Chapter 85
Thyroid and Adrenal Disorders in the Intensive Care Unit
The Nonthyroidal Illness Syndrome
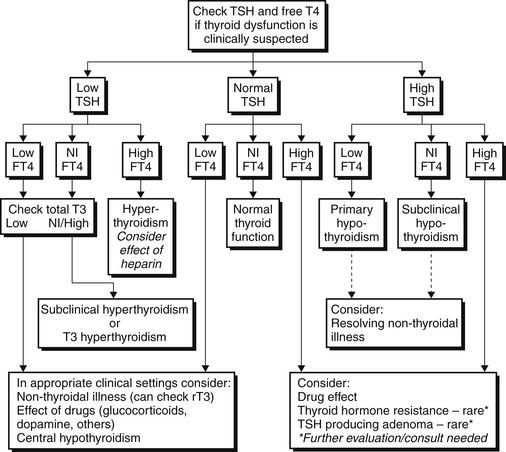
The nonthyroidal illness syndrome (NTIS) is an alteration in thyroid function tests seen in patients with acute or chronic illness with no prior history of thyroid dysfunction. Low serum T3 is the most common abnormality in NTIS; however, decreases in TSH and T4 are seen in more prolonged or severe illness. Elevations in reverse T3 (rT3) may also be evident (Figure 85.1).
Epidemiology and Etiology
Drugs used in the ICU can also affect both thyroid function and thyroid function tests. Corticosteroids and dopamine decrease both basal TSH secretion and pituitary response to TRH. Corticosteroids also decrease the peripheral conversion of T4 to T3. A number of other drugs commonly used in the ICU can affect the thyroid function or the interpretation of thyroid function tests. Drugs that can cause hypothyroidism include lithium, intravenous (IV) iodinated radiographic contrast agents, amiodarone, aminoglutethamide, thalidomide, interferon alpha, and interleukin-2. Drugs that can cause hyperthyroidism include iodine and amiodarone, interferon alpha, denileukin diftitox, and interleukin-2.
Diagnosis
TSH is usually suppressed, necessitating differentiation from hyperthyroidism. TSH is low enough to be undetectable in only 7% of NTIS patients (usually those who have received corticosteroids or dopamine). An undetectable TSH therefore suggests hyperthyroidism. Assessment of T4 and T3 levels differentiates between the two conditions, as these will be elevated only in hyperthyroidism (Figure 85.2).
Thyrotoxicosis
Etiology
Secondary hyperthyroidism is rare and caused by the excessive production of TSH from a pituitary adenoma. In this condition, the thyroid hormone levels are elevated but TSH is elevated or inappropriately normal.
Other causes of hyperthyroidism include excessive consumption of exogenous thyroid hormone which may be surreptitious. Amiodarone may also cause hyperthyroidism, either by increased thyroid hormone production or a destructive thyroiditis. Additional causes of hyperthyroidism are listed in Table 85.1.
TABLE 85.1
| Common Causes | Underlying Etiology | Diagnostic Features |
| Graves disease | Thyroid-stimulating immunoglobulin (TSI) binds to and stimulates the thyroid | Increased thyroid radioactive iodine uptake with diffuse uptake on scan, positive thyroperoxidase antibodies; raised serum thyroid-stimulating immunoglobulin; diffuse goiter; ophthalmopathy may be present |
| Toxic adenoma | Monoclonal autonomously secreting benign; thyroid tumor | Normal to increased thyroid radioactive iodine uptake with all uptake in the nodule on scan; thyroperoxidase antibodies absent |
| Toxic multinodular goiter | Multiple monoclonal autonomously secreting benign thyroid tumors | Normal to increased thyroid radioactive iodine uptake with focal areas of increased and reduced uptake on scan; thyroperoxidase antibodies absent |
| Exogenous thyroid hormone | Excess exogenous thyroid hormone | Low to undetectable thyroid radioactive iodine uptake; low serum thyroglobulin values |
| Painless postpartum lymphocytic thyroiditis | Autoimmune lymphocytic infiltration of thyroid with release of stored thyroid hormone | Low to undetectable thyroid radioactive iodine uptake; thyroperoxidase antibodies present; occurs within 6 months after pregnancy |
| Painless sporadic thyroiditis | Autoimmune lymphocytic infiltration of thyroid with release of stored thyroid hormone | Low to undetectable thyroid radioactive iodine uptake; thyroperoxidase antibodies present |
| Subacute thyroiditis | Thyroid inflammation with release of stored thyroid hormone; possibly viral | Low to undetectable thyroid radioactive iodine uptake; low titer or absent thyroid peroxidase antibody (TPO Ab) |
| Iodine-induced hyperthyroidism | Excess iodine | Low to undetectable thyroid radioactive iodine uptake |
| Drug-induced thyrotoxicosis | Lithium, interferon alpha; induction of thyroid autoimmunity (Graves disease) or inflammatory thyroiditis | Thyroid radioactive iodine uptake elevated in Graves disease or low to undetectable in thyroiditis |
| Amiodarone-induced thyrotoxicosis | Iodine-induced hyperthyroidism (type I) or inflammatory thyroiditis (type II) | Low to undetectable thyroid radioactive iodine uptake |
| Thyroid-stimulating hormone (TSH) secreting pituitary adenoma | Pituitary adenoma | Raised serum thyroid-stimulating hormone and alpha-subunit with raised peripheral serum thyroid hormones |
| Gestational thyrotoxicosis | Stimulation of thyroid gland thyroid-stimulating hormone receptors by human chorionic gonadotropin | Thyroid radioactive iodine uptake contraindicated in pregnancy; first trimester, often in setting of hyperemesis or multiple gestation |
| Molar pregnancy | Stimulation of thyroid gland thyroid-stimulating hormone receptors by human chorionic gonadotropin | Molar pregnancy |
| Struma ovarii | Ovarian teratoma with differentiation primarily into thyroid cells | Low to undetectable thyroid radioactive iodine uptake (raised uptake of radioactive iodine in pelvis) |
| Widely metastatic functional follicular thyroid carcinoma | Thyroid hormone production by large tumor masses | Differentiated thyroid carcinoma with bulky metastases; tumor radioactive iodine uptake visible on whole-body scan |
Adapted from Pearce EN: Diagnosis and management of thyrotoxicosis. Br Med J 332:1369-1373, 2006.
Diagnosis
Thyrotoxic patients have a decreased TSH (often undetectable) and increases in serum T4 and/or T3. Approximately 1% of patients will have isolated T3 thyrotoxicosis. A 24-hour radioactive iodine (RAI) thyroid uptake/scan or a pertechnetate scan will differentiate hyperthyroidism caused by increased hormone production from increased hormone release or exogenous intake. In the former, uptake will be elevated or normal, whereas in the latter, uptake will be low. RAI uptake may be falsely low in patients who have recently received iodinated contrast or amiodarone. Alternatively, pertechnetate scanning provides qualitative results in a few hours and an intense uptake of pertechnetate is indicative of increased thyroid hormone production. Either method must be interpreted with concurrent thyroid function tests.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree