134 Central Nervous System Infections
Infections of the meninges can be subclassified by the acuity of onset of symptoms. Bacterial infections almost exclusively cause acute meningitis syndrome, characterized by rapid (<48 hours) progression of fever, headache, and meningismus. In contrast, the subacute meningitis syndrome, frequently due to viruses, fungi, or mycobacteria, is more slowly evolving, with symptoms developing over several days to weeks (Table 134-1). The following sections outline approaches to acute meningitis and subacute CNS infection syndromes. These approaches prioritize the competing needs of obtaining a precise etiologic diagnosis versus instituting early antimicrobial therapy.
TABLE 134-1 Causes of Acute and Subacute Central Nervous System Infection Syndromes
| Acute Meningitis Syndrome |
| Rapid onset (<24-48 h) of fever, headache, or meningismus, with early cognitive impairment |
| Common |
| Pyogenic meningitis (pneumococcal, meningococcal, Listeria, other) |
| Uncommon |
| Viral encephalitis (especially herpes simplex), subarachnoid bleed, brain abscess (with rupture) |
| Rare |
| Viral meningitis, granulomatous meningitis (cryptococcal, mycobacterial), carcinomatous meningitis, brain tumor |
| Subacute Central Nervous System Infection Syndrome |
| Subacute onset (>24-48 h) of fever, headache, or meningismus, with no or gradual cognitive impairment |
| Common |
| Viral meningitis, viral encephalitis, rickettsial infection |
| Uncommon |
| Brain abscess, brain tumor, granulomatous meningitis |
| Rare |
| Cerebrovascular accident, carcinomatous meningitis |
 Bacterial Meningitis
Bacterial Meningitis
Anatomy
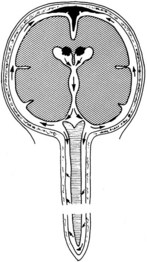
Bacterial meningitis is a pyogenic infection of the cerebral ventricles and the subarachnoid space, with bacteria usually confined to the nutrient-rich cerebrospinal fluid (CSF). CSF is formed in the choroid plexus of the ventricles, flows into the subarachnoid space at the cisterna magna and around the cerebral hemispheres, and is reabsorbed by the arachnoid villi (Figure 134-1). In adults, CSF is produced at a rate of approximately 500 mL/day, yet the CSF space averages only 140 mL in volume, consistent with rapid production and reabsorption. The cerebral and spinal subarachnoid spaces connect at the cisterna magna. Flow through the spinal subarachnoid space is of variable velocity and direction.
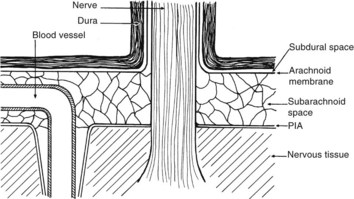
There are numerous potential and actual spaces among the layers of the meninges (Figure 134-2). Meningitis involves the actual space (i.e., the subarachnoid space), which consists of multiple interconnected compartments. The small size of the foramina of Luschka and Magendie allows unidirectional caudal flow toward the cisterna magna, where the CSF then moves either cephalad or into the spinal canal. This compartmentalization has implications for therapy, because the movement of medications and infectious agents depends on the rate and direction of CSF flow. A blockage at any of these levels may restrict entry of antibiotics into sites of ongoing infection.
Infectious agents can invade the CSF by at least three routes (Table 134-2). First, the vascular structures of the choroid plexus and pia and the vessels that traverse the subarachnoid space may serve as conduits during systemic bacteremia. A second less common route is direct invasion across the protective meninges. Physical disruption of the dura by trauma or surgery allows direct invasion of the subarachnoid space and should be considered in patients with a history of CSF leakage or rhinorrhea. Emissary veins provide another pathway for bacteria to spread from contiguous foci into the subarachnoid space. These veins traverse the skull and dura, directly connecting the soft tissues of the head and neck with the venous system of the brain and meninges, including the arachnoid villi. Although blood in the emissary veins usually flows away from the brain, the CNS veins and dural sinuses do not contain valves, and retrograde flow of bacteria is possible. Rarely, organisms may reach the ventricles or subarachnoid space from within the neural tissue; for example, rupture of a brain abscess into the ventricles may have disastrous effects.
TABLE 134-2 Routes by Which Bacteria May Enter the Subarachnoid Space
Pathophysiology
Neural damage occurs as a direct result of the host inflammatory response. The unique anatomy and composition of the CSF-filled compartments, combined with a paucity of host immunologic defenses, create a microenvironment that allows the persistence and proliferation of microorganisms.1 Polymorphonuclear leukocytes are not normal inhabitants of the CSF, and mobilization of these phagocytic cells is delayed during the early stages of infection. Similarly, the concentration of immunoglobulin in CSF is significantly less than that in serum, limiting the effectiveness of humoral immunity. Most important, complement, which plays a critical role in chemotaxis, phagocytosis, and intracellular killing, is virtually absent from normal CSF. Once in the CSF, bacteria induce leukocyte migration into the subarachnoid space, resulting in occlusion of cortical blood vessels, damage to nerve roots that traverse the subarachnoid space (see Figure 134-2), and impaired CSF flow (see Figure 134-1). Clinically, this manifests as cranial or spinal nerve dysfunction and hydrocephalus. Activation of leukocytes leads to an inflammatory cascade, with the release of cytokines, oxidants, and proteolytic enzymes. At the cellular level, this chain of events produces disruption of the blood-brain barrier and impaired cerebrovascular autoregulation.4 Increased intracranial pressure may result in transtentorial herniation or tissue hypoxia due to decreased tissue perfusion.
Clinical Course
Acute Meningitis Syndrome
Early recognition and therapy of acute meningitis syndrome are essential to minimize morbidity and mortality. The initial manifestation of the illness may be subtle, with a low-grade headache or fever. However, once meningeal symptoms (vomiting, severe headache, stiff neck) develop, the clinical course is dramatic. Patients appear “toxic,” and higher integrative functions may deteriorate rapidly. The classic clinical triad associated with bacterial meningitis, fever, neck stiffness, and altered mental status is present in only 44% of cases.6 A rash is highly suggestive of meningococcal infections; however, skin lesions are noted in only 64% of meningitis cases due to this organism.7 Other common signs and symptoms include headache and nausea. In the elderly, recognition of acute bacterial meningitis may be delayed by the absence of suggestive clinical findings. Nuchal rigidity, vomiting, and headache are significantly less frequently noted in the elderly; in contrast, seizures are present in 26% of patients ≥65 years of age.10 In addition, the elderly have a higher incidence of noninfectious conditions that may mimic acute meningitis syndrome (e.g., subarachnoid bleeding and malignancies involving the CNS) complicating the initial evaluation.
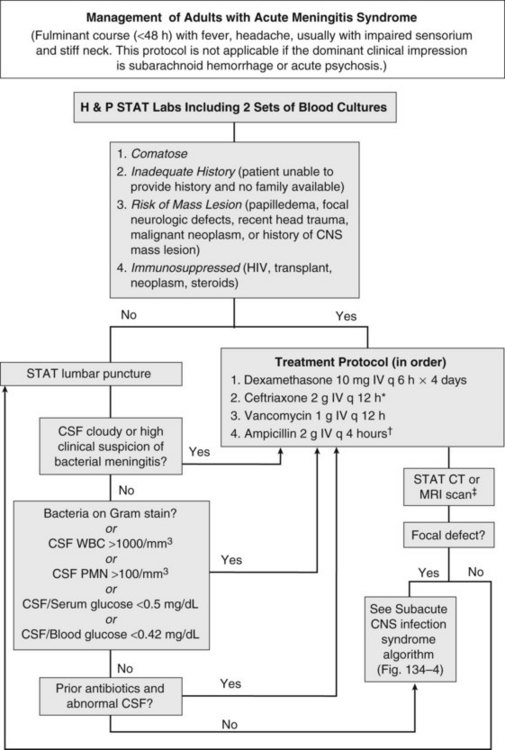
Acute meningitis syndrome represents an infectious disease emergency. Baseline predictors of adverse outcome in adults include advanced age, tachycardia (heart rate >120 beats/min), low Glasgow Coma Scale score, cranial nerve palsies, CSF WBC count less than 1000/mm3, and presence of gram-positive cocci on CSF Gram stain.8 This latter variable reflects the higher mortality associated with pneumococcal meningitis. Despite the availability of antibiotics active against all common causes of acute bacterial meningitis, in adults the overall mortality remains approximately 20%.6 A delay in antibiotic therapy increases the risk of an adverse outcome, particularly when progressive neurologic impairment occurs before receiving therapy.9 For this reason, recently published guidelines recommend the administration of empirical antibiotics to patients with a presumptive diagnosis of bacterial meningitis as soon as possible after presentation (Figure 134-3).5
