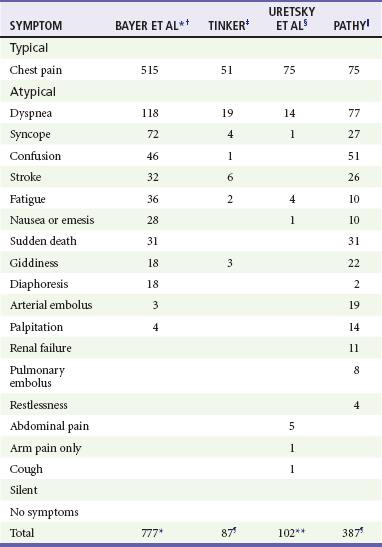
Chapter 78 Ischemic heart disease and CAD continue to be the leading causes of death among adults in many developed countries. Ischemic heart disease accounts for nearly 1 million deaths in the United States annually, of which approximately 160,000 occur in persons 65 years of age or younger. More than half of all deaths from cardiovascular disease occur in women, and CAD remains a major cause of morbidity and mortality in women beyond their middle to late fifties. The incidence of cardiovascular disease is expected to continue to increase owing to lifestyle and behavioral changes that promote heart disease.1 A significant reduction in age-adjusted mortality from CAD has occurred in the United States over the past four decades.2,3 In large part, the decline has been accompanied by diminished mortality from AMI. This decrease is a result of a reduction in the incidence of AMI by 25% and a sharp drop in the case-fatality rate. Reduction in cigarette smoking, management of lipids, and improved management of hypertension and diabetes mellitus undoubtedly play a role, along with significant advances in medical treatment. In 2005, 5.8 million patients were evaluated for chest pain or related complaints in EDs in the United States, constituting 5% of all ED visits. In 2004, 4.1 million visits to the ED had a primary diagnosis of cardiovascular disease, and over 1.5 million patients were hospitalized for a primary or secondary diagnosis of ACS.4–7 In addition, approximately 2% of patients with ACS are discharged from the ED. In the United States, approximately 900,000 persons every year experience an AMI, of whom 20% die before reaching the hospital, and 30% die within 30 days.8,9 The majority of fatalities from CAD occur outside the hospital, usually from an ACS-related dysrhythmia within 2 hours of onset of symptoms. For many patients who experience a nonfatal AMI, their lives are limited by an impaired functional status, anginal symptoms, and a diminished quality of life. The economic cost of ACS is estimated to be $100 to $120 billion annually.10 Acute myocardial infarction is defined as myocardial cell death and necrosis of the myocardium. The four-decade-old World Health Organization (WHO) definition for AMI has been replaced by clinical criteria developed jointly by the European Society for Cardiology and American College of Cardiology (ACC) that focus on defining infarction as any evidence of myocardial necrosis. This definition for an acute, evolving, or recent MI requires a typical rise and fall of a cardiac biochemical marker, currently troponin, with clinical symptoms, ECG changes, or coronary artery abnormalities based on interventional evaluation.11 The actual definition,11 referred to as the “Universal Definition of Myocardial Infarction,” includes the following; either one of these criteria satisfies the diagnosis for an acute, evolving, or recent MI: 1. Typical rise and gradual fall or more rapid rise and fall of biochemical markers of myocardial necrosis with at least one value above the 99th percentile of the upper reference limit (URL) and with at least one of the following clinical parameters: • ECG changes indicative of ischemia (T wave changes or ST segment elevation or depression) • Development of pathologic Q waves on the ECG • Imaging evidence of presumably new findings, such as a loss of viable myocardium or a regional wall motion abnormality Furthermore, regarding an established MI, any one of the following criteria satisfies this diagnosis11: • Development of new pathologic Q waves on serial ECGs. The patient may or may not remember previous symptoms. Biochemical markers of myocardial necrosis may have normalized, depending on the length of time since the infarct developed. • Imaging evidence of a region of loss of viable myocardium that is thinned and fails to contract, in the absence of a nonischemic cause. • Type 1—Spontaneous MI related to ischemia resulting from a primary coronary event, such as plaque erosion rupture, erosion, fissuring, or dissection with accompanying thrombus formation and vasospasm. Type 1 infarctions represent the “true” ACS event. • Type 2—MI secondary to ischemia caused by either increased oxygen demand or decreased supply, as seen in coronary artery spasm, coronary embolism, severe anemia, compromising arrhythmias, or significant systemic hypotension. • Type 3—Sudden unexpected cardiac death, including cardiac arrest, often with symptoms suggestive of myocardial ischemia, accompanied by presumably new ST segment elevation or new left bundle branch block (LBBB) pattern. Fresh coronary thrombus is noted via either angiography or autopsy; death occurs before appropriate sampling of the blood to detect the abnormal cardiac biomarker. • Type 4—MI associated with coronary instrumentation, such as occurring after percutaneous coronary intervention (PCI). For PCIs in patients with normal baseline troponin values, elevations of cardiac biomarkers above the 99th percentile URL are indicative of periprocedural myocardial necrosis. By convention, increases of biomarkers greater than 3 times the 99th percentile URL are designated as defining PCI-related MI. A subtype related to a documented stent thrombosis is similarly recognized. • Type 5—MI associated with coronary artery bypass grafting (CABG). For CABG in patients with normal baseline troponin values, elevations of cardiac biomarkers above the 99th percentile URL are indicative of periprocedural myocardial necrosis. By convention, increases of biomarkers greater than five times the 99th percentile URL plus any of the following are designated as defining CABG-related MI: AMI is further classified by findings on the ECG at presentation, as either STEMI or NSTEMI. Previous descriptors, such as transmural and nontransmural, as well as Q wave and non–Q wave MI, fail to adequately describe the coronary event and its related pathophysiology, electrocardiographic presentation, and pathologic outcome. The differentiation between STEMI and NSTEMI has important implications in terms of management, outcome, and prognosis for patients with AMI. In fact, the ACC and the American Heart Association (AHA) have separate clinical guidelines for the management of patients with UA/NSTEMI and those patients with STEMI.6,7,12 The underlying pathophysiology of ACS is myocardial ischemia as a result of inadequate perfusion to meet myocardial oxygen demand. Myocardial oxygen consumption is determined by heart rate, afterload, contractility, and wall tension. Inadequate perfusion most commonly results from coronary arterial vessel stenosis as a result of atherosclerotic CAD. Usually the reduction of coronary blood flow does not cause ischemic symptoms at rest until the vessel stenosis exceeds 95%. Myocardial ischemia, however, may occur with exercise and increased myocardial oxygen consumption with as little as 60% vessel stenosis.13 CAD is characterized by thickening and obstruction of the coronary vessel arterial lumen by atherosclerotic plaques. Although atherosclerosis is usually diffuse and multifocal, individual plaques vary greatly in composition. Fibrous plaques are considered stable but can produce anginal symptoms with exercise and increased myocardial oxygen consumption because of the reduction in coronary artery blood flow through the fixed, stenotic lesions. Vulnerable or unstable fibrolipid plaques consist of a lipid-rich core separated from the arterial lumen by a fibromuscular cap. These lesions are likely to rupture, resulting in a cascade of inflammatory events, thrombus formation, and platelet aggregation that can cause acute obstruction of the arterial lumen and myocardial necrosis.14 In the setting of UA, acute stenosis of the vessel is noted; complete obstruction, however, is encountered in only 20% of cases. In these cases, it is likely that extensive collateral vessel circulation prevents total cessation of blood flow, averting frank infarction.13 With AMI, the occlusive fibrin-rich thrombus is fixed and persistent, resulting in myonecrosis of the cardiac tissue supplied by the affected artery. Angiographic studies demonstrate that the preceding coronary plaque lesion is often less than 50% stenotic, indicating that the most important factors in the infarction are the acute events of plaque rupture, platelet activation, and thrombus formation rather than the severity of the underlying coronary artery stenosis. Appropriate pharmacotherapy for persistent anginal chest pain in the preadmission setting includes sublingual NTG, oral aspirin (acetylsalicylic acid [ASA]) that is preferably chewed, and intravenous morphine sulfate; the acronym MONA summarizes preadmission pharmacotherapeutic interventions (morphine, oxygen, nitroglycerin, and aspirin). Establishment of the diagnosis of ACS in this setting is difficult, however, as chest pain is a poor predictor of the diagnosis and adjunctive tools are limited.15 Preadmission 12-lead ECG offers high specificity (99%) and positive predictive value (93%) for AMI in patients with atraumatic chest pain while increasing the paramedic scene time by an average of only 3 minutes. This approach offers many advantages, including (1) earlier detection of STEMI, (2) ability to base the destination on the availability of PCI, and (3) more rapid reperfusion therapy.7 Preadmission 12-lead ECG would be necessary in the limited populations in whom preadmission fibrinolytic therapy might be applicable, such as those with prolonged out-of-hospital times (90-120 minutes). Traditionally, a history of risk factors for CAD is sought; these include male gender, age, tobacco smoking, hypertension, diabetes mellitus, hyperlipidemia, family history, artificial or early menopause, and chronic cocaine abuse. Approximately 80% of a population of more than 122,000 patients with known CAD had at least one of the four conventional risk factors (diabetes mellitus, cigarette smoking, hypertension, or hyperlipidemia).16 Cardiac risk factor burden has little impact on the ED diagnosis of ACS; however, in patients older than 40 years, ACS is 22 times more likely if four of the five major risk factors (diabetes mellitus, smoking, hypertension, hyperlipidemia, and family history) are present (compared with none).17 Nevertheless, Bayesian analysis indicates that risk factors are a populational phenomenon and do not increase or decrease the likelihood of any condition in any one patient. Thus the presence of an individual risk factor or a collection of risk factors is far less important in diagnosing acute cardiac ischemia in the ED than the history of presenting illness, prior diagnosis of ischemic cardiac disease in the patient, the presence of ST segment or T wave changes, or cardiac marker abnormalities.18 Risk assessment tools, such as the PURSUIT (Platelet Glycoprotein IIb-IIIa in Unstable Angina: Receptor Suppression Using Integrilin Therapy) risk model, the GRACE (Global Registry of Acute Coronary Events) risk model, and the TIMI (Thrombolysis in Myocardial Infarction) risk score, can be used to determine risk of death and ischemia in NSTEMI and STEMI. The TIMI risk score assigns a point each for seven factors based on history, cardiac markers, and the ECG. It can be accessed at www.timi.org.6 Although these tools may aid in decision-making and in risk stratification for patients to properly determine their disposition (telemetry bed vs. intensive care unit), none of them are designed to identify patients who may safely be discharged home. There are several nontraditional risk factors for coronary disease. Antiphospholipid syndrome, rheumatoid arthritis, human immunodeficiency virus (HIV),19 and particularly systemic lupus erythematosus (SLE) are associated with a higher risk of cardiovascular disease.20 Women with SLE who are 35 to 44 years of age are over more than 50 times more likely to have an MI than a similar age- and gender-matched Framingham population.21 The term angina refers to “tightening,” not pain. Classic angina pectoris may not be pain at all but rather a “discomfort,” with a “squeezing,” “pressure,” “tightness,” “fullness,” “heaviness,” or “burning” sensation. Classically, it is substernal or precordial in location and may radiate to the neck, jaw, shoulders, or arms. If the discomfort does extend down the arm, it classically involves the ulnar aspect. Discomfort in the left chest and radiation to left-sided structures is typical, but location and radiation to both sides or to only the right side may be consistent with angina. Radiation of the discomfort to the right arm or shoulder, or to both arms or shoulders, exceeds radiation to the left arm or shoulder in terms of likelihood of the chest pain being caused by ACS, although all exceed a positive likelihood ratio of 2.22,23 Furthermore, classic