used in combination. The local anesthetic is mixed in the same syringe as the corticosteroid solution. The primary corticosteroids on the market are listed in Table 27-1. There is no one particular steroid and local anesthetic combination that is a consensus choice. I use Kenalog, which is featured in the examples to follow. But if you decide to use Depo-Medrol or any other steroid, proper doses can easily be calculated: For
example, 40 mg of Kenalog is equivalent to 6 mg of Celestone (Table 27-1).
example, 40 mg of Kenalog is equivalent to 6 mg of Celestone (Table 27-1).
Table 27-1 Primary Corticosteroids for and Equivalent Doses | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
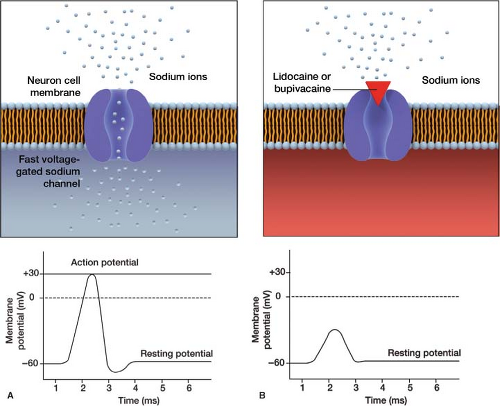
There are primarily two types of local anesthetics used for nerve blocks—lidocaine and bupivacaine—which both work by blocking fast voltage-gated sodium channels (Fig. 27-2). This prevents neuronal depolarization, blocking the painful nerve impulses that are sent to the brain. Dosages of 1% lidocaine and 0.25% bupivacaine are equivalent to each other,
but differ in their time of onset and length of duration (Table 27-2).
but differ in their time of onset and length of duration (Table 27-2).
Table 27-2 Lidocaine Versus Bupivacaine Onset and Duration of Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
|
When to use
Peripheral nerve blocks can be extremely useful in providing pain relief. In some cases, a nerve block alone can provide significant relief. In other cases, a nerve block may allow a patient to fully participate in physical therapy, a key component of pain relief. Physical therapy, which may have been limited previously by intense levels of pain, now becomes a method of treatment.
Peripheral nerve blocks can also be diagnostic, helping to localize the pain generator—for example, if it is unclear if pain in the chest wall is coming from the intercostal nerves or deeper visceral structures. A diagnostic intercostal nerve block can help resolve this question.
How to Perform the Procedure
The procedure is fully explained to the patient, all questions answered, and informed consent obtained. A “time out” is performed prior to the procedure including verbal confirmation of correct patient, procedure and procedural site.
Trigeminal Nerve Block: Ophthalmic (V1), Maxillary (V2), and Mandibular (V3) Branches
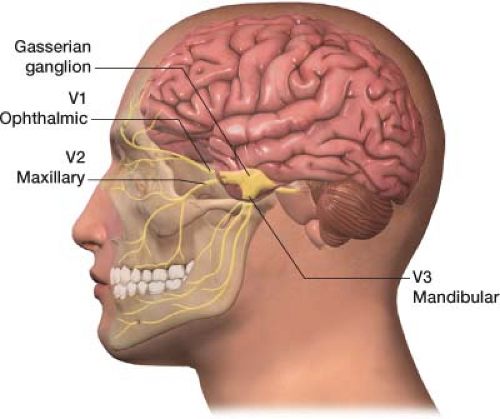
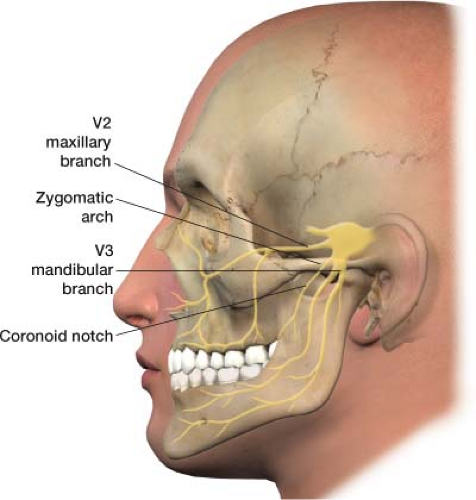
The trigeminal nerve is the fifth cranial nerve, responsible for sensory impulses originating from the face above the jaw line to the forehead. The trigeminal nerve has three branches: Ophthalmic (V1), maxillary (V2), and mandibular (V3) (Fig. 27-3). The ophthalmic, maxillary, and mandibular branches all originate from the base of the trigeminal nerve, the gasserian ganglion (Fig. 27-4). The pain of trigeminal neuralgia is most common in the maxillary (V2) and mandibular (V3) distributions. This sharp, electric pain radiates deep into the cheek, lips, and tongue, typically on one side of the face. It is described as brief episodes of stabbing and shock-like pain. Exacerbations often come in clusters, with complete remission between attacks.
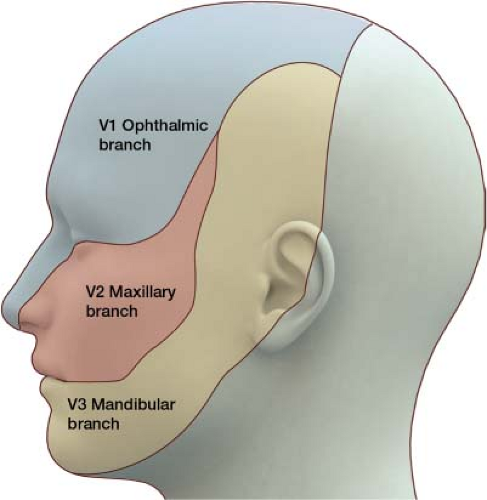 Figure 27-3 The trigeminal nerve has three divisions: Ophthalmic (V1), maxillary (V2), and mandibular (V3). |
Rather than blocking the trigeminal (gasserian) ganglion, usually the branches of the trigeminal nerve that correlates to the patient’s pain are blocked. A trigeminal (gasserian) block is technically challenging and carries substantial risk.3,4 Remember, pain is most common in the maxillary (V2) and mandibular (V3) distributions of the trigeminal nerve in trigeminal neuralgia, thus often only these two branches need to be blocked. These nerve blocks may also be useful for atypical facial pain as well as pain generated by oral/facial cancer.
Trigeminal Nerve Block: Ophthalmic (V1) Branch
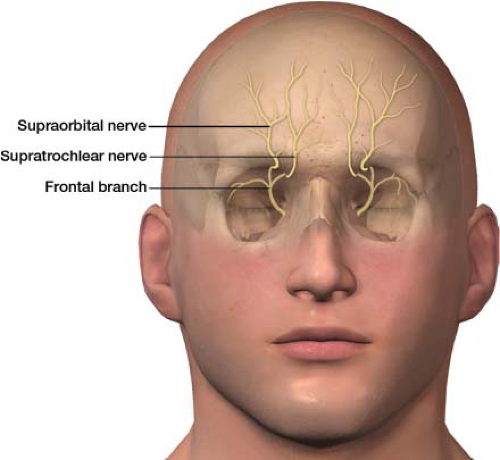
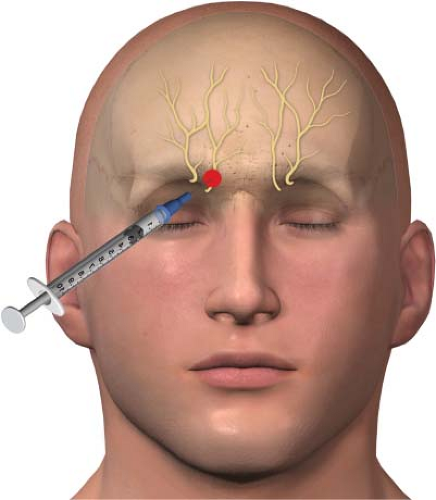
In clinical practice, branches of the ophthalmic (V1) branch of the trigeminal nerve are blocked, rather than the whole nerve, to treat pain around the eyes and nose. The ophthalmic nerve (the first division of the trigeminal nerve) splits into the lacrimal, frontal, and nasociliary branches. The frontal branch further divides into the supraorbital and supratrochlear nerves (Fig. 27-5). The supraorbital and supratrochlear nerves are the target of interest in an upper facial nerve block. The supraorbital nerve supplies sensory innervation to the upper eyelid, forehead, and scalp. The supratrochlear nerve supplies sensory innervations to the upper nose and sends branches to the conjunctivae and the upper eyelid.
Supraorbital and supratrochlear nerve block: The patient is placed in the supine position (face up). The primary landmark will be the bone of the superior orbit, in particular the supraorbital notch. The skin is then prepped with alcohol. A 3-mL syringe is filled with a mixture of a local anesthetic and a steroid; an 18-gauge needle is used to draw up the medication. I use a mixture of 2.5 mL of 0.25% bupivacaine and 20 mg of Kenalog as my therapeutic agent divided equally between the supraorbital and supratrochlear nerves. The 18-gauge needle is removed from the syringe once the medications have been drawn up, and a 1.5-in, 25-gauge needle is placed.
Push the eyebrow superiorly with your nondominant hand, which helps you to palpate the groove of the supraorbital notch, the site of the supraorbital nerve (Fig. 27-6). Using a felt-tip pen, place an “X” on the skin over the supraorbital notch. The needle is then placed through the “X,” aiming at the bone. The needle is advanced till bone is felt, which should be at
a very shallow depth. The needle is withdrawn 1 mm after bone contact. The patient is instructed to verbalize any dysesthesias during the injection to prevent injection into the nerve itself. If dysesthesias are felt, the needle is repositioned prior to injection. After negative aspiration, the therapeutic agent is injected—in this case, 1.5 mL of your mixed local anesthetic and steroid solution.
a very shallow depth. The needle is withdrawn 1 mm after bone contact. The patient is instructed to verbalize any dysesthesias during the injection to prevent injection into the nerve itself. If dysesthesias are felt, the needle is repositioned prior to injection. After negative aspiration, the therapeutic agent is injected—in this case, 1.5 mL of your mixed local anesthetic and steroid solution.
The supratrochlear nerve can be accessed by redirecting the needle to the upper medial aspect of the superior orbital bone. This site is just lateral to the base of the nose (Fig. 27-7). By using a single entry site to block both nerves, the procedure is both more efficient and more comfortable for the patient. The same steps are performed as just described regarding injection of the therapeutic agent after negative aspiration. The needle is withdrawn and a bandage placed if needed.
Trigeminal Nerve Block: Maxillary (V2) and Mandibular (V3) Branches
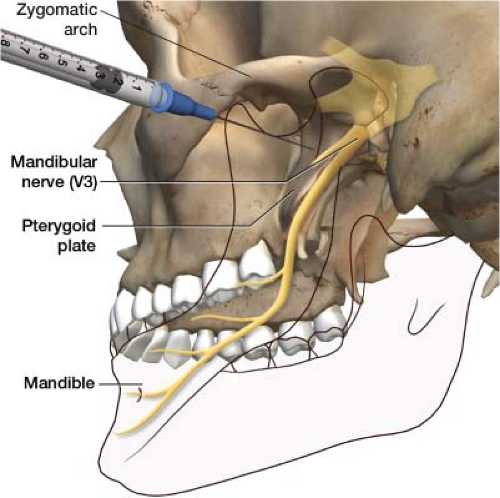
The coronoid notch approach can be used to block both the maxillary (V2) and mandibular (V3) branches of the trigeminal nerve (Fig. 27-8). The patient is positioned on the fluoroscopic table in a supine position (face up). There is a slight groove under the zygomatic arch, the coronoid notch, which is your entry point. This can be located by palpation. After the notch is identified, the patient is asked to hold the mouth slightly open. Using a felt-tip marking pen, place an “X” just inferior to the zygomatic arch (Fig. 27-9). The painful side of the face is draped and prepped in standard sterile fashion. The skin at the “X” is infiltrated with 2 to 3 mL of 2% lidocaine; use a 25-gauge needle to provide comfort during the procedure. The local anesthetic can be
drawn up with an 18-gauge needle before switching to a 25-gauge needle.
drawn up with an 18-gauge needle before switching to a 25-gauge needle.
 Figure 27-9 “X” on the skin showing needle entry site for a maxillary (V2) and mandibular (V3) nerve block, the coronoid notch approach. |
The fluoroscope is placed in the anteroposterior position. A 22-gauge, 2.5-in needle is advanced under fluoroscopy through the “X” in a perpendicular plane to the entry point on the skin till it comes in contact with the pterygoid plate, usually around 1.5 in deep (Fig. 27-10). This is the site of the mandibular nerve. After bone contact is made, the needle is withdrawn 1 to 2 mm. A 3-mL syringe with tubing containing contrast is connected to the needle. Tubing is used to enable you to see the contrast without having your hand obscure the image and protects your hand from radiation exposure. Inject the contrast, making sure it spreads properly (Fig. 27-11). Once proper contrast spread is seen, the 3-mL syringe containing the contrast is disconnected from the tubing. A 10-mL syringe with the therapeutic agent is connected to the tubing. I use a mixture of 2 mL of 0.25% bupivacaine and 20 mg of Kenalog as my therapeutic agent per division of the trigeminal nerve. Thus, if you are blocking both the maxillary (V2) and mandibular (V3) divisions, you would draw up 4 mL of 0.25% bupivacaine and 40 mg of Kenalog, dividing them equally over both nerves. The therapeutic agent is best drawn up before the procedure is started and placed on the sterile field, ready to go when needed. After negative aspiration, the therapeutic agent is then injected on to the mandibular nerve.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree