CHAPTER 68
Permanent Implant
INTRODUCTION
The use of intrathecal opioids dates back to August 16, 1898, when August Bier and his assistant Hildebrandt performed “cocainization of the spinal cord” on each other. Unfortunately, Bier was also the first to describe the complication of postdural puncture headache from his personal experience. The mechanism of opioids on the spinal cord was later confirmed in a rat model.1 Subsequently, intrathecal medication has been widely utilized for both anesthesia and analgesia. The use of implantable intrathecal drug delivery systems began in the early 1980s and is now indicated for use in patients with persistent chronic pain of malignant and nonmalignant origin that are either refractory to maximal medical therapy or dose limited due to significant side effects, and has been demonstrated to have a better side effect profile than systemic opiates alone.2 In addition, non-narcotic medications that can have minimal analgesia when administered systemically can be very effective when administered intrathecally.3
INDICATIONS
• Chronic intractable pain
• Malignant and nonmalignant in origin
• Refractory pain to maximal systemic medical therapy
• Dose limiting side effect to systemic medical therapy
• Intractable spasticity
AVAILABLE MEDICATIONS
• Medications may be compounded to suit individual needs
• Medications may be used individually or compounded in combination therapy
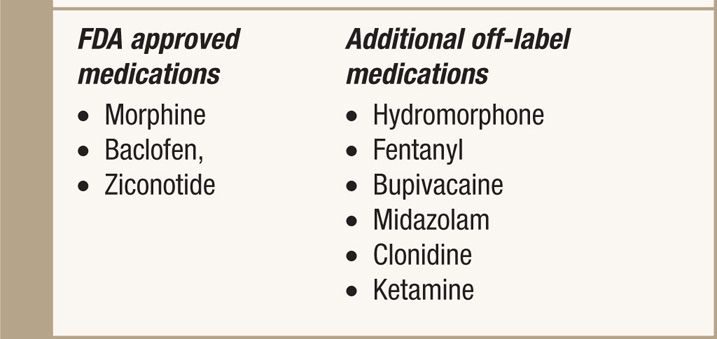
• FDA approved and off-label medications (Table 68-1)
TABLE 68-1. List of Medications Used in Intrathecal Pumps. Currently the Only Three FDA Approved Medications are: Morphine, Baclofen, and Ziconotide. These Medications Can be Delivered Individually or Compounded for Combination Therapy (Deer et al, 2012)
RELEVANT ANATOMY
The preferred catheter insertion site is below the conus medullaris, usually located at vertebral levels L1-L2 (Figure 68-1A). The catheter is anchored to the lumbodorsal fascia to prevent migration. The pump is usually placed in the left or right lower quadrant of the abdomen in the subcutaneous fat between the inferior costal margin and iliac crest. One may consider placing the pump in the subfacial space between the external and internal abdominal oblique muscles in the young and/or thin patients for decreased risk of wound breakdown and improved cosmesis (Figure 68-1B).
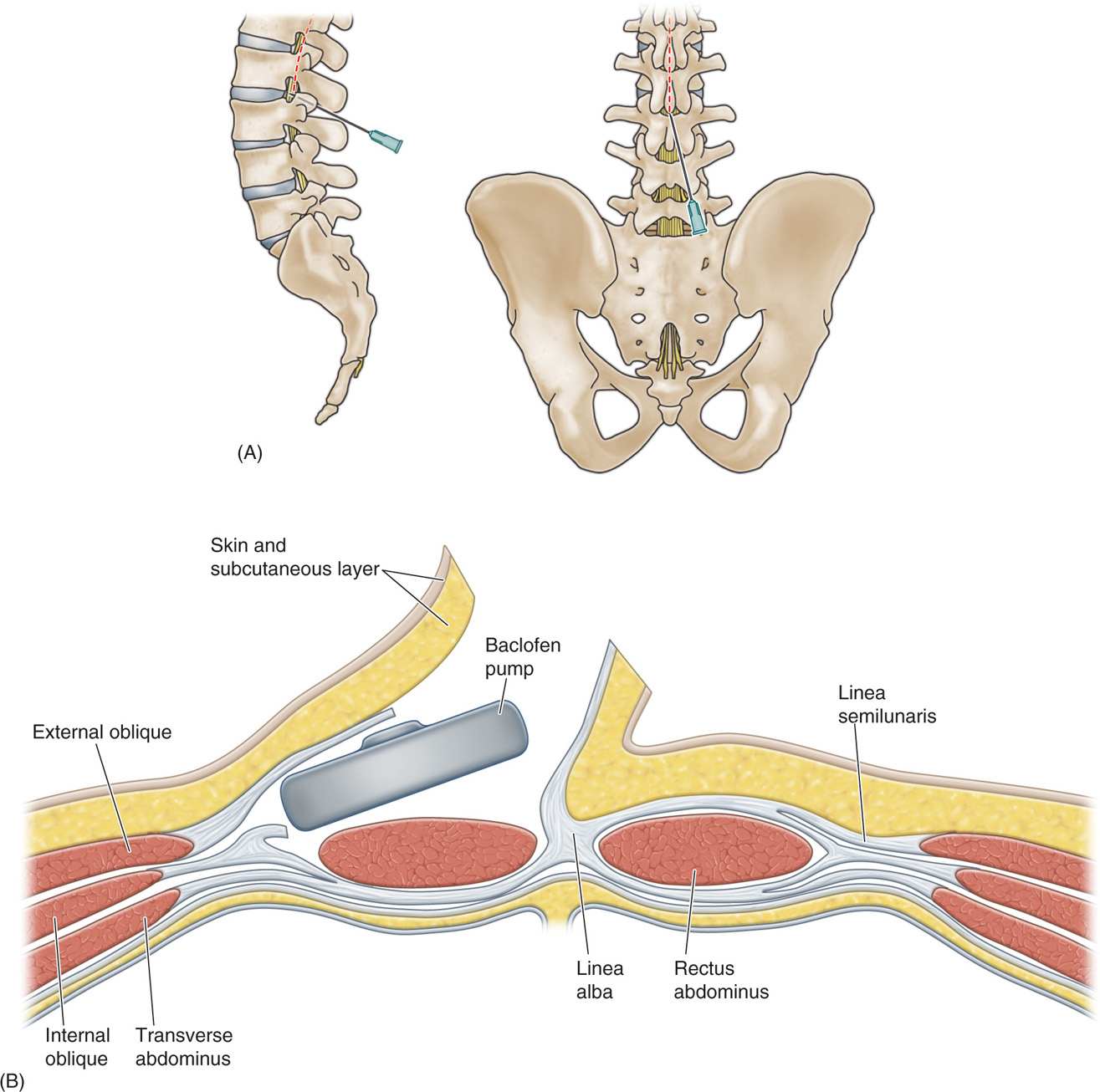