features of angina pectoris include exacerbation with exertion, a heavy meal, stress, or cold, and alleviation with rest. The onset of pain at rest in no way excludes the diagnosis of angina. Anginal discomfort characteristically lasts from 2 to 5 minutes up to 20 minutes, and it is rare for it to last only a few seconds or to endure for hours or incessantly, “all day” (Table 78-1). Table 78-1 Clinical Characteristics of Classic Anginal Chest Discomfort Adapted from Zink BJ: Angina and unstable angina. In Gibler WB, Aufderheide TP (eds): Emergency Cardiac Care. St. Louis, Mosby, 1994. Symptoms characteristically associated with angina pectoris, or other entities of ACS, include dyspnea, nausea, vomiting, diaphoresis, weakness, dizziness, excessive fatigue, or anxiety (Table 78-2). If these symptoms arise, either alone or in combination, as a presenting pattern of known ischemic coronary disease, they are termed anginal equivalent symptoms. Recognition that coronary ischemia may arise with an anginal equivalent rather than a classic symptom is the key to understanding the atypical presentation of ACS. Complaints of “gas,” “indigestion,” or “heartburn” in the absence of a known history of gastroesophageal reflux disease, or if the heartburn is different from the patient’s usual gastroesophageal reflux, or reproducible pain on abdominal palpation should raise suspicion of ACS. Gastroesophageal reflux disease is a common misdiagnosis in cases of missed ACS. Table 78-2 Symptoms of Acute Myocardial Infarction: Typical and Atypical *Patients able to report multiple symptoms; therefore total exceeds 777. †Bayer AJ, et al: Changing presentation of myocardial infarction with increasing age. J Am Geriatr Soc 34:263, 1986. ‡Tinker GM: Clinical presentation of myocardial infarction in the elderly. Age Ageing 10:237-240, 1981. §Uretsky BF, Farquhar DS, Berezin AF, et al: Symptomatic myocardial infarction without chest pain: Prevalence and clinical course. Am J Cardiol 40:498-503, 1977. ||Pathy MS: Clinical presentation of myocardial infarction in the elderly. Br Heart J 29:190-198, 1967. ¶Patients classified by principal symptom, although all patients with complaint of chest or epigastric discomfort were placed in typical group. **Same as ¶, except patients with epigastric complaints were placed in atypical group. Adapted from Scott PA, Gibler WB, Dronen SC: Acute myocardial infarction presenting as flank pain and tenderness: Report of a case. Am J Emerg Med 9:547, 1991. A description of typical symptoms (crushing, retrosternal chest pain or pressure) is often lacking in ACS; this may be a result of atypical features of the pain (e.g., character, location, duration, exacerbating and alleviating factors) or the presence of anginal equivalent symptoms (e.g., dyspnea, nausea, vomiting, diaphoresis, indigestion, syncope). Patients with an ultimate diagnosis of AMI or UA can have pain that is pleuritic, positional, or reproduced by palpation. Some patients describe their pain as burning or indigestion, sharp, or stabbing (see Table 78-2).23,24 In a large study of nearly 435,000 patients ultimately diagnosed with AMI, one third did not have chest pain on presentation.25 Multiple studies have identified risk factors for atypical presentation of ACS: diabetes mellitus, older age, female gender, nonwhite ethnicity, dementia, no prior history of MI or hypercholesterolemia, no family history of coronary disease, and previous history of congestive heart failure (CHF) or stroke.25–27 In patients with AMI or UA, atypical presenting complaints include dyspnea, nausea, diaphoresis, syncope, or pain in the arms, epigastrium, shoulder, or neck. Atypical features of ACS are present with increasing frequency in sequentially older populations. Before age 85, chest pain is found in the majority of patients with acute MI, although dyspnea, stroke, weakness, and altered mental status are notably present. In those older than 85 years, however, atypical symptoms are more common than chest pain, with 60 to 70% of patients older than 85 having an anginal equivalent complaint, especially dyspnea.27 Coincident ACS is more likely to occur in the elderly; patients with another acute condition (e.g., trauma, infection) should be scrutinized for concurrent ACS.28 Patients with diabetes mellitus are at heightened risk for ACS as well as an atypical presentation, such as dyspnea, nausea or vomiting, confusion, or fatigue. Medically unrecognized AMI can occur in 40% of patients with diabetes mellitus compared with 25% of a nondiabetic population, and myocardial scar unaccompanied by antemortem diagnosis of MI is three times more likely in diabetics.29 As with age and diabetes, female gender is an important risk factor for MI without chest pain. In some series, less than 60% of women reported chest discomfort at the time of their MI, with others reporting dyspnea, indigestion, or vague symptoms, such as weakness, unusual fatigue, cold sweats, sleep disturbance, anxiety, or dizziness.30 Finally, nonwhite racial and ethnic populations may have atypical symptoms in ACS.25 Compelling data demonstrate a disparity in treatment approach related to race in patients with acute manifestations of coronary heart disease.31 Whether this is related to the atypical nature of presenting symptoms in different racial groups is not clear. Although certain features of the chest pain history serve to increase or decrease the likelihood of ACS, none of them is strong enough to endorse discharge of the patient based on the history alone.24 The physical examination focuses on the cardiac, pulmonary, abdominal, and neurologic examinations, looking for signs of complications of ACS as well as alternative diagnoses for chest pain and the anginal equivalent syndromes (Table 78-3). Altered mental status, diaphoresis, and signs of CHF are all ominous findings in patients with symptoms consistent with ACS. Historical studies using untrained physicians identified chest wall tenderness or “reproducible” chest wall tenderness in up to 15% of patients ultimately diagnosed with AMI, but these data are highly suspect. The real incidence of truly reproducible chest wall tenderness (i.e., when the patient reliably identifies to the examiner that the pain produced on palpation is identical to the pain causing the patient’s presentation) in ACS is probably very small. It is suggested that patients with chest pain that is fully pleuritic, positional, or reproducible by palpation (the three Ps) are at low risk (yet not no risk) for ACS.22 Table 78-3 Key Entities in the Differential Diagnosis of Chest Pain Not surprisingly, atypical presentation of patients with ACS is associated with a delay in diagnosis and poorer outcomes. In the Second National Registry of Myocardial Infarction (NRMI-2) study, patients with MI without chest pain were significantly more likely to die in the hospital (23 vs. 9% for patients with chest pain) and were more likely to experience stroke, hypotension, or heart failure that required intervention, possibly reflecting the older age and greater comorbidity in this group.25 Patients with atypical symptomatology seek medical care later and are less likely to receive standard therapies, such as aspirin, beta-adrenergic blockers, heparin, fibrinolysis, and emergent reperfusion therapy.25 Patients 65 years of age or younger with NSTEMI have a 1% chance of dying during their hospitalization, but this risk is increased to 10% for patients ages 85 years and older.28 Approximately 2% to 4% of patients with acute MI in the ED are discharged without diagnosis.32 Missed ACS is the misdiagnosis that accounts for the largest amount of payment by emergency physicians in medical malpractice claims. Atypical presenting symptoms are an obvious causative consideration. Patients with undiagnosed ACS discharged from the ED are younger, more likely to be women or nonwhite, more likely to have atypical complaints, and less likely to have ECG evidence of acute ischemia.32,33 Among all patients with cardiac ischemia, women younger than 55 years seem to be at highest risk for inappropriate discharge. With respect to ECG findings, 53% of patients with missed AMI and 62% of patients with missed UA have normal or nondiagnostic ECGs. Finally, the risk-adjusted mortality ratio for all patients with acute cardiac ischemia is 1.9 times higher among nonhospitalized patients.32 Factors associated with misdiagnosis of ACS in medical malpractice closed claims analysis include physicians with less experience who document histories less