Obstetrical Anesthesia
Ferne R. Braveman
Barbara M. Scavone
Marcelle E. Blessing
Cynthia A. Wong
Key Points
Related Matter
Lung Volumes
Pudendal Nerve Block
C-Section Spinal
Fetal Heart Rate
Surgery Recommendations in Pregnancy
Physiologic Changes of Pregnancy
During pregnancy, there are major alterations in nearly every maternal organ system. These changes are initiated by hormones secreted by the corpus luteum and placenta. The mechanical effects of the enlarging uterus and compression of surrounding structures play an increasing role in the second and third trimesters. This altered physiologic state has relevant implications for the anesthesiologist caring for the pregnant patient. The most relevant changes involving hematologic, cardiovascular, ventilatory, metabolic, and gastrointestinal functions are considered in Table 40-1.
Table 40-1. Summary of Physiologic Changes of Pregnancy at Term | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Hematologic Alterations
Increased mineralocorticoid activity during pregnancy produces sodium retention and increased body water content. Thus, plasma volume and total blood volume begin to increase in early gestation, resulting in a final increase of 40% to 50% and 25% to 40%, respectively, at term. The relatively smaller increase in red blood cell volume (20%) accounts for a reduction in hemoglobin concentration (from 12 to 11 g/dL) and hematocrit (to 35%).1 Plasma expansion and the resultant relative anemia of pregnancy plateau at approximately 32 to 34 weeks’ gestation.2 The leukocyte count ranges from 8,000 to 10,000/mm3 throughout pregnancy. Several procoagulant factor levels increase during pregnancy, most notably fibrinogen, which doubles in mass. Anticoagulant activity decreases, as evidenced by decreased protein S concentrations and activated protein C resistance, and fibrinolysis is impaired. Increases in D-dimer and thrombin–antithrombin complexes indicate increased clotting and probable secondary fibrinolysis. Indeed, pregnancy has been referred to as a state of chronic compensated disseminated intravascular coagulation.3,4 These coagulation changes peak at the time of parturition.5 The platelet count is decreased in pregnant women, due to both dilution and increased consumption, and 6% to 15% of pregnant women at term have a platelet count <150 × 109/L, compared with only 1% of age-matched nonpregnant controls. A further 1% of women at term have platelet counts <100 × 109/L.6
Serum cholinesterase activity declines to a level of 20% below normal by term and reaches a nadir in the puerperium. However, it is doubtful that moderate succinylcholine doses lead to prolonged apnea in otherwise normal circumstances.7 Although the total amount of protein in the circulation increases, plasma protein concentration declines to <6 g/dL at term because of dilution from increased plasma volume.8 The albumin–globulin ratio declines because of the relatively greater reduction in albumin concentration. A decrease in serum protein concentration may be clinically significant because the free fractions of protein-bound drugs can be expected to increase.
Cardiovascular Changes
Arterial blood pressure decreases slightly because the decrease in peripheral resistance exceeds the increase in cardiac output. Additional increases in cardiac output occur during labor (when cardiac output may reach 12 to 14 L/min) and also in the immediate postpartum period because of added blood volume from the contracted uterus. These changes are exaggerated in multiple gestation pregnancies.11
Supine hypotensive syndrome, which occurs in 10% of pregnant women, occurs because the supine position leads to vena cava occlusion and thus decreased preload to the heart, resulting in lowered cardiac output and blood pressure, tachycardia, maternal mental status changes, nausea, and presyncope. Compression of the lower aorta in this position may further decrease uteroplacental perfusion and result in fetal asphyxia. From the second trimester, aortocaval compression by the enlarged uterus becomes progressively more important, reaching its maximum at 36 to 38 weeks’ gestation, after which it may decrease as the fetal head descends into the pelvis.12 Studies of cardiac output, measured with the patient in the supine position during the last weeks of pregnancy, have indicated a decrease to nonpregnant levels; however, this decrease was not observed when patients were in the lateral decubitus position.12 Therefore, left uterine displacement by placing a wedge under the right hip or providing 15 degrees left lateral pelvic tilt should be applied routinely during the second and third trimesters of pregnancy; a minority of women may remain susceptible to vena cava and aortic compression even at this degree of tilt.13
Changes in the ECG may also occur. In addition to heart rate increases, left axis deviation is observed in the third trimester, possibly due to upward displacement of the heart by the gravid uterus. There is also a tendency toward premature atrial contractions, paroxysmal supraventricular tachycardia, and ventricular dysrhythmias.14,15