Figure 68-1. (A) Anatomy for needle insertion. (B) Diagram illustrating the location of IT pump.
Basic Prerequisites
• Patient has tried and failed maximal systemic pain control
![]() Uncontrolled pain on maximal systemic medications
Uncontrolled pain on maximal systemic medications
![]() Uncontrolled side effects with systemic medications
Uncontrolled side effects with systemic medications
• Successful intrathecal trial
Methods of Trial for Intrathecal Pump
• In-patient continuous intrathecal catheter infusion.
• In-patient continuous epidural catheter infusion.
• Single or repeated epidural injections
• Single or repeated intrathecal injections
Basic Concerns and Contraindications
• Psychological evaluation to rule out secondary gain issues
• Comorbidities
• Anticoagulation therapy
• Immunocompromised condition and high risks for infection
PREOPERATIVE CONSIDERATIONS
Patients being considered for intrathecal drug delivery pump therapy should have a multidisciplinary care team which ideally includes a primary care physician, a pain psychologist, and a pain management specialist. Those who fail maximal systemic therapy or have dose-limiting side effects should undergo intrathecal trial via a single bolus lumbar puncture or an external drug delivery system through a temporary intrathecal catheter. Concurrently, noncancer or nonterminally ill patients should have a thorough pain psychology evaluation to rule out any potential secondary gain issues and/or untreated psychological comorbidities. Prior to placement of permanent intrathecal drug delivery pump, patients should undergo the necessary preoperative anesthesia evaluation. For patients who are taking anticoagulation medications, follow the latest American Society of Regional Anesthesia and Pain Medicine (ASRA) guidelines for neuraxial techniques. Finally, patients should have a clear understanding of potential complications and intrathecal drug side effects.
Pump pocket site should be marked with consideration for patient comfort, activities, belt lines, rib margins, wheelchair arms, and prostheses.
Editor’s Pearls
Prior to coming to the operating room, I like to place a nonsterile dummy pump on the abdomen to assess the patient’s tolerance for the device relative to their belt line, rib margin, and iliac crest. It is marked preoperatively.
Optimal pump position (Figure 68-2)
Figure 68-2. Optimal pump position.
• Superior to iliac crest.
• Below costal margin.
• Away from belt line.