clearly, admit fewer patients, and misinterpret the ECG. Bradydysrhythmia and atrioventricular (AV) conduction block occur in 25 to 30% of patients with AMI; sinus bradycardia is most commonly seen.34–36 Symptomatic bradydysrhythmias in the first few hours after inferior AMI tend to be atropine responsive; conduction abnormalities that appear beyond 24 hours of MI tend not to respond to atropine.37 Patients with AV block in the setting of anterior AMI tend to respond poorly to therapy and have a poor prognosis. Stroke may also complicate AMI, most commonly ischemic or thromboembolic. The major predisposing mechanisms with a recent MI are embolization from left ventricular mural thrombus with decreased ejection fraction, embolization from the left atrial appendage with atrial fibrillation, and hypercoagulability with concomitant carotid arterial disease. The rate of stroke is higher in the setting of MI (0.9% tapering to 0.1% at day 28 after MI) than in control subjects (0.014%).38 Hemorrhagic stroke is an obvious concern in the patient undergoing fibrinolytic therapy. The rate of hemorrhagic stroke with varying fibrinolytic agents is less than 1%, although the rate climbs in older patients. PCI lowers the overall risk of stroke compared with fibrinolytic therapy. Analysis of only fibrinolytic-eligible patients from the NRMI-2 database yields more than 24,000 patients treated with alteplase and more than 4000 who received primary angioplasty. The difference in stroke rate is highly significant (1.6% in the fibrinolytic group vs. 0.7% in the angioplasty group). Considering hemorrhagic strokes, the difference is again dramatic (1.0% in the fibrinolytic group vs. 0.1% in the angioplasty group).39 Hyperglycemia in the setting of AMI may be viewed as a complication, as well as a complicating disease process in AMI. Hyperglycemia is present in up to one half of all patients with STEMI, yet only one fifth to one fourth of those patients are recognized diabetics. Elevated glucose at the time of admission has independent negative implications for mortality rates in AMI patients. Although fasting blood sugar the day after presentation is a better predictor, an admission blood glucose level higher than 200 mg/dL is linked to similar mortality rates among diabetics and nondiabetics. There is a 4% mortality increase for nondiabetic patients for every 18-mg/dL elevation in blood glucose level. Hyperglycemia seems to induce a complex set of unfavorable cellular and biochemical circumstances, including negative effects on coronary flow and microvascular perfusion, as well as adverse effects on platelet function, fibrinolysis, and coagulation. Intravenous insulin therapy for glucose normalization is linked to improved outcomes in patients with STEMI as well as those in the medical intensive care unit. ACC/AHA guidelines acknowledge that tight control of blood glucose during and after STEMI decreases acute and 1-year mortality rates.40
Acute Coronary Syndrome
Epidemiology
Spectrum of Disease
Acute Myocardial Infarction
Pathophysiology
Clinical Features
Preadmission Evaluation
Emergency Department Evaluation
The Classic History
CHARACTERISTIC
MORE LIKELY TO BE ANGINA
LESS LIKELY TO BE ANGINA
Type of pain
Dull, pressure
Sharp, stabbing
Duration
2-5 min, often 15-20 min
Seconds or hours
Onset
Gradual
Rapid
Location
Substernal
Lateral chest wall, back
Reproducible
With exertion
With inspiration
Associated symptoms
Present
Absent
Palpation of chest wall
Not painful
Painful, exactly reproduces pain complaint
The Atypical History
Physical Examination
Acute myocardial infarction
Unstable angina
Stable angina
Prinzmetal’s angina
Pericarditis
Myocardial or pulmonary contusion
Pneumonia
Pulmonary embolism
Pneumothorax
Pulmonary hypertension
Pleurisy
Aortic dissection
Boerhaave’s syndrome
Gastroesophageal reflux
Peptic ulcer disease
Gastritis or esophagitis
Esophageal spasm
Mallory-Weiss syndrome
Cholecystitis or biliary colic
Pancreatitis
Herpes zoster
Musculoskeletal pain
Outcomes in Atypical Presentations
Missed Diagnosis of Acute Coronary Syndrome
Early Complications of Acute Myocardial Infarction
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Acute Coronary Syndrome
Only gold members can continue reading. Log In or Register to continue