Respiratory Changes
Airway resistance usually remains unchanged due to the competing effects of progesterone-induced relaxation of bronchiolar smooth muscle versus factors associated with increased airway resistance such as upper airway edema. Progesterone induces increases in minute ventilation, which increases from the beginning of pregnancy to a maximum of 50% above nonpregnant values at term. This is accomplished by a 30% to 50% increase in tidal volume and a small increase in respiratory rate. Alveolar dead space increases such that the dead space to tidal volume ratio remains unchanged. After delivery, as blood progesterone levels decline, ventilation returns to normal within 1 to 3 weeks.16
Metabolism
Basal oxygen consumption increases during early pregnancy, with an overall increase of 20% by term; CO2 production increases. However, increased alveolar ventilation leads to a reduction in the partial pressure of carbon dioxide in arterial blood (PaCO2) to 32 mm Hg and an increase in the partial
pressure of oxygen in arterial blood (PaO2) to 106 mm Hg. The plasma buffer base decreases from 47 to 42 mEq/L; therefore, the pH remains practically unchanged. The maternal uptake and elimination of inhalational anesthetics are enhanced because of the increased alveolar ventilation and decreased FRC. Also, the decreased FRC and increased metabolic rate predispose the mother to development of hypoxemia during periods of apnea/hypoventilation, such as may occur during airway obstruction or prolonged attempts at tracheal intubation.19
pressure of oxygen in arterial blood (PaO2) to 106 mm Hg. The plasma buffer base decreases from 47 to 42 mEq/L; therefore, the pH remains practically unchanged. The maternal uptake and elimination of inhalational anesthetics are enhanced because of the increased alveolar ventilation and decreased FRC. Also, the decreased FRC and increased metabolic rate predispose the mother to development of hypoxemia during periods of apnea/hypoventilation, such as may occur during airway obstruction or prolonged attempts at tracheal intubation.19
Human placental lactogen and cortisol increase the tendency toward hyperglycemia and ketosis, which may exacerbate pre-existing diabetes mellitus. The patient’s ability to handle a glucose load is decreased, and the transplacental passage of glucose may stimulate fetal secretion of insulin, in turn leading to neonatal hypoglycemia in the immediate postpartum period.20
Gastrointestinal Changes
Pregnant women are at increased risk for aspiration of gastric contents compared to the general population. Aspiration pneumonitis is estimated to occur in 0.1% of cesarean deliveries performed under general anesthesia.21 Airway difficulties present during pregnancy may contribute to this risk. In addition, gastric secretions are more acidic. Gastric emptying time is not prolonged during pregnancy, but overall gastrointestinal time is prolonged. In two recent studies of obese and nonobese, nonlaboring parturients at term, gastric emptying did not differ after ingestion of a moderate amount (300 mL) of water versus after an overnight fast.22,23 Recent obstetric anesthesia practice guidelines by the American Society of Anesthesiologists allow for oral intake of modest amounts of clear liquids in uncomplicated laboring patients and for similar intake in patients scheduled for uncomplicated cesarean delivery up to 2 hours prior to induction of anesthesia.24 However, the guidelines state that patients with additional risk factors for aspiration (e.g., morbid obesity, diabetes, difficult airway) or patients at increased risk for operative delivery (e.g., nonreassuring fetal heart rate [FHR] pattern) may have further restrictions of oral intake.
The lower esophageal sphincter (LES) may become distorted and incompetent and progesterone may decrease its tone. The risk of regurgitation depends, in part, on the gradient between the LES and intragastric pressures. The gravid uterus may increase intra-abdominal and intragastric pressures, decreasing the gradient. After succinylcholine administration in most patients, the gradient increases because the increase in LES pressure exceeds the increase in intragastric pressure. However, in parturients with “heartburn,” the LES tone is greatly reduced.25
Altered Drug Responses
The minimum alveolar concentration (MAC) for inhalation agents is decreased by 8 to 12 weeks’ gestation and may be related to an increase in progesterone levels.27 In addition, maximal cephalad block level after neuraxial administration of local anesthetics is higher in the second and third trimesters of pregnancy.28 Epidural venous engorgement, which decreases intrathecal volume, may lead to increased local anesthetic spread. Pregnancy increases median nerve sensitivity to lidocaine block29 and in vitro preparations from pregnant animals demonstrate increased susceptibility to local anesthetic blockade. This increased sensitivity may be due to progesterone or other hormonal mediators.
Placental Transfer and Fetal Exposure to Anesthetic Drugs
Most drugs, including many anesthetic agents, readily cross the placenta. Several factors influence the placental transfer of drugs, including physicochemical characteristics of the drug itself, maternal drug concentrations in the plasma, properties of the placenta, and hemodynamic events within the fetomaternal unit.
Drugs cross biologic membranes by simple diffusion, the rate of which is determined by the Fick principle, which states that:
where Q/t is the rate of diffusion, K is the diffusion constant, A is the surface area available for exchange, Cm is the concentration of free drug in maternal blood, Cf is the concentration of free drug in fetal blood, and D is the thickness of the diffusion barrier.
The diffusion constant (K) of the drug depends on physicochemical characteristics such as molecular size, lipid solubility, and degree of ionization. Compounds with a molecular weight of <500 Da are unimpeded in crossing the placenta, whereas those with molecular weights of 500 to 1,000 Da are more restricted. Most drugs commonly used by the anesthesiologist have molecular weights that permit easy transfer.
Drugs that are highly lipid soluble cross biologic membranes more readily. The degree of ionization is important because the nonionized moiety of a drug is more lipophilic than the ionized one. Local anesthetics and opioids are weak bases, with a relatively low degree of ionization and considerable lipid solubility. In contrast, muscle relaxants are more ionized and less lipophilic, and their rate of placental transfer is therefore more limited.
The relative concentrations of drug existing in the nonionized and ionized forms can be predicted from the Henderson–Hasselbalch equation:
The pKa is the pH at which the concentrations of free base and cation are equal. The ratio of base to cation becomes particularly important with local anesthetics because the nonionized form penetrates tissue barriers, such as the placenta. For the amide local anesthetics, the pKa values (7.7 to 8.1) are sufficiently close to physiologic pH so that changes in maternal or fetal acid–base status may significantly alter the proportion of ionized and
nonionized drugs present. At equilibrium, the concentrations of nonionized drug in the fetal and maternal plasma are equal. In an acidotic fetus, local anesthetics may be relatively more ionized than in maternal blood, and “ion trapping” may occur, leading to fetal drug accumulation.30
nonionized drugs present. At equilibrium, the concentrations of nonionized drug in the fetal and maternal plasma are equal. In an acidotic fetus, local anesthetics may be relatively more ionized than in maternal blood, and “ion trapping” may occur, leading to fetal drug accumulation.30
The effects of maternal plasma protein binding on the rate and amount of drug transferred to the fetus are not so well understood. In sheep, the low fetomaternal ratio of bupivacaine plasma concentrations has been attributed to the difference between fetal and maternal plasma protein binding, rather than to extensive fetal tissue uptake.31 However, if enough time is allowed for fetomaternal equilibrium to be approached, substantial accumulation of highly protein-bound drugs, such as bupivacaine, can occur in the fetus.32
Placenta
Maturation of the placenta can affect the rate of drug transfer to the fetus, as the thickness of the trophoblastic epithelium decreases from 25 to 2 mm at term. Uptake and biotransformation of anesthetic drugs by the placenta would decrease the amount transferred to the fetus. However, placental drug uptake is limited, and there is no evidence to suggest that this organ metabolizes any of the agents commonly used in obstetric anesthesia.
Hemodynamic Factors
Any factor decreasing placental blood flow (e.g., aortocaval compression, hypotension, or hemorrhage) can decrease drug delivery to the fetus. During labor, uterine contractions intermittently reduce perfusion of the placenta. If a uterine contraction coincides with a rapid decline in plasma drug concentration after an intravenous bolus injection, by the time perfusion has returned to normal, the concentration gradient across the placenta has been greatly reduced. Thus, an intravenous injection of diazepam, administered at the onset of contraction compared to during uterine diastole, results in less drug in infants of mothers in the former group.
