CHAPTER 31. The Pregnant Patient
Teri Campbell
Competencies
Get Clinical Tree app for offline access
1. Perform a focused assessment of the pregnant patient, which includes subjective and objective data related to the patient’s pregnancy.
2. Identify normal physiologic changes that occur during pregnancy.
3. Perform a focused assessment of the fetus before and during transport.
4. Initiate appropriate interventions for the patient in preterm labor.
5. Discuss common indications for transport of the high-risk obstetric case.
Complications that arise during pregnancy and place the pregnant patient at risk have many causes. Some complications may be related to the pregnancy itself, others may be related to preexisting medical conditions that may be aggravated by the pregnancy, and yet others may be related directly to the fetus.
Transport team personnel who provide care for the obstetric patient at risk must be prepared to assess obstetric factors so that stabilizing care can be provided in preparation for transport. For recognition of the pathologies associated with pregnancy, the transport clinician must have an understanding of normal physiologic changes that occur during pregnancy. The well being of the fetus and the mother must be considered. Identification of risk factors, early detection of possible complications, and interventions by the team during the transport can ensure a more favorable outcome for both the mother and the fetus. The transport team must be prepared to perform an obstetric assessment, determine strategies for transport, perform fetal monitoring, and initiate appropriate interventions. Complications are numerous and are usually multifocal. They can include, among other things, amniotic fluid embolism, delivery complications, diabetes in pregnancy, hemorrhagic complications, multiple gestation, pregnancy-induced hypertension (PIH) and related disorders, preterm labor (PTL) and related issues, and trauma in pregnancy. 21 The information gained with the general obstetric assessment (Box 31-1) aids the transport team in setting priorities for care during the transport. 2,4,7,8,13 The old cliché in transport and emergency medical service (EMS) was that all energies should be concentrated on the mother because “if you don’t save the mother, you can’t save the baby.” Although this adage is true in global terms, the transport team must be aware of maternal and fetal therapies that can improve survivability and decrease morbidity of the fetus as well as the mother.
BOX 31-1




General Obstetric Assessment
1. Age of patient: Age (for teenagers and for women over age 35 years) predisposes the obstetric patient to many complications.
2. Gravida/para: Gravid: The number of times pregnant, regardless of outcome. Parity is broken down into four sections. The first assessment is the number of term deliveries (after 36 weeks gestation), and the next section is the number of deliveries before 36 weeks gestation but less than 20 weeks. The next section is the number of abortions and miscarriages. The final section is the number of now living children. This more specific assessment of parity provides a tremendous amount of obstetric history. For example, a woman has been pregnant six times. She has two term deliveries, one preterm delivery, two abortions, and one baby who died of sudden infant death syndrome (SIDS). Her G/P is: G6 P 2122.
3. Estimated date of confinement (EDC): The EDC can be estimated from the first day of the last menstrual period (LMP) by using Nägele’s rule: Count back 3 months from the LMP and then add 7 days. The due date is accurate within 2 weeks. Palm operating programs have free versions for calculating LMP/EDC and gestational age.
4. Ultrasound scan: Has the patient had an ultrasound scan? How many? In the event of an uncertain or unknown LMP or irregular menses, an ultrasound scan performed between 12 and 30 weeks is reliable for dating the pregnancy within 2 weeks. An ultrasound scan can confirm the EDC estimated by the LMP. Early ultrasound scans performed before 12 weeks are accurate for dating within 1 week. An ultrasound scan is invaluable with any question about placental location, amount of amniotic fluid present, fetal presentation, expected fetal growth, or anomalies.
5. In addition to inquiry into medical history and allergies, obstetric history is of particular significance. The following information may be of some predictive value for the outcome of the current pregnancy:
a. Did the patient deliver vaginally or by cesarean section? Has she had a vaginal birth after a cesarean section? Observe for the location and extent of any abdominal scars.
b. Did she or the baby experience any delivery complications?
c. Did she experience any complications associated with any past pregnancies?
d. Has she had any preterm deliveries? At what gestation did she deliver, and what was the outcome?
e. Has she had either spontaneous or elective abortions? Was a dilation and curettage required?
f. How many living children does she have? What were the birth weights and genders of each child?
g. Has less than 1 year elapsed between the last delivery and commencement of the current pregnancy?
h. What was the length of her last labor?
6. Pertaining to the current pregnancy:
a. Is the patient having contractions? If so, when did the contractions begin? Has there been a change in the intensity or frequency of contractions? Is there accompanying backache, pelvic, or rectal pressure? How strong do the contractions palpate and how do they compare with patient reporting? What are the frequency, duration, and regularity of the contractions?
b. Is any vaginal bleeding or bloody show present? Is there active bleeding? Attempt to help the patient quantify the bleeding by the number of towels, pads, or amount of clothing soaked before arrival and observe for evidence of dried blood on the perineum, legs, and soles of the feet. Was the bleeding painless or associated with contractions or abdominal pain? Was the blood bright red or dark? Was mucus combined with the blood (bloody show)? When did the bleeding begin? Was there any previous activity that may have precipitated the bleeding?
c. Does the patient report leaking fluid vaginally? Does the patient believe her “bag of waters” has ruptured? Was there a gush or an intermittent trickle? A small leakage of clear fluid may be confused with urinary incontinence. Leakage of amniotic fluid is uncontrollable. What time did it happen? What color was the fluid: meconium-stained, dark (presence of blood in the fluid), or clear? Was an odor present? Is the Chux pad under the patient wet or pooling with fluid?
d. Does the patient smoke? If so, how much? Is there any evidence of alcohol or substance abuse? Attempt to ascertain from the patient the frequency and time of last usage.
e. Has the patient had an adequate weight gain? Does she appear malnourished or obese?
f. Has the patient had consistent prenatal care, no prenatal care, or limited prenatal care (three or fewer visits)? Obtain prenatal record if available because it provides a tremendous amount of obstetric information including history, ultrasound scan reports, laboratory reports, vital signs, etc.
g. Has there been any change in fetal activity in the past several days?
h. Is the patient currently taking any medications? If so, what is she taking and when was the last dosage?
i. Is the patient having any current medical problems or problems with this pregnancy?
j. Have any diagnostic tests been done?
7. Assess initial vital signs, including temperature: The blood pressure (BP), pulse, and respirations should be assessed every 15 minutes or as indicated. The obstetric patient should be positioned in the left lateral recumbent position before the BP is taken. When the patient is in the supine position, the gravid uterus may cause obstruction of the inferior vena cava, diminishing venous return to the heart, which may lead to supine hypotension. Consequently, uteroplacental blood flow is decreased, placing the fetus at risk for compromise.
8. Fetal heart tones (FHT): If the patient is currently being monitored with electronic fetal monitoring (EFM), evaluate the fetal heart rate (FHR) baseline and variability, observing for accelerations and decelerations. FHR should be assessed with Doppler scan if EFM is unavailable. FHR auscultations should be assessed every 15 minutes or less if any irregularities are noted. For strip interpretation, refer to the discussion in this chapter on fetal monitoring.
9. Fundal height (FH): FH should be measured in centimeters from the symphysis to the fundus. The fundal height roughly correlates to the gestation of the pregnancy in weeks. In the presence of hydramnios, multiple gestations, a large-for-gestation fetus, or a fetus with intrauterine growth restriction, the fundal height may not correlate with the gestation, signaling the possibility of complications. If no tape measure is available or the patient is unable to provide information, such as in the case of a trauma, assess the fundus in relation to the umbilicus. If the fundus is above the umbilicus, it is estimated the pregnancy is 20 to 24 weeks gestation.
10. Lightly palpate the fundus for strength, frequency, and duration of contractions: The fingertips can indent the fundus freely with mild contractions and slightly with moderate contractions; firm tension is noted with strong contractions. Between contractions, palpate the abdomen for localized or generalized tenderness and observe the patient’s coping response to the contractions. Gestures, posture, and facial expressions in response to contractions and verbal description should be noted. If the patient is in labor, observe for indications of advancing labor such as apprehension, restlessness, increasing difficulty coping with the contractions, screaming, nausea and vomiting, bearing-down effort, increase in bloody show, or a bulging perineum.
11. Roughly determine the fetal position with abdominal palpation: With the fingertips and palms, lightly palpate the fundus for the head or buttocks, moving down the sides to identify the fetal spine and small parts, and palpate the lower uterine segment for the presenting part. If the fetal position remains unclear, the fetus may be in a transverse lie. The FHT is heard most clearly over the fetal spine.
12. Assess cervical status as indicated by the presence of contractions: If the amniotic membranes are intact, cervical status just before departure should be documented. If the membranes are ruptured, a sterile vaginal examination (SVE) should never be attempted unless delivery is deemed imminent. In the presence of hemorrhage, an SVE should never be attempted unless a placenta previa has been ruled out with ultrasound scan. During transport, an SVE is not indicated unless signs of advancing labor are noted.
13. Observe for the presence of other risk factors that predispose the obstetric patient to complications.
DETERMINATION OF TEAM COMPOSITION FOR TRANSPORT OF THE PREGNANT PATIENT
Determination of the members of the transport team who transport the pregnant patient continues to generate controversy. Many pregnant patients are transported by teams composed of personnel who transport a variety of patients. 11 However, at times, the patient’s or fetus’s condition may warrant personnel with high-risk obstetric or neonatal experience. Dedicated maternal transport teams may be considered ideal from a patient care stand point but are often cost prohibitive or unavailable for many transport programs. The challenge becomes the adequate training of nonspecialty teams in competent assessment and care of the high-risk obstetric case. This controversy is not dissimilar to the training of transport teams in care for specialty patients who need intraaortic balloon pumps, left ventricular assist devices (LVADs), and other specialized equipment. Some suggested guidelines include3,16:
▪ A patient who is not in labor but needs transport for complications of pregnancy, such as preeclampsia or third-trimester bleeding, can probably be transported by a team with maternal and neonatal experience or a general transport team that has had high-risk obstetric (HROB) training from obstetric specialists.
▪ A patient not in labor but with severe preeclampsia ideally should be transported by a maternal transport team.
▪ A patient in labor may need both a maternal and a neonatal transport team. If this is not feasible or practical, the transport team may consider delaying the maternal transport and waiting until the baby is delivered. At this time, the neonate can be safely transported in a controlled environment. Individual program-directed guidelines are imperative in assisting with transport timing decisions.
If the transport team does not routinely transport high-risk obstetrical cases, the Commission on Accreditation of Medical Transport Systems9 recommends that the team members receive training in neonatal resuscitation. The transport vehicle should allow access to both the mother and the child in the case of a delivery during transport. Each transport program should have a policy and procedure in place that addresses when transport of a pregnant patient is and is not appropriate and what team members should provide care during transport. In addition, facilities are recommended to offer high-risk obstetric education, through the receiving facility or outside educators. Education directly leads to increased competency and confidence.
If transport services do include specialty teams as a part of their service, they must be sure that these individuals receive annual training and are equipped and dressed for transport. This training should include9:
▪ Use of restraint systems in the transport vehicle.
▪ Safety and survival skills.
▪ Emergency egress training.
▪ Postaccident/incident training.
Contraindications for initiation of maternal transport should be considered before leaving a referring facility. These contraindications include1,8:
▪ Inability to stabilize the mother’s condition, for example, inability to control bleeding.
▪ Acute fetal distress.
▪ Imminent delivery, especially in a vehicle that does not allow access to both the mother and child.
▪ Lack of maternal and neonatal experience by the transport team or no experienced personnel available to accompany the team.
▪ Hazardous weather conditions that may prolong the transport time.
GENERAL STRATEGIES FOR TRANSPORT
The primary survey and obstetric physical assessment should be completed in a very short time because of concerns with the crew and the patient being out of belts for adjustment of the external fetal monitor. The primary survey is completed as it is in every other patient. The obstetric assessment is completed in the secondary survey. Pertinent information obtained from the patient may be gathered as the situation permits during the course of the transport. In a life-threatening situation for the mother, the fetus, or both, lifesaving measures must take precedence. During transport, or preferably before, the team should perform the following assessments and interventions3,8,19:
1. Place the patient in a left lateral or right lateral recumbent position or displace the uterus with a wedge if the patient cannot be turned. Displacing the uterus from the inferior vena cava provides optimal return to the heart for maintenance of adequate mean arterial pressure (MAP).
2. Note the patient’s temperature if possible. The American College of Obstetricians and Gynecologists (ACOG) recommends assessment of vital signs every 15 minutes for any patient in labor or at high risk.
3. Note fetal heart tones (FHTs). Initiate continuous electronic fetal monitoring (EFM) if available and applicable. Concerns for continuous fetal monitoring en route include vibration and movement that cause an intermittent fetal heart rate (FHR) tracing, the EFM monitoring of maternal heart rate and not the fetal heart rate, and the need for the crew members and patient to be out of belts for monitor adjustment. FHR assessment with Doppler scan should be performed at least every 15 minutes per ACOG guidelines. Document fetal movement as either present or absent. Documentation should also include current fetal movement patterns to previous fetal movement patterns. For example, is the fetus moving the same amount, more or less compared with usual movement patterns? Decreasing fetal movements may indicate progressive hypoxia and decreasing perfusion. In addition, the uterus must be palpated for contractions with assessment for frequency and duration and subjective assessment of strength.
4. Start an intravenous line with a large-bore 18-gauge or 16-gauge catheter and blood tubing. Use lactated Ringer’s or a 0.9 normal saline (NS) solution with an infusion rate of up to 125 mL/h or titrate volume with consideration for renal, cardiac, and pulmonary status. A maintenance solution of D5LR may be used with patients who do not need intravenous fluid (IVF) boluses.
5. Provide supplemental oxygen with a nonrebreather mask as indicated by FHR pattern or maternal condition.
6. Monitor oxygen status with pulse oximetry, maintaining a level of 98% to 100%.
7. Have a qualified clinician assess uterine contractions and cervical status before departure. Although the assessment is subjective, a commonly used tool is palpation of the fundus during contractions. If at the peak of the contraction, the fundus palpates similar to the consistency of a nose, the contraction is considered mild. If the contraction palpates as firm as the chin, it is considered a moderately strong contraction; and if it palpates as firm as a forehead, it is considered a strong contraction. Keep in mind that this assessment is subjective and patient perceptions should also be taken into consideration. Sterile vaginal examinations (SVEs) may be inappropriate for patients with ruptured bag of membranes or for concerns with infection. Sterile speculum examinations are more appropriate for this patient population.
8. Note and quantify any bleeding. Blood loss should be assessed as objectively as possible. Use pad counts, weighing of blue chux pads, or actual description on the pad (i.e., a 1 × 4-inch stain). Clots can be measured in a graduated cylinder or suction canister. Assess whether blood loss is associated with any pain or contractions.
9. Observe for leaking of fluid vaginally. Note the color and odor of the fluid and presence or absence of contractions.
Emotional and psychologic support provided to the obstetric patient at risk and her family is as vital an aspect as the emergency care provided. The team should encourage the patient to express and verbalize her anxiety, fear for the fetus, and concern regarding complications. The transport team should assess the patient’s knowledge of the situation, encourage questions, and use the opportunity for patient education. The vocabulary used should be based on the education and employment background of the patient. Because most patients have never been transported via air or ground ambulance, the team should explain all medications, procedures, and equipment to allay apprehension about the unfamiliar circumstances. The team should also reassure family members about the current condition of the patient and answer any questions they may have regarding the diagnosis, treatment, or destination. This is true of all patients.
INFERIOR VENA CAVA SYNDROME
The inferior vena cava (IVC) was once described by a cardiovascular surgeon as having the consistency of a wet paper towel roll. Use this description to imagine the weight of the fetus, amniotic fluid, placenta, and uterus compressing the inferior vena cava. Keep in mind that the IVC is responsible for transporting venous blood from the lower part of the body back to the right atrium where it can travel through its normal cardiac flow and then be returned to the central circulation as oxygenated blood. When a gravid (pregnant) patient lies supine, she quite effectively compresses the IVC. Venous blood return is dramatically decreased, which causes a logical decrease in cardiac output. If blood is not brought to the heart, the heart does not have volume to pump out. This condition is called the inferior vena cava syndrome (IVCS). IVCS greatly mimics hypovolemic shock. Patients present with hypotension (decreased cardiac output [CO]), reflexive tachycardia (as a result of decreased CO), skin parameter changes (cool diaphoretic skin), and potential mental status changes (decreased cerebral blood flow leads to nausea, vomiting, and decreased level of consciousness [LOC]). Decreased blood flow to the uterus occurs secondarily. The limited amount of blood is shunted to the mother’s vital organs, sacrificing the fetus. Unfortunately for the fetus, the uterus is not considered a vital organ. If fetal monitoring is in place, signs of fetal distress are seen as a result of hypoplacental perfusion. Luckily, for awake and alert patients, IVCS is rarely a problem because they cannot tolerate lying supine if the weight of the gravid uterus is enough to compress the IVC. The patients experience nausea and naturally flip themselves to their sides. This action increases the patency of the IVC, and the patients feel better as venous blood is returned. The greatest potential problem occurs when the pregnant patient is obtunded or unconscious, for example, a trauma patient. Appropriately, EMS puts these patients on a LBB and cervical collar. A towel roll under the backboard in the area of the patient’s hip relieves the compression of the IVC. At this time, the patient’s condition should dramatically improve as blood is now able to return to the heart. If the patient is indeed in hypovolemic shock, then an objective assessment can now be made (e.g., left or right side). For years, we have all learned that the pregnant patient has to be on her left side to increase venous and placental blood flow. The IVC lies slightly to the right midline under the uterus. In theory, if the patient is turned to the left, the IVC is the most patent. We now know that the IVC is also patent if the patient is tilted or turned to the right. In addition, if the patient cannot be turned, the uterus can be manually displaced by gently pushing or pulling it to the left and off the IVC. IVCS is the most common mistake healthcare professionals make in the care of the pregnancy patient. We are not accustomed to “tilting” our patients, and this condition is often forgotten. Prevention is key because these patients already have compromised conditions. This is a great example of how an intervention cannot only improve the maternal condition but can also save the life of the fetus.
FETAL MONITORING BEFORE AND DURING TRANSPORT
Fetal monitoring may be accomplished with intermittent Doppler scan auscultation, which is used most frequently for short transports, and with EFM, used for longer transports (approximately 30 minutes or more). An external ultrasound scan device records FHTs, and a tocodynamometer detects subjective uterine activity. 11,12,16,17 The mode of FHR assessment, be it continuous or intermittent via Doppler scan, is often program and institution dependant. One has not been clearly identified over the other as superior during transport. The ACOG does not make a statement on which method of fetal monitoring is recommended. However, they do state that FHR assessment should be done a minimum of every 15 minutes during transport.
Continuous assessment of fetal well being is best accomplished with electronic fetal monitoring. FHTs are recorded simultaneously with uterine activity. The EFM works by sending ultrasound waves into the uterus; the monitor detects the fastest moving objects, which are typically the valves on the fetal heart. This information or sound is returned to the monitor where it is converted to a number on the graph paper. The monitors have a program designed to detect heart rates between approximately 50 and 190 bpm. A limitation of the EFM is that if the FHR nears the lower limits of the monitor capacities the EFM may erroneously detect the motion of one heart beat as two separate beats. This error is called “doubling” the FHR; the monitor paper graphs the FHR at twice the actual heart rate. Conversely when the FHR nears the upper limits, the monitor may only recognize every other motion as a heartbeat and “halving” of the FHR occurs. So, how does one accurately assess the FHR with an EFM? The key is the audible signal on the monitor. The audible signal of the monitor remains accurate and can be compared against the printed FHR tracing to confirm accuracy. In addition, the maternal pulse should be compared with the FHR tracing to confirm that the maternal signal is not being traced. This error has occurred in circumstances in which the fetus is dead in utero and the EFM then monitors the maternal aorta fluctuations and misinterprets them as a fetal heart rate. Subtle changes in the FHT are often the earliest indication of hypoxia caused by uteroplacental insufficiency or umbilical cord compression. Recognition of normal FHR tracing permits abnormalities to be realized quickly. Appropriate intervention should be aimed at correcting or alleviating the source of insult; at a minimum, actions should improve placental/uterine perfusion.
Historically, conflicting interpretations have been seen. Most of this conflict has revolved around nomenclature, classification, and significance of EFM patterns. As a result of these inconsistent interpretations, the National Institute of Child Health and Human Development (NICHD) developed a defined terminology and classification system. This system was later adopted by the ACOG and Association of Women’s Health, Obstetric and Neonatal Nurses (AWHONN). This chapter reflects all of the NICHD nomenclature.
Baseline Assessment
The first parameter to be assessed is the FHR baseline. The baseline is the average of the FHR and typically is between 110 and 160 bpm. The baseline is assessed over a minimum 10-minute period. In addition, it is assessed between contractions and periodic or episodic changes. Periodic changes are accelerations or decelerations that occur to the fetal heart rate as a result of contractions. Episodic changes are accelerations/decelerations that occur in the FHR as a result of fetal movement. The FHR baseline is the approximate mean FHR rounded to increments of 5 bpm over a 10-minute period. In any 10-minute section, a minimum of a 2-minute period of no contractions or periodic or episodic changes is needed to determine the FHR baseline. Evaluation of the FHR baseline is a critical step in FHR interpretations. It allows interpretation of trends that occur in the baseline and can reflect subtle changes in the fetal environment.
Fetal Heart Rate Abnormalities
Variability
Fluctuations in the FHR reflect interplay between the sympathetic and parasympathetic branches of the autonomic nervous system. A constant pull from the sympathetic nervous system increases the heart rate and from the parasympathetic nervous system decreases the heart rate. These normal variations between each fetal heartbeat give the FHR tracing its “squiggly” appearance. Variability is the single most important factor in prediction of fetal well being, based on EFM monitor interpretation. Fetal movement remains the number one nonmonitor indicator of fetal well being. Similar to assessment of the baseline, variability can only be assessed between contractions and episodic and periodic changes (Figure 31-1).
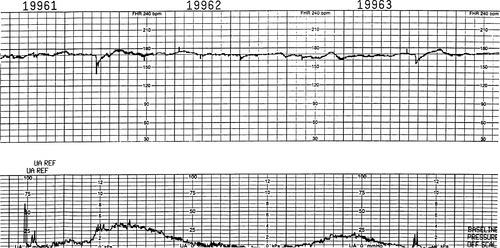 |
| FIGURE 31-1 Reduced variability and tachycardia. Note the almost absent beat-to-beat variability and reduced long-term variability as recorded by fetal scalp electrode; also note the tachycardic baseline. |
Moderate variability is indicative of an adequately oxygenated, normal PH, mature and intact autonomic nervous system. Moderate variability is defined as fluctuations in the FHR that range between 6 and 25 bpm. The presence of moderate variability is reassuring because it indicates that the fetus is tolerating blood flow changes within the uterus.
Minimal variability is defined as fluctuations in the FHR that are equal to or less than 5 bpm. This variability is often associated with fetal hypoxia and acidosis. However, benign or expected minimal variability can occur in certain clinical situations; for example, the fetal variability can decrease if the mother has received any narcotics. Narcotics are a central nervous system (CNS) depressant; anything that decreases the maternal CNS can decrease fetal CNS as it passes through the placenta. As the maternal drug is metabolized and excreted, the effects on the fetus should also be seen to diminish. Persistent minimal variability is not associated with maternal drug use, and other causes of decreasing variability must be assessed. Another common reason for minimal variability is immaturity in gestational age, which is associated with CNS immaturity. Fetuses that are less than 32 weeks gestation show less variability because the autonomic nervous system (ANS) may not yet be fully developed. Fetal arrhythmias and cardiac or CNS anomalies may also be responsible for minimal or absent variability. These causes show variability changes from the time monitoring is initiated. The most common reason for benign minimal variability is fetal sleep patterns. The fetus has frequent sleep periods that range from 20 to 40 minutes. A key assessment pearl is that although fetal sleep patterns are common, they are transient in nature and rarely last longer than 40 minutes. In addition, moderate variability should be documented before and after the sleep pattern. As blood flow decreases across the placenta or umbilical cord, less oxygen is available for the fetus. If the hypoxic events are corrected, the fetus usually tolerates them well. However, if the cause of decreased perfusion is not corrected, an eventual decrease in variability is noted. Careful evaluation of the FHR tracing reflects trending of decreasing variability (minimal or absent), which is a warning sign that the fetus is losing compensatory mechanisms and fetal hypoxia and acidosis are increasing. Interestingly, the incidence rate of actual fetal acidemia in the presence of minimal or absent variability and decelerations is only 23%. 16 However, the presence of these signs still remains an indicator for intervention. This intervention includes measures to increase placental and uterine perfusion, such as maternal position change, intravenous (IV) fluid bolus to increase maternal volume/perfusion, and application of supplemental O 2.
Increased or marked variability of more than 25 beats of fluctuation may be one of the earliest signs of hypoxia. Although it may also be an indication of increased fetal activity, constant assessment and reevaluation is necessary. If continuous EFM is used during transport, the clinician should keep in mind that a greater degree of variability may be recorded than is actually present because of vibrations from the vehicle. Special notice of long-term variability and other reassuring signs must be made, including the presence of fetal movement. Variability should be assessed with NICHD terminology such as moderate, minimal, absent, or marked (see Table 31-1).
| NICHD Terminology | Fluctuations | Indications |
|---|---|---|
| Moderate | 6-25 bpm | Well-oxygenated, nonacidotic, intact/mature CNS |
| Minimal | ≤5 bpm | Hypoxia, acidosis, sleep patterns, maternal drug use, cardiac or CNS insult, prematurity |
| Absent | Undectable from baseline | Hypoxia, acidosis, sleep pattern, maternal drug use, cardiac or CNS insult |
| Marked | >25 bpm | Fetal movement or early hypoxia |
Periodic Changes/Episodic Changes
Periodic changes are changes that occur in the FHR tracing related to uterine contractions. Episodic changes are changes in the FHR tracing that are not related to uterine contractions and are often related to fetal movement. The FHR may accelerate, decelerate, or not respond.
Acceleration.
Accelerations above the baseline are usually associated with fetal movement but may occur during contractions (Figure 31-2). Because the hypoxic fetus with metabolic acidosis is unable to accelerate its heart rate, accelerations are viewed as a sign of fetal well being. The true definition of acceleration is a transient increase above the baseline greater than 15 bpm for 15 seconds or longer and typically lasting less than 2 minutes in duration. This definition is applied for fetuses that are greater than 32 weeks in gestation. The definition of acceleration for a fetus younger than 32 weeks is a transient increase over baseline of greater than 10 bpm for at least 15 seconds and typically less than 2 minutes. Uniform accelerations are accelerations that occur with each contraction and are uniform in shape and size. This acceleration pattern may be associated with breech presentation or early and mild cord compression. In very early labor, the contractions are not strong and the fundus or top of the uterus gently compresses the breech during contractions, which causes a sympathetic response and accelerations are noted. Later, as the contractions increase in strength and the head of the fetus is pushed down into the pelvis, uniform decelerations are seen, discussed in the section on early decelerations.
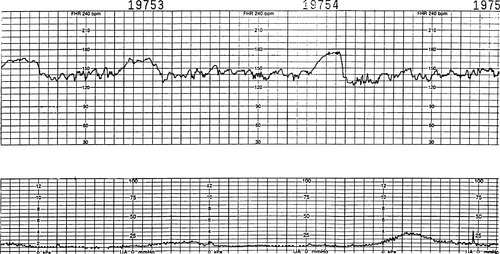 |
| FIGURE 31-2 Accelerations. With use of an ultrasound scan transducer, accelerations of approximately 20 beats above the baseline may be noted. Long-term variability is present, and the FHR baseline range is approximately 135 to 145 bpm. |
Although the presence of accelerations and moderate variability are excellent monitor signs of fetal well being, fetal movement remains the best nonmonitor indicator of fetal well being. Only well-oxygenated fetuses with normal pH levels have consistent fetal movement patterns. Certainly all fetuses have different movement patterns that are unique to themselves. During transport, the clinician should assess the current movement pattern compared with the usual movement pattern. For example, a fetus that is known to have movements of at least 15 times an hour that is now reported to have only moved once in the past 2 hours shows a significant deviation from normal patterns and further assessment and documentation are necessary. The transport team should take note of fetal movements, whether the mother has noticed a decrease, increase, or no change in fetal movement. Decreased fetal movement is indicative of hypoxia. Although a decrease in fetal movement may be anticipated, such as in the event of maternal trauma and blood loss, the transport team is expected to inform the maternal fetal medicine physicians of this change in fetal status.
Variable Deceleration.
Variable decelerations can occur at any time during a contraction (Figure 31-3). The shape may also vary and is frequently V-shaped or W-shaped. The decelerations are known as variable because of the varied shape and timing. Cord compression is typically responsible for these decelerations. However, they may also occur from head compression as a result of vagal stimulation in the second stage of labor (pushing). Physiologically, as the cord is compressed, blood flow through the umbilical cord is decreased. Baroreceptors cause a short increase in the FHR to compensate. The blood flow is further impeded, and a sharp decrease occurs in the FHR. As the umbilical cord compression is relieved, the FHR responds with a quick increase above the baseline in an attempt to increase oxygenation status. After a short period of time, typically less than 5 to 10 seconds, the FHR returns to baseline with moderate variability. Initially, variables may have a characteristic appearance; frequently, a short acceleration is observed, followed by a rapid deceleration for some seconds and then a rapid rise and a short acceleration before a return to the FHR baseline. The short period of increased FHR before and after the deceleration is known as shoulders. Shoulders are associated with moderate variability and indicate that the fetus still has the ability to compensate with varying amounts of placental blood and available oxygen. Shoulders are considered a reassuring characteristic associated with variable decelerations. If the fetus continues to have variable decelerations that are progressively more consistent or deeper in nadir, often this compensatory response is lost and only the sharp deceleration is seen without shoulders noted. The abrupt onset and sharp decline with usual rapid return to baseline makes these decelerations easily identifiable. Cord compression may occur in a variety of circumstances. After the membranes rupture, less fluid is available to cushion the cord. Variables usually occur in response to uterine contractions but also may occur in response to fetal movement in the absence of contractions when membranes are ruptured. If a nuchal cord, short cord, or cord entanglement is present, variables usually result.
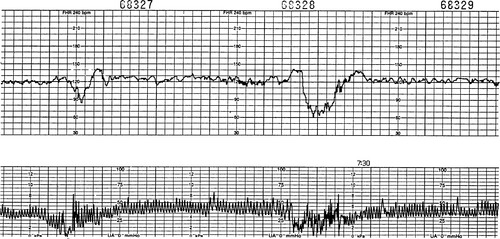 |
| FIGURE 31-3 Variable decelerations. Note the variable decelerations in the presence of average variability. The accelerations before and after the deceleration (also called shoulders) are a reflection of adequate variability. |
In the past, these decelerations have commonly been described as mild, moderate, or severe, depending on the drop in FHR. However, to remain consistent with NICHD nomenclature, a more conclusive method is to describe the deceleration. This description should include the depth (nadir), duration, shape, and return variability status. A better indicator of the fetal response is reflected in the FHR baseline, variability, and changes in the variable decelerations. Signs that the fetus is losing its ability to tolerate the stress of repeated cord compression or that the cord compression is becoming more severe include a deeper deceleration that lasts longer, a slow return to baseline, loss of shoulders, and decreased variability. Overshoots are another type of acceleration seen with variable decelerations. These differ from shoulders in the sense that these accelerations are seen only after the variable deceleration as the compromised fetus attempts to improve oxygenation status. These off shoots are typically well above baseline. Offshoots are associated with minimal to absent variability and are considered nonreassuring . In interpreting the tracing, careful observations of any changes in FHT reveal more than what has just occurred during the last contraction.
Late Decelerations.
Late deceleration begins at or after the apex (peak) of the contraction. They gradually decelerate in a uniformed “U” shape and return to the FHR baseline well after the contraction is over (Figure 31-4). By definition, late decelerations must be recurrent, which means they occur in greater than 50% of all uterine contractions (National Institute of Child Health and Human Development [NICHD]). 3 Physiologically, during a normal contraction, a minimal amount of oxygen always crosses the placenta. A healthy fetus can tolerate this decrease in oxygen via its oxygen reserves. A compromised fetus that has experienced prolonged or chronic hypoxia does not have oxygen reserves available and therefore cannot tolerate the decrease oxygen availability found during contractions. Not until the contraction is over and the maternal-fetal exchange can resume does the FHR return to baseline. Late decelerations always mean uteroplacental insufficiency: either the placenta, the fetus, or the uterus presents with conditions that interfere with normal exchange of oxygen between the mother and fetus. Use the analogy of a child being dunked in a pool. The child tolerates the dunking early on, but if it continues, the child becomes fatigued, loses O 2 reserve, and ends up gasping every time he surfaces. When a contraction is stronger, the insufficiency is greater and the deceleration is proportional. However, “size does not matter.” With severe hypoxia, the myocardial depression may be such that the heart is unable to decelerate in response to the stress of the contraction, and very subtle late decelerations are seen accompanied by a flat FHR baseline. Simply stated, the fetus becomes so hypoxic and acidotic that it cannot render a response to the impeded blood flow. The fetus is too sick to “wave the white flag.” A FHR tracing that is tachycardic with minimal or absent variability and subtle late decelerations is considered an ominous FHR tracing and interventions must be made to improve fetal oxygenation. These interventions include measures to increase placental perfusion, such as maternal position change, supplemental O 2 via a nonrebreather mask, IVF boluses, and possible delivery, if improvement is not noted.
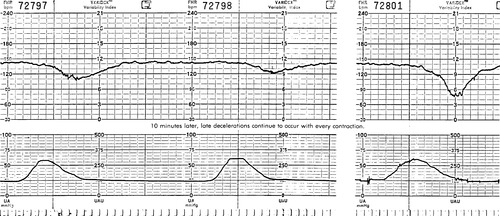 |
| FIGURE 31-4 Late decelerations. Note the onset of deceleration at the apex of the contraction. Also note the minimal variability, slow recovery, and the proportional deceleration observed. |
Uteroplacental insufficiency may result from numerous maternal and fetal conditions, such as pregnancy-induced hypertension (PIH), diabetes mellitus (DM), cardiovascular or kidney disease, chorioamnionitis, smoking, a fetus that is past maturity, and fetal hydrops. Uteroplacental insufficiency may also result from decreased placental perfusion in placental abruption or previa, uterine hypertonus as a result of oxytocin stimulation, and hypotension. As with variable decelerations, evaluation of late decelerations with respect to FHR baseline, variability, and changes noted over time is necessary in evaluation of the well being of the fetus. Even an otherwise healthy well-oxygenated fetus can experience late decelerations in the presence of acute hypotension and hypoxia. An example of this is a mother that is obtunded and was laid flat on her back on a back board. This position alone can cause IVCS and rapid decrease in maternal venous blood return, which then causes maternal hypotension that leads to profound fetal hypotension and results in late decelerations. Once the maternal position is tilted to the right lateral position (RLP) or left lateral position (LLP), the blood flow improves and the late decelerations resolve.
Early Decelerations.
Early decelerations are innocuous decelerations that begin close to the beginning of the contraction and end close to the end of the contraction. These decelerations appear to mirror the contractions. As the head is compressed, the vagus nerve is stimulated and causes a parasympathetic response that leads to a deceleration. The deceleration ends as the contraction ends because the vagus nerve (head) is no longer compressed. Early decelerations are usually associated with moderate variability and are considered benign in nature. These decelerations frequently occur in active labor when the cervix has dilated 4 to 7 cm, or they may be seen in early labor with breech presentation because, in this position, the head is compressed into the pelvic cavity with contractions. A transport consideration is that a sterile vaginal examination (SVE) or sterile speculum exam (SPE) should be preformed before transport to rule out advanced cervical dilation. In FHR interpretation, late decelerations should not be confused with early decelerations. Accurate placement of the tocotransducer over the fundus ensures that contractions are recorded correctly. Also, consider other reassuring attributes such as moderate variability and the presence of accelerations or fetal movement to help discern early from late decelerations.
Sinusoidal.
Sinusoidal is a rare FHR pattern in which a uniform sine wave pattern occurs. This FHR remains within normal baselines but has an obvious unusual appearance. It is often described as an undulating pattern that is saw-toothed in appearance. Possible causes of this pattern are fetal hypovolemia or anemia; it may occur in cases of erythroblastosis fetalis, accidental tap of the umbilical cord during amniocentesis, fetomaternal transfusion, placental abruption, or another type of accident. Variability is absent or minimal, and accelerations are not seen. When this pattern is recognized, rapid delivery is usually recommended unless the underlying pathology can be corrected. For example, in the case of severe fetal anemia, a blood transfusion can be administered to the fetus via a percutaneous umbilical cord sampling (PUBS) procedure. Once the anemia is resolved, the sinusoidal pattern is resolved. However, a more commonly observed FHR pattern is a pseudosinusoidal or undulating pattern that is not pathologic (Figure 31-5). This pattern is linked to maternal drug administration, both prescribed and illicit.
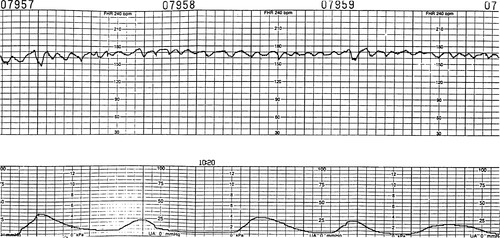 |
| FIGURE 31-5 Sinusoidal pattern. Note the jagged, nonuniform pattern observed after intravenous administration of butorphanol (Stadol), 1 mg, which resolved spontaneously after 20 minutes. |
Bradycardia.
An FHR of less than 110 bpm for a period of 10 minutes or longer is defined as bradycardia. Bradycardia can occur as result of numerous acute or chronic conditions and can be hypoxic or nonhypoxic in nature. Many term fetuses and those past maturity may have a stable baseline between 100 and 120 bpm, reflecting a more mature fetal neurologic system that is more parasymphathetically controlled. In the absence of hypoxia, adequate variability and accelerations are also noted.
Bradycardia is a response of increased parasympathetic tone and is reflected by a decrease in fetal cardiac output in the presence of hypoxia. The fetus can tolerate sustained bradycardia for only a short length of time before becoming acidotic. Bradycardia can be acute as a result of severe cord compression. It can occur minutes before delivery when the cord is drawn into the pelvis in the second stage or with a cord prolapse. Bradycardia can also occur with hypertonic or tetanic contractions as seen with placenta abruptio and any event that causes maternal hypotension. In the presence of chronic hypoxia, bradycardia is usually a late occurrence. Evaluation of variability determines how the fetus tolerates the stress.
Tachycardia.
A FHR of more than 160 bpm for a period of 10 minutes or longer is considered tachycardia. Tachycardia is a response of increased sympathetic tone and is reflected by a compensatory mechanism to increase cardiac output in the presence of transient hypoxia. A decrease in variability is generally associated with tachycardia. This decreased variability and increased rate is reflective of the fetus returning to a more primitive or sympathetic response. Fetal tachycardia may be maternal or fetal in origin. Factors that contribute to tachycardia include smoking, maternal fever, use of beta-sympathomimetic agents, fetal anemia, fetal hypovolemia, fetal tachydysrhythmias, chorioamnionitis, and maternal hyperthyroidism.
Fetal distress is a loose term that implies that the fetus is in danger of hypoxia and metabolic acidosis. Late and variable decelerations are sources of stress to the fetus, whereas variability is an indicator of how the fetus tolerates the stress.
Assessment of FHR tracings should follow a systematic approach with these sequential steps:
Assessment of baseline.
Assessment of variability with NICHD standards.
Assessment of periodic/episodic changes, including accelerations or decelerations.
Assessment for the presence of tachycardia or bradycardia.
At any time that the transport team considers that the FHR tracing is not reassuring, medical control or receiving maternal fetal medicine physicians must be contacted for further orders. Interventions should be made to improve placental perfusion, including maternal position change, supplemental O 2 via a nonrebreather mask, and IVF boluses. If the transport team is unsure of the FHR interpretation, the FHR strip can be faxed to the receiving facility for interpretation. If the FHR is not reassuring en route, medical control should be contacted (if possible) and diversion should be considered for signs of ominous fetal distress.
Nonreassuring signs of fetal well being include a significant increase or decrease in the FHR baseline during a period of several hours, a wandering baseline, a spontaneous decrease in variability or a decrease in variability as labor progresses, bradycardia or tachycardia with reduced variability, subtle late decelerations, or any combination of these signs.
Abnormal FHR tracings are observed in situations of congenital anomalies. Frequently, variability is reduced or absent, and tachycardia or bradycardia may be noted. Table 31-2 summarizes comparative signs of acute and chronic distress. Whatever the mechanism of insult to the fetus, the plan of action for possible fetal distress is intrauterine resuscitation.
| IUGR, Intrauterine growth restriction. | |
| Chronic Distress (Occurs Over Time) | Acute Distress (Occurs Suddenly) |
|---|---|
| Mechanism of insult | |
| Uteroplacental insufficiency | Umbilical cord compression or uteroplacental insufficiency |
| Signs of IUGR, decreased fetal movements | Initially, no indication of fetal compromise |
| Contributing factors | |
| PIH (preeclampsia) | Cord prolapse |
| Cardiac or kidney disease | Placental abruption |
| Severe anemia | Hypotension (vena cava compression, epidural anesthesia, hemorrhage) |
| DM (class B-R) | Hypertonic contractions |
| Postdate pregnancy | Placenta previa with hemorrhage |
| Rh isoimmunization | |
| Chorioamnionitis | |
| Smoking | |
| Fetal response (progression differs depending on circumstances) | |
| Tachycardia | Variable decelerations |
| Increased variability | Prolonged decelerations |
| Decreased variability | Tachycardia |
| Late decelerations | Increased variability |
| Bradycardia | Decreased variability |
| Late decelerations | |
| Bradycardia | |
The key formula (LOCK) is as follows:
L:Place the patient in the left lateral or right lateral recumbent position.
O:Provide supplemental oxygen, via 100% nonrebreather mask.
C:Correct or improve contributing factors.
K:Keep reassessing the FHR and intervene when indicated.
Contributing Factors to Fetal Distress
Interventions that must be performed by the transport team when signs and symptoms of fetal distress are present are as follows:
1. Hypotension: Initiate a 500-mL intravenous fluid bolus, depending on the condition of the patient. If the patient has no comorbidities that promote pulmonary edema, the maternal patient can receive 2 L of crystalloids to improve maternal and placental perfusion. Correct for supine hypotension with a change to the left lateral or right lateral position, uterine displacement, or a towel roll under the backboard.
2. Hypertonic or tetanic contractions: Consider the use of terbutaline, 0.25 mg, administered subcutaneously or via intravenous push. Check to ensure that the patient’s heart rate is less than 120 bpm before administering the medication. The terbutaline can be repeated every 10 minutes for three doses; however, program protocol should always guide therapy.
3. Rule out cord prolapse. A sterile vaginal examination is used to confirm the presence of a cord. Lift the presenting part of the cord to relieve the cord compression and reposition the patient, following recommendations provided in this chapter.
4. Assess for placental abruption or other complications that may affect the FHR.
5. Change the position of the mother. If the left lateral position does not relieve the cord compression as indicated by continued variable decelerations, reposition the mother to the right side, to the hands and knees, or last, to the knee-chest position. A transport consideration is the inability to restrain the maternal patient in the knee-to-chest position in some vehicles, especially small helicopters.
6. Assess for signs of maternal hemorrhage. Changes in FHR, including tachycardia and loss of variability, are often the first signs of maternal blood loss. The maternal patient has 40% to 50% more blood volume at term and therefore can mask signs of hypovolemia. In addition, the maternal patient shunts blood to her vital organs, and the uterus is not considered a vital organ.
If the patient is located in an outlying area where the transport time is expected to be lengthy, evaluation of the FHT for reassuring signs of fetal well being aids in the decision to transport the mother or to deliver the fetus at the referring facility to increase the chance of fetal survival. With this in mind, the accepting maternal fetal medicine physicians make the ultimate decision of whether or when to transport. At times, the primary reasons for transport are maternal and fetal conditions that cause a nonreassuring FHR tracing. In these circumstances, a nonreassuring FHR strip does not preclude or exclude maternal transport. Appropriate indications for maternal/fetal transport are if the referring facility is unable to offer specialized or high-risk obstetric services or a neonatal intensive care unit (NICU). Likewise, if the transport is expected to be short and the time needed by the referring facility to prepare for a cesarean section is longer than the estimated transport time, maternal transport is recommended. The intent of the transport is to attain the most expedient delivery of the mother and fetus in a facility most capable of dealing with the mother and fetus at risk. Studies show improved neonatal outcomes if the fetus is delivered at a perinatal network with a level III nursery.
The transport team may consider use of Doppler scan auscultations or EFM for transports. Advantages and disadvantages are found with both methods. Doppler scan auscultation is performed a minimum of every 15 minutes. This method does not allow for assessment of variability but can assess for FHR and responses to contractions if auscultation is performed during and after contractions. Continuous EFM allows for assessment of variability; however, vibration of the aircraft may artificially add artifact. Remember that this can be a problem in any transport vehicle. Because of position constraints in the aircraft, the EFM may slip and monitor maternal heart rate, which causes the transport team and patient to frequently be out of belts so the monitor can be adjusted. The FHR strip should clearly identify when maternal heart rate is monitored so it is not later assessed to be fetal distress. Trained and competent individuals must accomplish this monitoring. The age-old controversy with continuous fetal monitoring during transport is what to do with the information found. For example, if the fetal condition acutely deteriorates into fetal distress, is there adequate time to divert to another facility? Does diversion to another facility outside of the level III hospital offer services other than the referring facility? Both forms of monitoring are accepted by the ACOG as appropriate and safe. Regardless of the method chosen, if the FHR reflects acute or chronic signs of deterioration or distress, appropriate interventions must be made to alleviate the cause and to increase uterine/placental perfusion. These interventions and their responses need to be documented.
NORMAL PHYSIOLOGIC CHANGES IN PREGNANCY
For true appreciation and recognition of the pathologies that may be associated with pregnancy, a review of normal physiologic changes is necessary. This review of systems is brief and highlights key areas. 5,6,10,14,19
Airway: The failure rate with oral intubation is four times higher in the pregnant patient than in the nonpregnant patient. The trachea of the term pregnant patient tends to be anterior, and the epiglottis is reported to be friable.
Hematalogic: By term, the maternal blood volume has increased by 40% to 50%. The greater increase is in plasma then in red blood cells (RBCs), platelets, etc. As a result, a normal dilutional anemia is often associated with pregnancy. In cases of maternal blood loss, maternal hypotension is often not noted until the patient has lost approximately 30% of the blood volume because of the extra volume created during the pregnancy. Platelets and fibrinogen slightly increase during pregnancy and cause the pregnant patient to be in a hypercoagulable state, which allows faster and more efficient clotting to prevent hemorrhage after delivery.
Respiratory: Tidal volume increases by about 40%, and respiratory rate slightly increases; thus, pregnant women have compensated respiratory alkalosis. Alveolar ventilation increases by 65%.
Cardiac: CO increases by about 50% (related to the increased blood volume). Heart rate (HR) slightly increases by about 10 beats by term. Blood pressure is normotensive (for the patient) in the first trimester, decreased in the second trimester, and returned to normotensive in the third trimester. This change is greatly related to a pregnancy hormone called relaxin. A key assessment point is that the blood pressure should never be elevated over 140/90 mm Hg during pregnancy. If it is, a pathology is always indicated.
Gastrointestinal: Slowing of peristalsis and resulting constipation occur. The stomach empties slowly, and the pregnant patient is at a high risk of aspiration with altered levels of consciousness. This effect is also related to the hormone relaxin. Because of this effect, a pregnant woman is always considered to have a full stomach no matter when her last meal was. Increased salvation is common and frequent suctioning may be needed in the case of oral intubation. An increased incidence of cholelithiasis is found during pregnancy.
Renal: Increased renal filtration of glucose and sodium occurs during pregnancy. BUN and CR levels are both lower during pregnancy. Elevated levels of either renal function laboratory tests indicate pathology. Protein is a large molecule and should not be passed renally in the absence of pathology.
Uterus: The uterus becomes the largest intraabdominal organ. Uterine and placental perfusion increases to 600 to 800 mL of blood per minute at term. A very high risk of maternal hemorrhage is found in the presence of uterine or placental injury.
Musculoskeletal: The abdominal viscera become stretched and distended because of the growing uterus. These distorted viscera may cause abdominal pain to be referred. The effects of the hormone relaxin also cause the symphysis pubis cartilage to slightly separate, thereby increasing pelvic instability. The gravid uterus causes the patient’s center of gravity to be altered, and an increase in falls may be noted. The thoracic cavity also expands during pregnancy to allow greater lung expansion because the lungs have less distance to elongate as a result of the gravid uterus.
Liver: The liver is the only organ to not increase efficiency during pregnancy. Hepatic function values remain the same as nonpregnant values. However, if elevations of serum glutamate oxaloacetate transaminase (SGOT)/serum glutamate pyruvate transaminase (SGPT), or aspartate aminotransferase (AST)/alanine aminotransferase (ALT), are noted, significant pathology is implied.
COMPLICATIONS OF PREGNANCY AND DELIVERY
In the following discussions regarding complications of pregnancy and delivery, general obstetric assessment and implementation of general guidelines for transport are assumed to have already been performed, including assessment of fetal well being.
Amniotic Fluid Embolism/Anaphylactic Syndrome of Pregnancy
Amniotic fluid embolism is now known as anaphylactic syndrome of pregnancy. Previous thought was that amniotic fluid gained access to the maternal circulation during labor or delivery or immediately after delivery, resulting in obstruction of the pulmonary vasculature. In addition, particulate matter in the amniotic fluid, such as meconium, lanugo hairs, fetal squamous cells, bile, fat, and mucin, was also thought to embolize. The belief now is that the process is more likely to be an anaphylactic reaction to the amniotic fluid and the fetal cells they contain. Treatment is primarily supportive and usually necessitates immediate intubation, mechanical ventilation, pressor administration, and treatment of coagulopathy. In the United States, amniotic fluid embolism causes 10% of maternal deaths. The complication is very rare and is frequently fatal, with a maternal mortality rate nearing 90%. Amniotic fluid embolism is often initially misdiagnosed as result of the vague clinical picture of surviving patients and missed autopsy findings in fatal cases. Unfortunately, the rapid progression of this syndrome is associated with a high maternal mortality that is often not diagnosed until postmortem.
Etiology and Pathophysiologic Factors
The route by which amniotic fluid enters the circulatory system of the mother is not clear. The most frequently suggested sites of entry are lacerations in the endocervical veins during cervical dilation and lacerations in the lower uterine segment, the placental site, and uterine veins at sites of uterine trauma. Under the pressure of uterine contractions, amniotic fluid gains access to the circulatory system of the mother and travels quickly to the pulmonary vasculature, where embolization and anaphylactic reaction quickly ensue.
Factors that have been associated with amniotic fluid embolism include uterine rupture, cesarean section, and the use of uterine stimulants to induce labor, which produces hypertonic contractions. Other factors that place the obstetric patient at risk are a large fetus, placenta previa, placental abruption, intrauterine fetal death, meconium in the amniotic fluid, multiparity, precipitous delivery, knee-chest position, and maternal age of more than 30 years.
Disseminated intravascular coagulation (DIC) is a complication that can be expected, although the pathway is unclear. Uterine atony and postpartum hemorrhage are also frequent complications. Acute cor pulmonale, right heart failure, and pulmonary edema follow.
Assessment
Of the predisposing factors that the patient may have, sudden acute dyspnea is the most characteristic symptom, followed by profound cyanosis and sudden shock. Other symptoms may include chest pain, restlessness, anxiety, coughing, vomiting, pulmonary edema with pink frothy sputum, seizures that are frequently confused with eclamptic seizures, and coma. If the patient has delivered, the transport team should watch for symptoms of postpartum hemorrhage caused by uterine atony.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