• Avoid areas of previous incisions and/or radiation.
• Catheter access port should be oriented uniformly to facilitate pump refill.
• Obese patients should have pump pocket position marked in the standing position to account for movement of large panniculus.
Patient Position
• Patient is placed in the lateral decubitus position allowing for simultaneous access to both the patient’s lumbosacral spine and abdomen.
• The patient’s back should be perpendicular and their spine should be parallel to the floor.
• The fluoroscopy compatible surgical table is slightly flexed at the hip to further open up the space between the patient’s rib cage and pelvic brim giving greater exposure to the abdomen for pump pocket placement.
• The patient’s arms are placed slightly above their head to allow for C-arm access and pillows are placed between the arms to alleviate pressure points.
• The legs are slightly flexed and padded to avoid pressure points.
• The entire abdomen and lumbosacral spine is prepped with antiseptic solution and draped with a large Ioban dressing (3M, Minneapolis, MN) for added sterility (Figure 68-3).
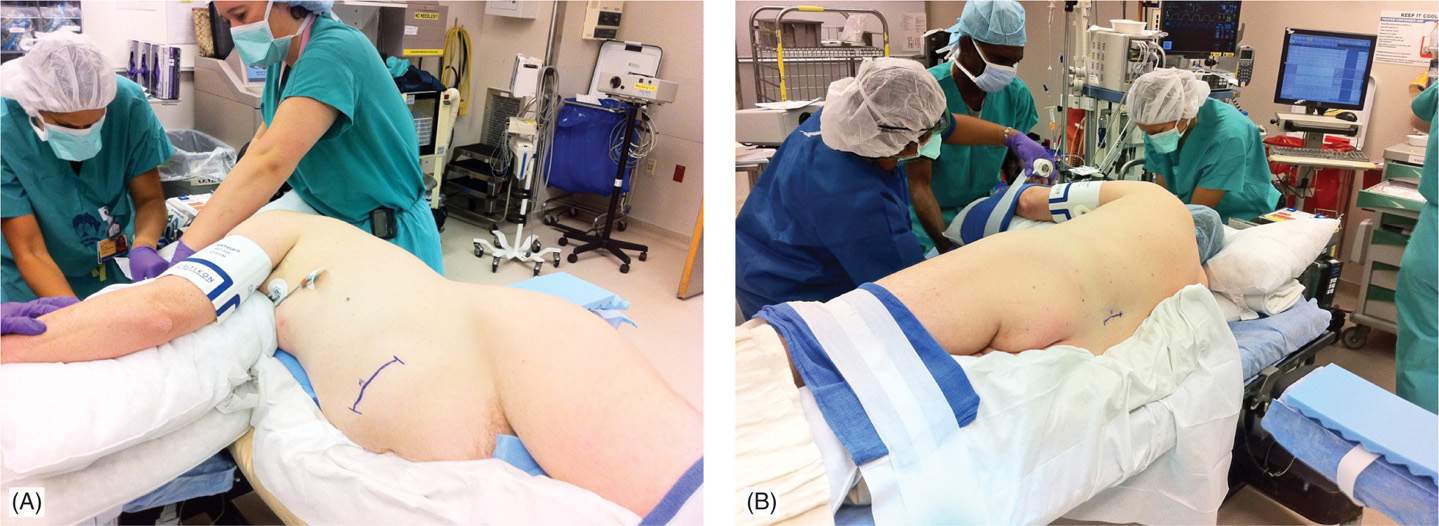
Figure 68-3. Proper positioning of the patient allows for access to both intrathecal catheter placement in the lumbosacral spine and pump placement in the anterior abdomen. (Used with permission from Dr. M. Bottros and Dr. K. Williams, Blaustein Interventional Pain Treatment Center, Johns Hopkings Univeristy, Baltimore, MD.)
Fluoroscopic Views
Fluoroscopic guidance should be utilized for all intrathecal catheter placements to ensure patient safety and to verify the anatomic location of the catheter. A C-arm fluoroscope is sterilely draped and positioned in the cross-table anteroposterior (AP) configuration during the case. This allows for C-arm access for both AP and lateral views during the procedure. The position of the image intensifier can be positioned either at the patient’s abdomen or back. Placement of the larger image intensifier at the abdomen allows for added room behind the patient for placement of the intrathecal catheter, however, this configuration may create increased scatter radiation for the surgeon and operating room staff.
Equipment
There are now multiple commercially available intrathecal drug delivery pumps include programmable (SynchroMed II, Medtronic, Minneapolis, MN, and Prometra, Flowonix, Mt. Olive, NJ) and constant flow (Codman 3000, Johnson and Johnson, Raynham, MA). The programmable SynchroMed II or Prometra pump offers the versatility of dose adjustments via telemetric flow control without having to replace the medication within the reservoir but its usage is limited by the battery lifespan (5-10 years). Constant flow configuration of the multiple nonprogrammable pumps offers unlimited usage due to its contained propellant design but dose adjustments require replacement of remaining drug with a refill of a different concentration. The manufacturers offer models with various reservoir volumes ranging from 16 to 40 mL. Choice of equipment should be based on the individual medication requirements of the patient and personal patient preference (Table 68-2).
TABLE 68-2. Comparison of The Two Commercially Available Intrathecal Delivery Pumps

Anesthesia
The operation can be successfully performed under general anesthesia, regional anesthesia or local anesthesia with sedation. Anesthesia with sedation allows for direct patient feedback during catheter placement, which may help alert the surgeon of potential nerve or spinal cord injury; however, in certain cases, patients cannot tolerate lumbar and abdominal incisions without a deeper level of sedation.
Implantation Technique
The pump implantation procedure is described in 6 simplified steps as follows:
• Accessing the intrathecal space
• Anchoring the catheter
• Creating the pump pocket
• Tunneling the intrathecal catheter
• Pump placement and catheter connection
• Wound closure and dressing
Once the patient is anesthetized or adequately sedated, a single prophylactic dose of parenteral antibiotic is given within 60 minutes of incision.
Accessing the Intrathecal Space
• Under direct fluoroscopic guidance, the patient’s L3-L4 intervertebral space is located with fluorography in the AP view.
• The overlying skin is infiltrated with a local anesthetic (we prefer 1% lidocaine with 1:200,000 epinephrine) with a 25-gauge needle.
• May elect to access the intrathecal space percutaneously with the 14-gauge Tuohy needle first then create the necessary skin flaps by making a vertical incision that incorporates the needle site.
• We prefer to make the vertical paramedian skin incision prior to accessing the intrathecal space. A 3- to 4-cm vertical paramedian incision is made over the L3-L4 intervertebral space.
• Once the skin is incised sharply with a skin blade, the dissection is carried down to the lumbodorsal fascia with electrocautery and blunt dissection.
• Dissection is carried out laterally 1 to 2 cm to the lumbodorsal fascial plane to create small tissue flaps and have adequate space for the anchor and excess catheter.
• Meticulous hemostasis should be obtained at this point.
• The skin is retracted with a Weitlaner self-retaining retractor.
• Next, the intrathecal space is accessed by shallow paramedian approach with a 14-gauge Tuohy needle.
• The shallow angle of the paramedian approach reduces the potential for shearing or excess stress on the catheter, minimizes catheter kinking, and facilitates advancing the catheter to the desired level.
• Subarachnoid position of the needle is confirmed radiographically and clinically with positive return of cerebral spinal fluid (CSF).
• The Tuohy needle bevel is then turned cephalad, the stylet is removed and the catheter is quickly inserted into the needle to minimize CSF leakage.
• Under live fluoroscopic guidance in the AP view, the catheter is advanced to the desired vertebral level.
Editor’s Pearls
Minimize nerve root trauma during intrathecal needle insertion by avoiding muscle paralysis. This should be discussed with the anesthesiologist prior to induction of general anesthesia.
Anchoring of Catheter
Once the catheter is placed, the intrathecal pump should be prepared on the back table by filling it with the desired medication and allowing adequate time for the pump to prime.
• Before the Tuohy needle is removed, a purse string suture is made within the lumbodorsal fascia and loosely placed around the Tuohy catheter complex. We prefer a nonabsorbable braided suture such as an O-TiCRON (Covidien, Mansfield, MA) or O-Ethibond (Ethicon, Piscataway, NJ).
• Before the guidewire and Tuohy needle are completely withdrawn, the purse string suture is tied down snug. This may decrease CSF leak around the catheter.
• Next the Tuohy needle and catheter guidewire are carefully removed while keeping the catheter in place.
• Proximal catheter position is checked with fluoroscopy and a small clamp is placed at the distal tip to prevent further CSF leakage.
• The catheter is then anchored to the lumbodorsal fascia using premade anchoring devices that are supplied by the pump manufacturer.
• The anchor is first carefully secured to the catheter with the same nonabsorbable braided O suture and then catheter-anchor complex is sutured to the lumbodorsal fascia.
Creating the Pump Pocket
• The pump pocket is made by first infiltrating the skin over the planned pocket site with local anesthetic.
• Using a skin blade, an 8-cm horizontal incision is made at the superior aspect of the pocket.
• Care should be taken to avoid a position too close to the rib margin. Typically 3 to 4 cm below the rib margin is sufficient.
• This incision is dissected to a depth of approximately 1.5 cm into the subcutaneous tissue with both electrocautery and blunt dissection.
• The pump pocket should be no more than 2.5 cm deep allowing for future programming and medication refill.
• Keeping parallel to the skin, a pocket is created with sharp and blunt dissection. One may use the pump as a template of pocket size.
• Meticulous hemostasis should be maintained.
Tunneling the Intrathecal Catheter
• Once the pump pocket is created, the intrathecal catheter is tunneled laterally from the insertion site to the abdominal wall, where the pump will reside.
• A malleable tunneling device is carefully tacked in the subcutaneous plane from the abdominal incision toward the back incision.
• It is imperative to keep the tunneling trocar superficial in the subcutaneous tissue or risk major vascular and/or bowel injury.
• Avoid placing tunneling device in subcutaneous plane to minimize the risks of penetration through skin.
• The catheter is fed through the tunneling device and trimmed to the appropriate length.
• A gentle strain relief loop is created at the catheter insertion site.
• Catheter position should be verified radiographically and clinically with positive CSF return.
Pump Placement and Catheter Connection
• Final catheter length should be recorded as part of the standard pump parameters.
• After the catheter is properly connected to the pump, excess catheter is coiled deep to the pump.
• One may elect to use a Dacron pouch around the pump to encourage fibrosis and prevent pump migration or flipping.
• We prefer to secure the pump directly to the external abdominal fascia with interrupted nonabsorbable braided suture through prefabricated anchors found on the pump.
Wound Closure and Dressing
• Prior to wound closure, both incisions are irrigated with warm antibiotic impregnated saline solution.
• The incisions are closed in a layered fashion.
• Use absorbable 1-O Vicryl for inverted interrupted sutures at the deep fascial layer.
• Followed by 3-O Vicryl at the subcutaneous layer.
• The skin is closed with a running subcuticular 4-O monocryl suture.
• The closed incision is reinforced with tissue glue and left open to air.
• If programmable pump is used, the appropriate settings are verified through telemetry.
• Patient is woken up, and taken to the recovery room for postoperative monitoring and safe discharge to floor or home.
POSTPROCEDURAL FOLLOW-UP
Most pump implantation patients are discharged home on the same day but one may elect to admit patients with complex comorbidities overnight for observation. Close monitoring should be provided for incisional bleeding and/or infection. All post-implant patients should be followed up with wound checks and possible suture/staple removal within 2 weeks of surgery. Patient will require several follow-up appointments for titration of intrathecal medication dosage.
POTENTIAL COMPLICATIONS
• Acute postoperative complications
![]() CSF leak
CSF leak
![]() Postdural puncture headache
Postdural puncture headache
![]() Wound infection
Wound infection
• Catheter-related complications
![]() Kinking
Kinking
![]() Breakage
Breakage
![]() Granuloma
Granuloma
• Medication-related complications
![]() Respiratory depression
Respiratory depression
![]() Sedation
Sedation
![]() Pruritus
Pruritus
![]() Constipation
Constipation
![]() Urinary retention
Urinary retention
![]() Nausea/vomiting
Nausea/vomiting
![]() Sweating
Sweating
![]() Hypogonadism
Hypogonadism
![]() Decreased libido
Decreased libido
• Long-term complications
![]() Pump failure
Pump failure
![]() Displaced catheter
Displaced catheter
![]() Cracked or kinked catheter
Cracked or kinked catheter
![]() Human error in programming
Human error in programming
PITFALLS AND CLINICAL PEARLS
• Accessing the intrathecal space below, the level of the conus medullaris will minimize spinal cord injury.
• Keeping the needle bevel parallel to the spine at dural entry may help minimize future CSF leak.
• Placement of catheter tip near the spinal level of pain complaints will keep looking for proof in literature.
• Prelubricating the guidewire with saline may help during removal.
• Keeping the catheter and guidewire straight during removal will prevent damage to the catheter.
• Leaving the Tuohy in place while making the purse string stitch prevents inadvertent catheter damage from the suturing needle.
• Keeping the guidewire in distal catheter while tightening the purse string suture.
• Avoid withdrawing the catheter through the Tuohy needle to avoid shearing of the catheter.
• Keep pocket parallel to skin to avoid slanted pump position or pump flipping.
• Avoid making the incision over the final pump position.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