Coupled with the need for urgent treatment is the need for urgent diagnosis. Identification of a pathogen allows the clinician to tailor the antibiotic regimen based on susceptibility patterns, and has prognostic and therapeutic implications. However, situations arise when lumbar puncture is unavoidably delayed. This may be due to anatomic factors that make lumbar puncture technically difficult or the need to perform neuroimaging studies to exclude a contraindication to lumbar puncture. If a significant delay in obtaining CSF is anticipated, antibiotics should be given immediately after peripheral blood cultures are obtained. The yield of CSF culture decreases within as little as 15 minutes following administration of antibiotics.11 Nevertheless, the risk of delaying treatment outweighs the need to make a microbiological diagnosis. Despite the inhibitory effect of prior antibiotics on bacterial culture and Gram stain, the absolute neutrophil count and neutrophilic pleocytosis remain suggestive of bacterial meningitis,12,13 and a full course of empirical therapy should be completed if CSF parameters are consistent with this diagnosis.
Historically, the perceived risk (and legal consequences) of uncal herniation following lumbar puncture in the presence of an intracranial mass lesion led to the ubiquitous use of neuroimaging. More recently, studies have challenged this practice, citing the potential deleterious effect of computed tomography (CT) scan–related delays in the initiation of therapy or the compromising effect of premature sterilization of CSF cultures.9,11,16 Even among patients with an abnormal CT scan, only a minority of cases have radiographic findings precluding lumbar puncture.14 For this reason, guidelines suggest that neuroimaging prior to lumbar puncture should be reserved for patients with compromised immune systems (e.g., human immunodeficiency virus [HIV] infection, use of immunosuppressive medications, or organ transplantation), history of an intracranial mass lesion, abnormal level of consciousness, papilledema, or focal neurologic deficit.5 In the absence of one of these features, the recommendation is to proceed directly to lumbar puncture, followed by immediate administration of empirical antibiotics.5
Subacute Central Nervous System Infection Syndrome
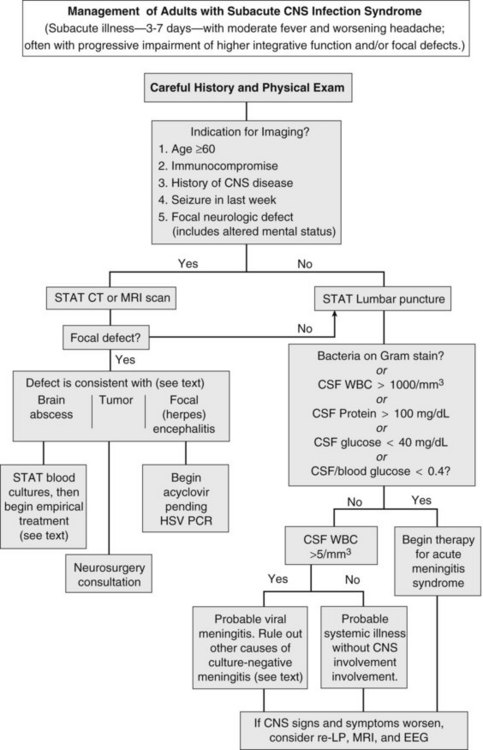
Herpes simplex encephalitis, brain abscess, and meningitis due to fungi, mycobacteria, fastidious bacteria (e.g., Rickettsia rickettsii, Treponema pallidum), or viruses all produce fever, worsening headache, and progressive impairment of higher integrative functions. On occasion, carcinomatous meningitis, brain tumor, and subarachnoid bleeding cause similar findings (see Table 134-1). To avoid inappropriate therapy and unnecessary hospitalization, the decision to institute antimicrobial therapy should be carefully weighed. However, if pyogenic meningitis is still a possibility, empirical antimicrobial therapy should be begun, as outlined in the previous section.
The first priority when managing subacute CNS syndrome is rapid diagnosis (as opposed to the rapid-therapy approach to acute meningitis syndrome). With this syndrome, the physician has time to carefully evaluate the patient and relevant laboratory data (Figure 134-4). Peripheral blood granulocytosis (>10,000/mm3), CSF cell counts over 1000/mm3, CSF protein concentration over 100 mg/dL, and CSF glucose concentrations below 40 mg/dL favor a bacterial cause, and these patients should be given empirical antibiotics for acute meningitis syndrome until a specific diagnosis is made or bacterial cultures return negative.
Additional diagnostic studies may be indicated for subacute infections. Serologic testing for HIV should be performed, because the spectrum of infectious agents is much broader among HIV-infected individuals. Testing for enteroviruses (CSF polymerase chain reaction [PCR] or viral culture), Cryptococcus (cryptococcal antigen), neurosyphilis (VDRL), mycobacterial infection (culture or PCR of CSF), herpes simplex virus (CSF PCR), tickborne infections (Ehrlichia, Rickettsia, Lyme disease), and arboviral encephalitides (West Nile virus) should be individualized based on patient characteristics, severity of illness, knowledge of local pathogens, and season. Despite intensive diagnostic testing, a pathogen is identified in only two-thirds of patients with subacute meningitis syndrome.17
Epidemiology
The epidemiology of bacterial meningitis has evolved in the last twenty years. The incidence of bacterial meningitis in the pediatric population has decreased markedly in the United States following the widespread use of conjugate vaccines active against Haemophilus influenzae type B, Neisseria meningitidis, and Streptococcus pneumoniae.18Conversely, the incidence of nosocomial bacterial meningitis caused by resistant strains of Enterobacteriaceae, Pseudomonas aeruginosa, and Staphylococcus aureus is increasing following surgery or instrumentation of the CNS.2
Therapy
Antibiotics
The choice of empirical antibiotics is based on knowledge of the likely causative agents, which vary based on host characteristics (e.g., age, immunocompromise), site of acquisition (nosocomial versus community acquired), and local resistance patterns. Recommendations for empirical therapy are listed in Table 134-3, with dosages commonly used for the treatment of CNS infections listed in Table 134-4.3,5 Pneumococci and meningococci remain the most common causes of community-acquired meningitis in immunocompetent adults younger than 50 years.3 In the last decade, pneumococci that are intermediately (minimum inhibitory concentration [MIC] >0.12 to 1 µg/mL) or highly (MIC >2 µg/mL) resistant to penicillin have emerged as important pathogens. Penicillin-resistant pneumococci are typically multidrug resistant; however, many isolates remain sensitive to third-generation cephalosporins, and all are susceptible to vancomycin.
TABLE 134-4 Antimicrobial Dosages for Central Nervous System Infections
| Drug | Dosage (by Total Body Weight) | Usual Dosage (for 70-kg Adult) |
|---|---|---|
| Acyclovir | 10 mg/kg IV q 8 h | 700 mg IV q 8 h |
| Ampicillin | 30 mg/kg IV q 4 h | 2 g IV q 4 h |
| Cefotaxime | 30 mg/kg IV q 6 h | 2 g IV q 6 h |
| Ceftazidime | 30 mg/kg IV q 8 h | 2 g IV q 8 h |
| Cefepime | 30 mg/kg IV q 8 h | 2 g IV q 8 h |
| Ceftriaxone | 30 mg/kg IV q 12 h | 2 g IV q 12 h |
| Meropenem | 40 mg/kg IV q 8 h* | 2 g IV q 8 h |
| Metronidazole | 7.5 mg/kg IV q 6 h | 500 mg IV q 6 h |
| Nafcillin | 30 mg/kg IV q 4 h | 2 g IV q 4 h |
| Penicillin G | 60,000-70,000 units/kg IV q 4 h | 4 million units IV q 4 h |
| Tobramycin or gentamicin† | 2 mg/kg IV load, then 1.7 mg/kg q 8 h‡ | 140 mg IV load, then 120 mg IV q 8 h‡ |
| Intrathecal | 0.1 mg/kg/d | 5-10 mg/d |
| Intraventricular | 0.1 mg/kg/d | 5-10 mg/d |
| Trimethoprim-sulfamethoxazole | 5 mg/kg IV q 6 h | 350 mg IV q 6 h§ |
| Vancomycin | 15 mg/kg IV q 6 h | 500 mg IV q 6 h‡ or 1 g IV q 12 h |
* Pediatric dosage. Adults should receive usual dosage.
† Regardless of which aminoglycoside is used, only preservative-free preparations should be used.
‡ Adjust dosage based on serum levels.
§ Dosage indicates trimethoprim component.
Antimicrobial therapy should be directed against the most common causes of bacterial meningitis. In the absence of a positive CSF Gram stain, initial therapy for adults with community-acquired meningitis should include a third-generation cephalosporin such as cefotaxime or ceftriaxone, as well as vancomycin. Vancomycin should never be used alone as initial therapy because of its marginal CNS penetration and lack of activity against gram-negative organisms. Empirical ampicillin therapy for Listeria monocytogenes should be added for adults aged 50 years or older or for patients with T-cell immunocompromise (e.g., on chronic steroid therapy), pregnant women, or patients with significant use of alcohol.3,5 If the CSF Gram stain shows gram-positive rods suggestive of Listeria, intravenous (IV) gentamicin should be added. For patients intolerant of penicillins, trimethoprim-sulfamethoxazole is an acceptable alternative for treatment of Listeria meningitis.
In contrast to community-acquired meningitis, organisms causing nosocomial meningitis reflect the highly resistant strains endemic to the hospital. Empirical therapy for patients suspected to have nosocomial meningitis must therefore be directed against staphylococcal species (both coagulase-positive and coagulase-negative strains) and multidrug-resistant strains of gram-negative bacilli, including P. aeruginosa and Acinetobacter baumannii (see Figure 134-3). Empirical treatment in this population should therefore include vancomycin as well as an antipseudomonal cephalosporin (ceftazidime or cefepime) or carbapenem. Imipenem is active against Pseudomonas and achieves therapeutic levels in the CSF; however, because this agent lowers the seizure threshold, it is relatively contraindicated for meningitis. Meropenem, a related carbapenem, is less epileptogenic and is therefore preferred for this indication.19
Initial antibiotic choices can be refined when sensitivity patterns become available, typically in 2 to 3 days. The duration of therapy in bacterial meningitis varies with the pathogen and the clinical response. Although there have been few randomized studies evaluating the optimal duration of therapy, 7 days of treatment for H. influenzae and N. meningitidis meningitis is typically sufficient,20 whereas S. pneumoniae requires 10 to 14 days of therapy.5,17 Adults with pneumococcal meningitis may have predisposing infections including pneumonia, sinusitis, otitis, or rarely endocarditis. Although therapy for meningitis usually treats the primary cause, endocarditis requires prolonged therapy with bactericidal antibiotics.
For all causes of bacterial meningitis, abnormalities of the CSF (high protein and cell counts) persist for days to weeks. Resolution of symptoms (e.g., fever, leukocytosis, meningismus) should serve as adequate evidence of successful therapy. In a patient who responds poorly to 48 hours of therapy, repeat lumbar puncture and head CT or MRI are indicated. Repeat lumbar puncture is particularly important for detecting clearance of bacteria from the CSF in patients with cephalosporin-resistant pneumococcal meningitis who demonstrate a slow clinical response.19 Patients with culture-negative pyogenic meningitis and suboptimal clinical response should also have repeat lumbar puncture to ensure response to empirical antibiotics. Ongoing or worsening CSF parameters suggest infection with either resistant bacteria or with a pathogen more typically associated with subacute meningitis syndrome (see Table 134-1).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree