IV: MANAGEMENT OF COMMON OBSTETRIC CONDITIONS THROUGHOUT PREGNANCY |
|
Common General Surgical Emergencies in Pregnancy | 21 |
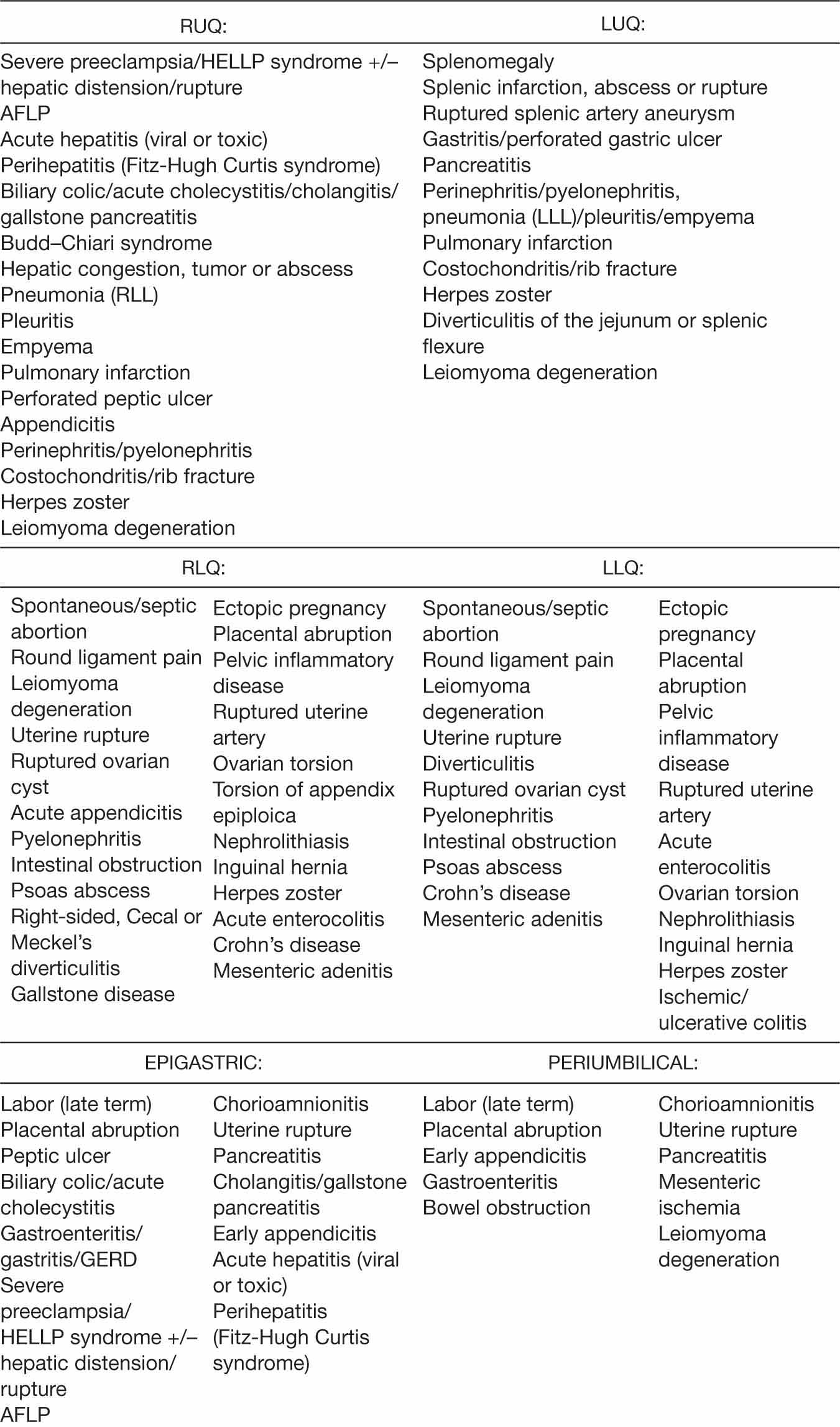
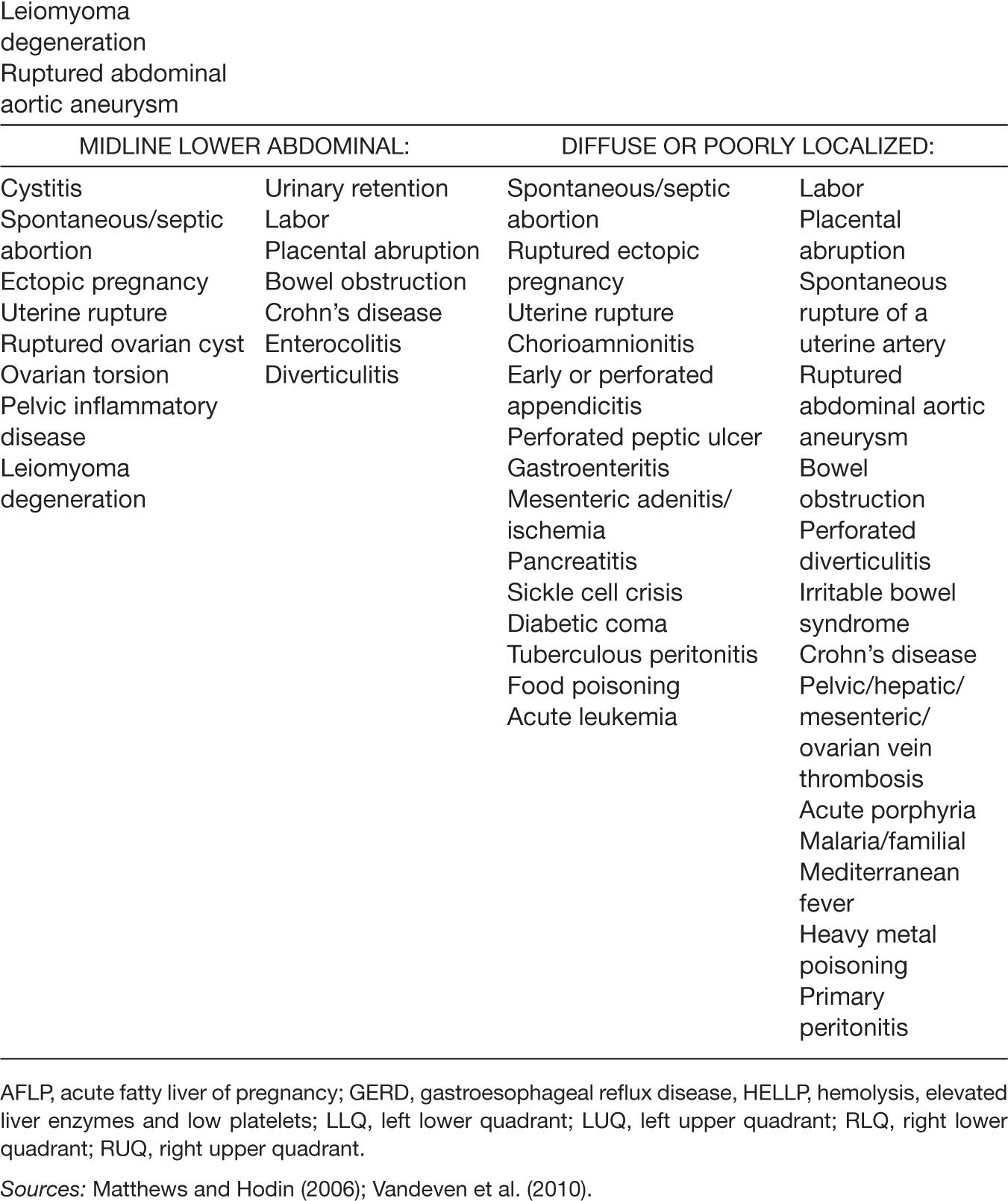
Abdominal discomfort is a common presenting complaint in pregnancy, and often has a benign, physiologic cause. However, severe pain, or pain associated with peritoneal signs on physical examination, is not considered to be normal in pregnancy. The differential diagnosis of such pain in a pregnant woman is extensive, as shown in Table 21.1, and includes both obstetric and nonobstetric conditions.
Emergent general abdominal surgery is performed in approximately 0.2% of pregnant women, most commonly for acute appendicitis, acute cholecystitis, and bowel obstruction (Gilo, Amini, & Landy, 2009). Following a brief discussion of general surgical considerations during pregnancy, each of these conditions is reviewed in detail.
GENERAL SURGICAL CONSIDERATIONS DURING PREGNANCY
Several considerations need to be taken into account during the evaluation, diagnosis, and treatment of a pregnant woman for any disorder that may require emergent surgical intervention. Foremost are the anatomic and physiologic changes that take place in a woman’s body as a result of being pregnant. These changes may alter the presentation of a given disease or make a certain diagnosis more or less likely. Obstetric complications, when present, may have an effect on these factors as well. In addition, the well-being of the fetus must be addressed throughout the encounter as well as during any surgery that is deemed necessary. This can be challenging, especially as a delay in diagnosis or treatment can jeopardize the health or life of the mother, fetus, or both.
Physiologic Leukocytosis of Pregnancy
A complete discussion of the pregnancy-induced anatomic and physiologic changes can be found in most general obstetric texts. The changes that are relevant to the evaluation of each surgical condition reviewed in this chapter are highlighted in the respective discussions. However, the physiologic leukocytosis of pregnancy deserves special mention, as knowledge of this change is relevant to all three conditions and to the evaluation of acute abdominal pain in pregnancy, in general. The normal white cell count in a pregnant woman ranges from 6,000 to 13,0000 cells/mm3 in the first and second trimesters, up to 16,000 cells/mm3 by term, and may reach the 20,000 to 30,000 cells/mm3 range during labor (Vandeven, Adzick, & Krupnick, 2010). This contrasts with the normal nonpregnant white cell count, which ranges from 4,000 to 11,000 cells/mm3. This fact complicates the interpretation of an elevated white cell count in pregnancy, and may require increased reliance upon other elements in the evaluation of a pregnant woman presenting with acute abdominal pain.
TABLE 21.1 Differential Diagnoses for Acute Abdominal Pain in Pregnancy by Location
Diagnostic Imaging Considerations
When diagnostic imaging is indicated in a pregnant woman, the safety of fetal radiation exposure must be weighed against the risk to the pregnant woman 234or fetus of a delayed diagnosis. In most instances, the latter is greater. The most commonly used diagnostic imaging studies expose the fetus to significantly less than 5 rad of ionizing radiation, below which there is no evidence for an increased risk of pregnancy loss, fetal anomalies, fetal growth restriction, or developmental delay of the child (American Congress of Obstetricians and Gynecologists [ACOG], 2016). For instance, a routine chest radiograph exposes the fetus to less than 1 millirad (mrad), while an abdominal flat plate exposes the fetus to 140 mrad. Both are considered acceptable in pregnancy.
Graded compression ultrasonography (US) is the initial imaging modality of choice in pregnant women. This is because it involves no discernable radiation exposure, employs no contrast agents, and there has been no documentation of any biologic effects to mother or fetus over a long history of use (ACOG, 2016; Long, Long, Lai, & Macura, 2011). In addition, results are rapid, and additional pelvic pathology can be detected.
When US is not sufficient to make an accurate diagnosis, noncontrast magnetic resonance imaging (MRI) is the next recommended study. It utilizes the magnetic properties of tissues to create images, and therefore, like US, involves no fetal exposure to ionizing radiation. Gadolinium, the contrast agent commonly used for MRI, crosses the placenta and is excreted into the amniotic fluid by the fetus. Though no adverse effects to the fetus or to the pregnancy have been reported when administered, data are sparse, and the potentially long half-life of gadolinium once it enters the fetal circulation is concerning. In addition, MRI has been shown to be highly accurate without contrast; therefore, it is not used in pregnancy (Long et al., 2011). In addition, as with US, additional pelvic and lower abdominal pathology can be detected simultaneously.
Although the fetus is exposed to ionizing radiation and iodinated contrast during a computed tomography (CT) scan (approximately 20 mrad for a chest CT with abdomen shielded, 150–200 mrad for an abdominal CT with uterus shielded, and 2 rad for a pelvic CT), protocols that decrease this exposure without affecting performance are available and advocated for pregnant women (Long et al., 2011; Vandeven et al., 2010). Additional pelvic and abdominal pathology can be detected as well, though it can take up to 2 hours to administer oral contrast.
Timing of Surgical Intervention
Urgently indicated surgery is acceptable in a pregnant woman regardless of trimester. Alternatively, elective surgery is best deferred until the postpartum period. Per the most recent ACOG Committee Opinion (ACOG, 2015), indicated but nonurgent surgery in pregnant women is best deferred to the early to mid-second trimester. Erroneous causal associations between the surgery and the frequent adverse pregnancy outcomes seen in the first and third trimesters are thereby avoided. These potential outcomes include spontaneous abortion (33%) and preterm contractions/labor (9%), respectively. In addition, by following these guidelines, the fetus would not be exposed to potentially harmful anesthetic agents during the period of organogenesis. However, it is important to note that standard concentrations of commonly used anesthetic agents have not been shown to have teratogenic effects in humans regardless of gestational age (ACOG, 2011). Furthermore, the gravid uterus may obliterate the operative field in the third trimester and make surgery more technically difficult.
ACOG also recommends that when surgery is required during pregnancy it should be performed at an institution where the fetus can be monitored as appropriate, and where an obstetric provider is available, able to interpret the 235results, and able to intervene by performing an emergency cesarean section intraoperatively if indicated. Informed consent for emergent delivery on the part of the pregnant woman is essential when possible. Finally, neonatal and pediatric services are required in the event that the fetus is delivered and is viable (ACOG, 2011).
Route of Surgery
Laparoscopic surgery is considered safe in pregnancy and is therefore a viable alternative to laparotomy for surgical emergencies (Spight, Hunter, & Jobe, 2015; Vandeven et al., 2010). In general, the type of surgery chosen is based upon the skills of the surgeon, the availability of equipment and staff, the gestational age at the time of surgery, and the usual patient characteristics taken into account when planning laparoscopic surgeries on nonpregnant individuals, including body mass index and airway access. Open entry techniques are preferred, and dependent positioning is advised in order to shift the uterus off of the inferior vena cava. Lower insufflation pressures are advocated than those used for nonpregnant women, though it has been shown that the intrauterine pressure created by a pneumoperitoneum of 15 mmHg is much less than that found with mid-pregnancy, nonlaboring uterine contractions (Spight et al., 2015). Overall, despite the fact that some adverse fetal outcomes have been reported with negative diagnostic surgeries (Ito, Ito, Whang, & Tavakkolizadeh, 2012; Liang, Anderssen, Jaffe, & Berger, 2015; McGory et al., 2007), the rates of most complications for common nonobstetric surgeries performed via either laparoscopy or laparotomy in pregnant women are not increased above those in nonpregnant women (Moore et al., 2015; Silvestri et al., 2011; Vandeven et al., 2010).
Fetal Monitoring During Surgery
Hemodynamic stability of the mother during surgery does not necessarily imply adequate placental perfusion or fetal oxygenation. When possible, intraoperative fetal monitoring can identify the need for changes in maternal positioning and/or cardiorespiratory status in order to maintain fetal well-being. Therefore, it is recommended by ACOG that a fetal heart rate be documented for a previable fetus both before and after the procedure. Continuous fetal monitoring is recommended for a viable fetus if all of the previously listed general requirements for surgery during pregnancy apply, and if the surgery itself is amenable to safe interruption or modification in order to allow for an emergent delivery. If all of these conditions are not met, then electronic fetal heart rate and contraction monitoring are advised immediately before and after the procedure, to allow for documentation of fetal well-being as well as the presence or absence of contractions (ACOG, 2011).
Anesthesia
A full discussion of the issues encountered by the anesthesiologist in managing pregnant women is outside the scope of this chapter. Briefly, the risks and benefits of the type of anesthesia and individual medications chosen are carefully weighed in conjunction with current obstetric and pediatric knowledge in preparing for the indicated surgery. When possible, regional anesthesia is 236preferred, as it eliminates many of the risks and problems encountered with general anesthesia, including difficult/failed intubation and oxygen desaturation. Aspiration prophylaxis with sodium citrate or other medications is generally administered prior to the procedure regardless of the type of anesthesia chosen. Intravenous, inhalation, and neuromuscular blockade medications are titrated to effect, given the potential for pregnancy to alter patient sensitivity to these agents. And, most importantly, maternal hypotension, possible with any type, is avoided as best as possible in the interest of maintaining uterine blood flow (American Society of Anesthesiologists Task Force on Obstetric Anesthesia [ASATFOA], 2016).
Additional Recommendations
The following additional recommendations are based upon observational studies, expert opinion, the results of trials in nonpregnant individuals, and a knowledge of the anatomic and physiologic changes that occur in a woman’s body as a result of being pregnant. A woman 18 to 20 weeks pregnant or more is best positioned at a 15% left lateral tilt to prevent the cardiovascular decompensation that may result from compression of the aorta and inferior vena cava by the uterus is a direct supine position. A wedge placed under the woman’s right hip is also acceptable. Antibiotics are administered according to the usual guidelines for the procedure being performed, with attention to and avoidance of medications that have been associated with reported fetal toxicities and teratogenic effects if alternatives are available. Deep venous thrombosis (DVT) prophylaxis with sequential compression devices is implemented during surgery of any type or duration, as pregnancy is a well-known hypercoagulable condition. The decision to use systemic anticoagulants, as usual, is dependent upon the nature of the procedure itself, any additional risk factors of the woman herself (obesity, immobilization, personal or family history, etc.), and the nature of the expected course of recovery.
When surgery is performed prior to 7 to 9 weeks gestation, and a corpus luteum cyst is compromised or removed in the process, progesterone supplementation is indicated up to 9 completed weeks gestational age (Pritts & Atwood, 2002). With surgery at or near viability, tocolytics are not recommended prophylactically, but only for treatment of preterm labor when present. Prophylactic glucocorticoids may be considered for surgeries that occur between 24 and 34 weeks gestation if the underlying process or procedure is thought to place the pregnant woman at increased risk for preterm labor. They are not recommended, however, in cases where an infection has spread systemically, as they may undermine the efforts of the maternal immune system to eliminate the infection.
ACUTE APPENDICITIS
Acute appendicitis is the most common general surgical emergency among pregnant women, and accounts for 25% of nonobstetrical surgery performed during pregnancy (Abbasi, Patenaude, & Abenhaim, 2014). The incidence is approximately 1 in 766 births (Liang et al., 2015), and was previously reported to be on par with the incidence in nonpregnant women. However, a recent cohort study of over 350,000 pregnancies from 1997 to 2012 reported that the diagnosis is actually 35% less likely during pregnancy (Zingone, Sultan, Humes, & West, 2015). The highest rates in pregnant women were found during the second 237trimester, and the lowest were during the third trimester. Appendiceal rupture is more likely when surgery is delayed more than 20 to 24 hours from symptom onset (Abbasi et al., 2014; Bickell, Aufses, Rojas, & Bodian, 2006; Yilmaz, Akgun, Bac, & Celik, 2007). Therefore, it is not surprising that rupture is more likely in pregnant women, upon whom reluctance on the part of surgeons to operate is more likely to delay treatment, especially during the third trimester. A timely diagnosis is crucial, given that elevated rates of adverse outcomes, including maternal sepsis and preterm labor, are seen under these circumstances (Abbasi et al., 2014).
PRESENTING SYMPTOMATOLOGY
The classic presentation of acute appendicitis includes a report of vague periumbilical pain thought to be due to luminal obstruction of the appendix by a fecalith or by lymphoid hyperplasia. This is a referred pain relayed by visceral mechanisms as a result of the increased pressure in the appendix. It is followed by anorexia, nausea, and vomiting, also mediated by visceral mechanisms. As the inflammatory process progresses beyond the appendix itself and affects the overlying peritoneum of the right lower quadrant or pelvis, the pain shifts to that region. This is reported to be the most common presenting symptom of appendicitis in pregnancy (Liang et al., 2015; Mourad, Elliot, Erickson, & Lisboa, 2000; Yilmaz et al., 2007). Fever often follows, generally low grade unless the appendix is perforated. Diffuse abdominal pain is also more common with perforation. Additional symptoms that may be present include urinary complaints, diarrhea, or constipation, all owing to the proximity of the bladder and bowel to the inflamed appendix. Though microscopic hematuria, pyuria, or bacteriuria are present in one third to more than one half of patients with acute appendicitis (Yilmaz et al., 2007), a complaint of frank hematuria is rare.
In the pregnant woman, especially with advancing gestational age, the presentation may be less “classic,” though perhaps not as much as originally thought. Though the growing uterus shifts the location of the appendix a few centimeters superiorly during the pregnancy, the right lower quadrant pain has been found to be in close proximity to McBurney’s point regardless of the gestational age of the pregnancy (Hodjati & Kazerooni, 2003; Mourad et al., 2000). This is in contrast to the classic teaching that the pain is displaced superiorly as well, though some recent smaller studies have reported this to be the case (House, Bourne, Seymour, & Brewer, 2014). In addition, since the uterus lifts the anterior abdominal wall away from the appendix as it grows, thereby preventing direct contact between the area of inflammation and the anterior parietal peritoneum, the presentation of the pain may be significantly less pronounced. Gastrointestinal symptoms may also be more subtle, since the uterus intervenes between the appendix and the bladder and bowel in many cases. Though 58% to 77% of pregnant women with appendicitis report some degree of nausea and vomiting, complaints may consist only of simple indigestion, mild bowel irregularity, or generalized malaise (Vandeven et al., 2010).
HISTORY AND DATA COLLECTION
In addition to the history of the onset and progression of pain and upper gastrointestinal symptoms in a pregnant woman suspected of having acute appendicitis, a complete review of systems is recommended to make an accurate diagnosis. In particular, certain urinary or bowel symptoms may make an alternate 238diagnosis more likely, since pregnancy-induced physiologic changes render these organs highly susceptible to compromise. Inquiry regarding pregnancy and fetal status is also crucial, as a history of vaginal bleeding or abdominal trauma coincident with the onset of pain would clearly increase concern for an obstetric etiology for the presenting symptoms.
In pregnancies that have reached fetal viability, fetal heart rate abnormalities may also increase concern for pregnancy-related causes of abdominal pain, though they can occur with nonobstetric etiologies as well.
PHYSICAL EXAMINATION
Pregnant women with acute appendicitis appear variably uncomfortable, depending how early in the process they present for care. A documented low-grade fever and elevated heart rate, in addition to the common presenting complaints, would support a diagnosis of early acute appendicitis. A fever above 39.4°C (103°F) is especially concerning for a perforated appendix.
As previously mentioned, the abdominal examination findings on a pregnant woman with acute appendicitis may be attenuated by the presence of the gravid uterus. Though the point of maximal tenderness in the right lower quadrant is still consistently close to McBurney’s point (1.5–2 inches from the anterior superior iliac spine in the direction of the umbilicus), rebound and guarding are inconsistently found (Vandeven et al., 2010). Additional peritoneal signs that are less sensitive and variably influenced by the intervening uterus include Dunphy’s sign (increased pain with coughing or movement), Rovsing’s sign (pain in the right lower quadrant with palpation of the left lower quadrant, indicative of right-sided peritoneal irritation), the psoas sign (right lower quadrant pain with passive hip extension, indicative of retrocecal appendiceal inflammation), and the obturator sign (right lower quadrant pain with passive flexion of the right hip and knee, followed by internal rotation of the right hip, indicative of internal obturator inflammation, and therefore a “pelvic” appendix). Cervical motion tenderness may be present on pelvic examination, as may pelvic tenderness upon rectal examination, especially in the case of a retrocecal or pelvic appendix.
LABORATORY AND IMAGING STUDIES
While the diagnosis of appendicitis is based primarily upon clinical findings, a complete blood count (CBC) is usually obtained to screen for leukocytosis. A retrospective review of the pregnancies of over 66,000 consecutive deliveries at a single hospital was performed in the late 1990s, during which the mean leukocyte count among expectant mothers with confirmed appendicitis was found to be 16,400 cells/mm3, compared to 14,000 cells/mm3 for those found to have a normal appendix (Mourad et al., 2000). A left shift or bandemia in the differential, thought previously to favor appendicitis or another infectious process, was found to be nondiagnostic in the same study. When the overall white count is normal, appendicitis is unlikely, while a white count above 20,000 cells/mm3 in a nonlaboring patient is concerning for an appendiceal perforation.
As previously mentioned, urinalysis in a woman with appendicitis may show microscopic hematuria or pyuria, and this does not discount the diagnosis. In addition, in the absence of biliary disease, mild elevations in serum 239bilirubin (>1.0 mg/dL) have been associated with a perforated appendix (Sand et al., 2009).
Though an experienced examiner may be able to diagnose appendicitis based upon history, physical exam, and laboratory findings alone, imaging is recommended when the diagnosis is not certain. The diagnosis of appendicitis is made by US with 86% sensitivity and 81% specificity (Terasawa, Blackmore, Bent, & Kohlwes, 2004) if a thick-walled, noncompressible, blind-ended tubular structure, with a diameter greater than 6 mm, is present in the right lower quadrant, as shown in Figure 21.1.
If a normal appendix is not clearly visualized by US, which has been reported to be the case up to 97% of the time (Lehnert, Groos, Linnau, & Moshiri, 2012), additional imaging with MRI is advised, where available. In the largest study to date, the sensitivity and specificity of MRI in the detection of appendicitis were 100% and 93%, respectively. A negative predictive value of 100% was also noted (Pedrosa, Lafornara, Pandharipande, Goldsmith, & Rofsky, 2009). The diagnostic finding of an enlarged (>6 mm), fluid-filled appendix is shown in Figure 21.2.
When clinical evaluation and US results are not conclusive, or MRI is not readily available, CT is recommended (Long et al., 2011). CT is available in most institutions and has well-established diagnostic value for appendicitis in the general population, with a sensitivity of 94% and a specificity of 95% (Teresawa, Blackmore, Bent, & Kohlwes, 2004). A meta-analysis of three retrospective studies in pregnant women reported sensitivity of 85.7% and a specificity of 97.4% (Basaran & Basaran, 2009). The main findings suggestive of appendicitis on CT are an enlarged (>6 mm) nonfilling or occluded tubular structure, inflammation as manifested by appendiceal wall thickening or enhancement, and/or periappendiceal fat stranding in the right lower quadrant, as seen in Figure 21.3.
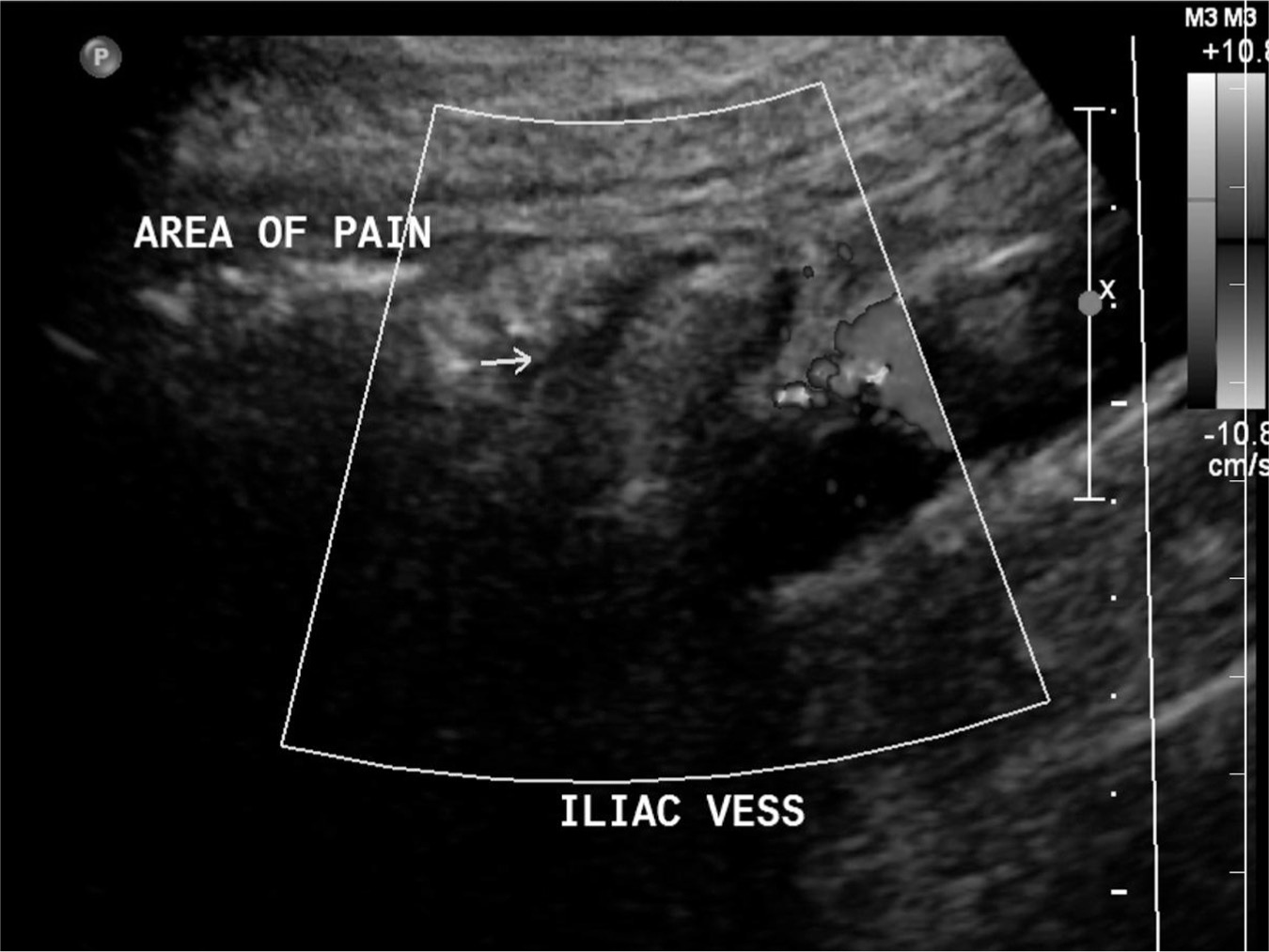
Figure 21.1 Ultrasound appearance of acute appendicitis
Source: Courtesy of Department of Radiology, Women & Infants Hospital, Providence, RI.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








