PRECAUTIONS
In addition to vaccination, several types of precautions for use in patient care settings are critical to protecting the HCW. Making sense of them and knowing when to implement each can be confusing. The CDC has published guidelines outlining the recommended approach to infection control as well as the definition and application of the various precautions, including the specific personal protective equipment (PPE) to be used for each category. There are four types of precautions recommended in the CDC guidelines (3):
- Standard (previously termed universal)
- Contact
- Droplet
- Airborne
The CDC’s 2007 Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings is available online (3). Appendix A of that document lists recommended precautions for an extensive list of diseases and conditions including links to updated information on emerging pathogens such as Ebola viral disease (EVD).
Standard Precautions
Standard precautions are those that are applied when the practitioner comes into contact with patients, regardless of the diagnosis. Standard precautions include hand hygiene before and after contact with every patient and between anatomic sites on the same patient. Based on the clinical task and setting, additional precautions may be required. When providing care or performing procedures during which the HCW may come into contact with blood and body fluids (including secretions and excretions, but excluding sweat), broken skin, and mucous membranes, use of PPE is required. This may include gloves, masks, eye protection, face shield, and/or gowns.
Other considerations include special handling of patient care equipment, including sharps and other instruments and correct disposal and cleaning of linen and other contaminated items. It is important to note that, while not all of these additional precautions apply in every circumstance, the standard precaution of hand hygiene is truly universal and must be done before and after every patient contact. Even in clinical situations where there has been no visual contamination with blood, body fluids, secretions, excretions, or contact with contaminated items, compliance with hand hygiene must be stressed. Hands must be cleaned immediately after gloves are removed if they were worn. In addition, hand hygiene should be performed whenever indicated to avoid transfer of microorganisms to other patients and the environment, such as after contact with intravenous tubing or a monitor even when there has been no direct patient contact. An alcohol-based hand sanitizer with at least 60% alcohol should be used for most routine care. Alcohol-based hand sanitizers have been shown to be more convenient, faster, and more efficient than hand washing (4,5), and many institutions have these available throughout patient care areas. However, alcohol-based regimens are not recommended in all situations. Hand washing with soap and water is recommended in situations where visible soiling on hands occurred or in environments where spores may be present such as when caring for patients with Clostridium difficile.
In order to decrease pathogens that can be transmitted on HCW clothing, there are now HCW clothing restrictions in Britain including the elimination of ties, except bow ties, and white coats with long sleeves. While these HCW clothing restriction proposals are not currently being practiced in the United States, some ICUs have instituted policies that HCWs wear only surgical scrubs while providing patient care and that white coats be removed prior to entering ICU rooms. At this time, these recommendations are not part of the CDC’s standard precautions, but may become so in the future.
Gloves
Clean, nonsterile gloves are appropriate when contact with blood, body fluids, secretions, excretions, broken skin, mucous membranes, and items visibly contaminated by these fluids are expected. Gloves should be changed between tasks and procedures on the same patient if contact with material containing a high concentration of microorganisms occurs and between different anatomic sites on the same patient. Gloves should be removed immediately after use and hand hygiene performed before contact with the environment to avoid contamination of surfaces. Between contact with the next patient, hand hygiene must always be performed and new gloves donned if indicated; failure to do so is an infection control hazard (6). According to CDC guidelines, the use of gloves does not replace the need for hand hygiene. Gloves may have small, unapparent perforations or may be torn during use. Also, hands can become contaminated during removal of gloves (6) and clean gloves may be contaminated with dirty hands prior to donning making hand hygiene after glove removal critical.
Mask, Eye Protection, and Face Shield
The mucous membranes of the HCW, including those of the eyes, nose, and mouth, are at risk for exposure when performing procedures or patient care tasks that may generate aerosols or droplets. Bronchoscopy, open suctioning, and intubation are examples of aerosol-generating procedures (AGPs). Masks, goggles (eye protection), or face shields should be worn to protect the mucous membranes of the HCW.
Gowns
A clean, nonsterile gown that is fluid resistant or impermeable should be used during procedures and patient care activities when the clothing or skin of an HCW may be at risk for exposure or contamination. A gown should be worn if the patient is incontinent of urine or stool, has an ileostomy, colostomy, or wound drainage not covered, or contained by a dressing. Gowns should be removed immediately after patient contact and should not be worn outside of the patient’s room. Hands may be contaminated during gown removal and hand hygiene should be performed prior to contact with the environment or other patients.
Patient Care Equipment and Linen
If patient care equipment is soiled with blood, body fluids, secretions, and excretions, the HCW should don appropriate PPE to prevent skin and mucous membrane exposures, contamination of HCW clothing, and transfer of microorganisms to other patients and the environment. Multi-use equipment should be appropriately cleaned or processed prior to being used for the care of another patient. Single-use items should be disposed of in the appropriate manner. Adequate procedures for the routine cleaning and disinfection of environmental surfaces, beds, bedrails, bedside equipment, and other high-touch surfaces should be established. Soiled linen should be handled and transported in a manner that prevents skin and mucous membrane exposure and contamination of HCW clothing (7).
Contact Precautions
Contact precautions are designed to reduce the risk of transmission of epidemiologically important microorganisms and certain infestations caused by lice or scabies. Transmission of these organisms or conditions may take place either by direct or indirect contact. Direct contact transmission from patient to staff includes physical transfer of microorganisms to the HCW from an infected or colonized patient. This usually takes place via skin-to-skin contact during patient care activities. Indirect transmission occurs when an HCW comes into contact with a contaminated intermediate object in the patient’s environment, particularly high-touch surfaces such as monitors, medication pumps, bedrails, bedside tables, commodes, and sinks. Pathogenic organisms that can remain viable on the surfaces in the patient’s environment for extended periods of time include methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococcus (VRE), Gram-negative organisms such as Pseudomonas spp. and Acinetobacter spp., and C. difficile (6).
Contact precautions involve appropriate patient placement, hand hygiene, gloves, gowning, and precautions when transporting patients and when using patient care equipment. Patients should be placed in a private room if possible; however, the door to the room may be left open. When a private room is not available, the patient can be placed in a room with a patient who has colonization or active infection with the same microorganism but no other infection (cohorting). If a private room is not available and cohorting is not achievable, the epidemiology of the microorganism should be considered and the patient population taken into consideration when determining patient placement. An example of this would include the avoidance of placing an immunocompromised patient in the same room as a patient with a resistant organism.
Gloves and hand hygiene should be used as with standard precautions, and gowns should be used if there will be any contact with the patient, environmental surfaces, or items in a patient’s room. However, unanticipated contact with the patient and/or the environment may occur. Therefore, many hospitals require a gown and gloves be worn upon entry to the room even if substantial contact with the patient or environment is not anticipated. Patient transport should be limited only to essential purposes, and if the patient is transferred out of the room, care should be taken to ensure minimal risk of transmission to other patients and environmental surfaces or equipment. Patient care equipment such as stethoscopes, thermometers, and intravenous pumps should be dedicated to a single patient (or cohort) to avoid sharing between patients, minimizing transfer of organisms.
Droplet Precautions
Droplet precautions aim to reduce the risk of spreading infectious agents when droplets formed by secretions are expelled from the respiratory tract during sneezing, coughing, talking, and open suctioning. Transmission in this manner occurs when infectious droplets greater than 5 μm in size come into contact with the mucous membranes (conjunctiva, nose, mouth) of an HCW. Droplet transmission requires close contact between the infected patient and the HCW, as droplets do not remain suspended in the air and generally travel only short distances, usually less than 3 feet. However, data from experimental studies with smallpox and experience during the severe adult respiratory syndrome (SARS) outbreak showed that droplets containing these viruses could reach persons up to 6 feet away. Because of this, the CDC suggests that masks and face protection be donned 6 to 10 feet from a source patient requiring droplet precautions. According to CDC recommendations, because droplets do not remain suspended in the air, special air handling and ventilation are not required to prevent droplet transmission, and the door to the patient’s room may remain open (3).
Droplet precautions apply to any patient known or suspected to be infected with epidemiologically important pathogens that can be transmitted by infectious droplets, such as Neisseria meningitidis, influenza, Bordetella pertussis, rhinovirus, rubella, Haemophilus influenzae type B, adenovirus, Mycoplasma pneumoniae, and parvovirus B19. Patients should be placed in a private room or cohorted. Transport should be limited to essential purposes only. If transport becomes necessary, a surgical mask should be placed on the patient to minimize dispersal by droplets.
Hospitals should implement programs that encourage respiratory etiquette among patients, visitors, and HCWs. This includes covering sneezes and coughs with tissues which are then disposed of immediately, followed by hand hygiene. If tissues are not available sneezes or coughs should be covered by the upper arm or elbow and not directed into the hands. Visitors and patients may be asked to wear masks if they are experiencing upper respiratory symptoms (7). To prevent transmission to both patients and other HCWs, many hospitals require that HCWs with respiratory illnesses be medically evaluated prior to patient contact.
Airborne Precautions
Airborne precautions are designed to reduce the risk of transmission of respiratory infectious agents which are carried in airborne droplets less than or equal to 5 μm in size (droplet nuclei) or can attach to dust particles in the environment. Airborne transmission occurs by dissemination and inhalation of these droplet nuclei and dust particles which can be dispersed widely by air currents or may travel through ventilation systems. They may be inhaled by a susceptible host within the same room or by a patient several rooms or floors away from the source patient because a single ventilation system often serves multiple patient rooms. Therefore, special air handling and ventilation are required to prevent airborne transmission. Airborne precautions apply to patients known or suspected to be infected with epidemiologically important pathogens such as Mycobacterium tuberculosis (MTb), rubeola virus (measles), and varicella-zoster virus (VZV, chickenpox) (8). The patient should be placed in an airborne infection isolation (AII) room that has monitored negative air pressure in relation to the surrounding areas, 6 to 12 air exchanges per hour, and appropriate discharge of air outdoors or monitored high-efficiency filtration of room air before the air is recirculated to other areas in the hospital. The room door should be kept closed and the patient should remain in the room except for medically necessary tests. If a private AII room is not available, the patient should be cohorted with a patient infected with the same microorganism but with no other infection. Consultation with infection prevention and control professionals is advised before patient placement in the event no AII rooms are available (3).
An N95 or higher respirator must be worn to provide an adequate barrier to various airborne organisms including M. tuberculosis (9,10). Powered air-purifying respirators (PAPRs) may be used in these settings as well. PAPRs do not require fit testing and may be worn by HCWs with facial hair. Per CDC, HCWs with documented immunity to measles or chickenpox need not wear respiratory protection. However, hospital policies may dictate all HCWs wear respiratory protection regardless of immune status. Susceptible persons should not enter the room of patients known or suspected to have rubeola or VZV if immune caregivers are available. If susceptible persons must enter the room of a patient known or suspected to be infected with rubeola or VZV, they should wear an N95 respirator or PAPR.
Patient transport should be limited to the movement and transport of the patient from the room for essential purposes only. If transport or movement is necessary, minimize patient dispersal of droplet nuclei by placing a surgical mask on the patient. The patient is not required to wear an N95 respirator or PAPR during transport (3).
BLOOD-BORNE PATHOGENS
Handling of sharps (needles, scalpels, sutures and other sharp instruments or devices) during use, cleaning, or disposal should be done with extreme care to avoid percutaneous injury. Blood-borne pathogen training is mandatory for HCWs who have exposure to blood and body fluids per the Occupational Safety and Health Administration (OSHA) Blood-borne Pathogen Standard (11). Safe practices include never recapping any needle, avoiding manipulation of sharps using both hands, avoiding hand-off of a sharp from one HCW to another or using techniques that involve the point of a needle being directed toward any part of an HCW’s body. Used needles should not be removed from disposable syringes by hand. Bending, breaking, or otherwise manipulating used needles should not be done. Used disposable syringes and needles, scalpel blades, and other sharp items should be placed in an appropriate puncture-resistant container which should be located as close as practical to the area in which the items are being used. Reusable sharps can be placed in a puncture-resistant container for transport to a reprocessing area. The Needlestick Safety and Prevention Act of 2000 directs employers to evaluate their medical devices on an ongoing basis and make efforts to convert as many devices as possible to safer products (11).
Human Immunodeficiency Virus
Human immunodeficiency virus (HIV) is the etiologic agent of acquired immunodeficiency syndrome (AIDS). Two species of HIV, HIV-1 and HIV-2, infect humans. HIV is transmitted primarily by exposure to blood and other body fluids from an HIV-infected patient. The three primary methods of transmission are via unprotected sexual intercourse, vertical transmission (mother to child), and contaminated needles (either occupational exposure or with the use of intravenous drugs of abuse). In the United States, blood products are screened for HIV, and transfusion-associated transmission essentially has been eliminated. Potentially infectious fluids include blood and blood-containing fluids; fluids from other sites such as semen, vaginal secretions, and cerebrospinal fluid (CSF); and, synovial, pleural, peritoneal, pericardial, and amniotic fluids.
After percutaneous or mucosal exposure, HIV replicates within dendritic cells and spreads via lymphatics to the bloodstream where CD4+ cells become infected. The delay in systemic spread leaves a “window of opportunity” for postexposure prophylaxis (PEP) using antiretroviral drugs. The most common HCW exposure is via percutaneous injury. Other types of exposures among HCWs include mucous membrane exposure, nonintact skin exposure, and bites. The average risk of HIV transmission to HCWs after a percutaneous injury is approximately 0.3% (12,13) and 0.09% after a mucous membrane exposure (14). Transmission of HIV via nonintact skin exposure has been documented (15); however, the risk for transmission via this route is much less than for mucous membrane exposures (16). The risk for transmission after exposure to fluids or tissues other than HIV-infected blood has not been quantified but is believed to be lower than for blood exposures.
Several factors may affect the risk of HIV transmission after an occupational exposure. Increased risk is associated with a larger quantity of blood from the source patient such as contained in a hollow bore needle, percutaneous injury that occurs during a procedure involving a needle placed directly into a vein or artery of the source patient, a deep tissue injury in the HCW and blood exposure from a patient with a high HIV viral load.
HIV Postexposure Management
Occupational exposures to HIV require urgent medical evaluation. The goal of postexposure management is to deliver HIV PEP to the HCW with a high-risk exposure within 2 hours. A number of occupational HIV PEP guidelines have been published (17,18). The initial step following an occupational exposure to blood and body fluids is prompt treatment of the exposure site including washing wounds and skin sites with soap and water and flushing mucous membranes with water. Squeezing the injured area to expel blood should not be done. The use of local antiseptics at the injury site is not contraindicated, although there is no evidence of efficacy.
The HCW should immediately report the exposure to facilitate rapid testing of the source patient for HIV, hepatitis B (HBV), and hepatitis C (HCV). PEP should be initiated for the HCW if indicated. The source patient should be tested for HIV immediately. FDA-approved rapid tests can produce HIV test results within 30 minutes, with sensitivities and specificities similar to those of first- and second-generation enzyme immunoassays (EIAs). Third-generation chemiluminescent immunoassays can detect HIV-specific antibodies 2 weeks sooner than conventional EIAs and generate test results in an hour or less. Fourth-generation combination p24 antigen—HIV antibody (Ag/Ab) tests produce both rapid and accurate results, and p24 antigen detection allows identification of most infections during the window period (the time period between HIV infection and the development of detectable HIV antibodies). Rapid determination of source patient HIV status provides essential information about the need to initiate and/or continue PEP. As per the U.S. Public Health Service Guidelines, regardless of which type of HIV testing is employed, all of the above tests are acceptable for determination of source patient HIV status (17). It is not necessary to investigate whether the source patient is in the window period unless acute antiretroviral syndrome is suspected (17). HIV RNA polymerase chain reaction (PCR) testing for routine screening of source patients is not recommended. As per the CDC, if the source patient is HIV negative, no further testing of the HCW is indicated. However, other guidelines recommend that if the source patient has been at risk for HIV exposure in the preceding 6 weeks, then an HIV RNA PCR should be performed on the source patient. Depending on the results, PEP is either continued or stopped (Figure 10.1) (18).
The severity of exposure is no longer used to determine the number of drugs to be offered in an HIV PEP regimen. A regimen of three or more antiretroviral drugs is now recommended for all occupational exposures to HIV. The drug regimen selected should have a favorable side-effect profile as well as a convenient dosing schedule to facilitate both adherence to the regimen and completion of the recommended 4-week course of PEP. As of 2015, the preferred HIV PEP regimen includes emtricitabine plus tenofovir with either raltegravir or dolutegravir. This once-a-day regimen is tolerable, potent, and conveniently administered, and it has been associated with minimal drug interactions (17). Expert consultation for HIV PEP is recommended for an exposure report delayed more than 72 hours, unknown source (needle in sharps disposal container or laundry), known or suspected pregnancy in the HCW, breastfeeding in the HCW, known or suspected resistance of the source HIV to antiretroviral agents, toxicity of the initial PEP regimen, or significant underlying illness in the HCW. HCWs should have follow-up within 72 hours of the occupational exposure regardless of whether they take PEP or not. If local expert consultation is not available, the National Clinicians’ Post-Exposure Hotline (PEPline) can be consulted at 888-448-4911.
Hepatitis Viruses
Several hepatitis viruses have been described, including hepatitis A, B, C, D, E, and G. Hepatitis A and E are transmitted by the fecal/oral route, usually by contaminated food. They cause acute hepatitis that is generally self-limited and confers immunity to future infections. In a small percentage of cases, hepatitis E can develop into an acute severe liver disease that is often fatal, especially in pregnant women. Hepatitis D is caused by delta virus and can only replicate in the presence of HBV. By far, HBV and HCV pose the greatest threat to HCWs.
Hepatitis B
HBV is endemic in many parts of the world, causes both acute and chronic hepatitis, and is a major cause of hepatocellular carcinoma. The virus is transmitted through exposure to blood and body fluids. Routes of transmission include unprotected sexual contact, blood transfusions, use of contaminated needles and syringes, vertical transmission, and occupational exposure including needlesticks and mucous membrane exposure. As with many blood-borne viral pathogens, the risk of transmission from a blood and body fluid exposure is closely related to the volume of blood and the number of copies of virus present in the blood of the source patient. The percutaneous injury site should be washed with soap and water, and the mucous membrane flushed with water. Squeezing the injured area to expel blood should not be done.
Per the CDC guidelines, risk of transmission of HBV is also related to the HBV envelope antigen (HBeAg) status of the source patient. In patients who were both hepatitis B surface antigen (HBsAg) and HBeAg positive, the risk of developing clinical hepatitis from a needle injury was 22% to 31%. The risk of developing serologic evidence of infection was 36% to 62%. If the source patient was HBsAg positive with a negative HBeAg, the risk of developing clinical hepatitis from a needle injury was 1% to 6%, and the risk of developing serologic evidence of HBV infection was 23% to 37% (19). Blood exposure and percutaneous injuries are among the most efficient modes of transmitting HBV since blood has the highest titers of HBV compared to other body fluids. HBsAg is also found in several other body fluids, including breast milk, CSF, stool, nasopharyngeal washings, saliva, semen, sweat, and synovial fluid (20). When investigations of outbreaks were performed, most infected HCWs could not recall a percutaneous injury but rather recalled caring for a patient who was HBsAg positive (21–24). HBV has been shown to survive in dried blood at room temperature for at least 1 week (25), and it is possible that contact with environmental surfaces is a potential risk for HBV transmission as has been shown in hemodialysis units (23,26–28).

FIGURE 10.1 Health care worker HIV postexposure prophylaxis. The strategy for source patient evaluation and PEP recommended by the CDC is shown by the blue pathway. The orange pathway details additional steps recommended by other guidelines (18) when the source patient’s rapid HIV test is negative. For both pathways, the choice of antiretrovirals for PEP (gray box) should be verified at www.aidsinfo.nih.gov, since the regimens change as new antiretrovirals become available.
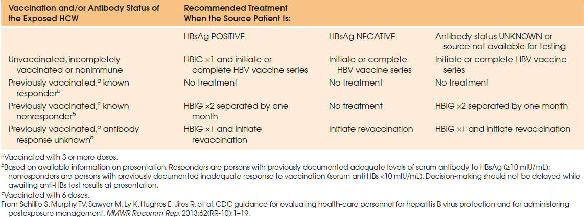
The key factor in preventing HBV infection in the health care setting is vaccination. The OSHA Blood-borne Pathogen Standard requires employers to offer the HBV vaccine series to all employees who have exposure to blood-borne pathogens (11). The worker may opt out of the series but they can change their mind at any time and be given the vaccination series. HBV vaccination is part of the routine immunization schedule for infants and children in the United States; by the age of 18 months, children who are up to date on their vaccinations have been fully immunized for HBV. As with childhood vaccination, the protocol for adult immunization consists of three doses of the vaccine given intramuscularly in the deltoid muscle. For those whose HBV vaccination series is interrupted, there is no need to restart the series over again. Vaccination can resume based on where in the series the patient was at the time of the interruption. Follow-up testing to document seroconversion is recommended, and repeating the vaccination series for nonresponders is indicated.
| TABLE 10.2 Health Care Worker Postexposure Prophylaxis for Hepatitis B | |||
 | |||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