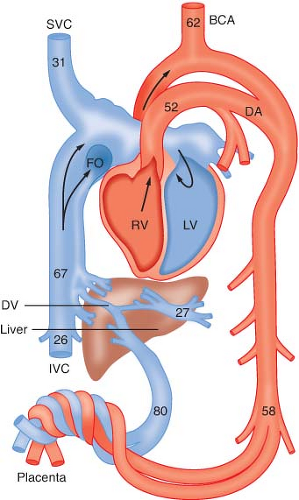
Several characteristics of the fetal circulation delay equilibration between the umbilical arterial and venous blood, and thus delay the depressant effects of anesthetic drugs (Fig. 40-1). The liver is the first fetal organ perfused by the umbilical venous blood, which carries drug to the fetus. Substantial uptake by this organ has been demonstrated for a variety of drugs, including thiopental, lidocaine, and halothane. During its transit to the arterial side of the fetal circulation, the drug is progressively diluted as blood in the umbilical vein becomes admixed with fetal venous blood from the gastrointestinal tract, the lower extremities, the head and upper extremities, and finally, the lungs. Because of this unique pattern of fetal circulation, continuous administration of anesthetic concentrations of nitrous oxide during elective cesarean sections caused newborn depression only if the induction-to-delivery interval exceeded 5 to 10 minutes. Rapid transfer of inhalation agents, including halothane, enflurane, and isoflurane, results in detectable umbilical arterial and venous concentrations after 1 minute.35 Because of the rapid decline in maternal plasma drug concentrations, administration of thiopental or thiamylal as a single-bolus injection not exceeding 4 mg/kg was followed by fetal arterial concentrations of barbiturate below a level that would result in neonatal depression.36
Fetal regional blood flow changes can also affect the amount of drug taken up by individual organs. For example, during asphyxia and acidosis, a greater proportion of the fetal cardiac output perfuses the fetal brain, heart, and placenta. Infusion of lidocaine resulted in increased drug uptake in the heart, brain, and liver of asphyxiated baboon fetuses compared with control fetuses that were not asphyxiated.37
Fetus and Newborn
Any drug that reaches the fetus undergoes metabolism and excretion. In this respect, the fetus has an advantage over the newborn in that it can excrete the drug back to the mother once the concentration gradient of the free drug across the placenta has been reversed. With the use of local anesthetics, this may occur even though the total plasma drug concentration in the mother may exceed that in the fetus because there is lower protein binding in fetal plasma.31 There is only one drug, 2-chloroprocaine, that is metabolized in the fetal blood so rapidly that even in acidosis, substantial accumulation in the fetus is avoided.34
In both the term and the preterm newborn, the liver contains enzymes essential for the biotransformation of amide local anesthetics. The metabolic clearance in the newborn is similar to, and renal clearance greater than, that in the adult. Elimination half-life is prolonged in the newborn due to a greater volume of distribution of the drug. Prolonged elimination half-lives in the newborn compared with the adult have been noted for other amide local anesthetics.
It is not completely understood whether the fetus and the newborn are more sensitive than the adult to the depressant and toxic effects of local anesthetics. The relative central nervous and cardiorespiratory toxicity of lidocaine has been studied in adult ewes and lambs (fetal and neonatal).38 The doses required to produce toxicity in the fetal and neonatal lambs were greater than those required in the adult, although serum concentrations at which toxicity occurred were not different. In the fetus, this was attributed to placental clearance of drug into the mother and better maintenance of blood gas tensions during convulsions. In the newborn, a larger volume of distribution was thought to be responsible for the higher doses needed to induce toxic effects.
Bupivacaine has been implicated as a possible cause of neonatal jaundice because its high affinity for fetal erythrocyte membranes may lead to a decrease in filterability and deformability, rendering them more prone to hemolysis (see Chapter 42). However, studies failed to show increased bilirubin production in newborns whose mothers received bupivacaine for epidural anesthesia during
labor and delivery.39 Finally, observational neurobehavioral studies have revealed subtle changes in newborn neurologic and adaptive functions. In the case of most anesthetic agents, these changes are minor and transient, lasting for only 24 to 48 hours.
labor and delivery.39 Finally, observational neurobehavioral studies have revealed subtle changes in newborn neurologic and adaptive functions. In the case of most anesthetic agents, these changes are minor and transient, lasting for only 24 to 48 hours.
Analgesia for Labor and Vaginal Delivery
Most women experience moderate-to-severe pain during parturition. In the first stage of labor, pain is caused by uterine contractions, associated with dilation of the cervix and stretching of the lower uterine segment. Pain impulses are carried in visceral afferent type C fibers accompanying the sympathetic nerves. During the first stage of labor, pain is referred to the T10 to L1 spinal cord segments. In the late first and second stages of labor, additional pain impulses from distention of the vaginal vault and perineum are carried by the pudendal nerves, composed of lower sacral fibers (S2 to S4).
Well-conducted obstetric analgesia, in addition to relieving pain and anxiety, may have other benefits. Pain may result in maternal hypertension and reduced uterine blood flow. During the first and second stages of labor, epidural analgesia blunts the increases in maternal cardiac output, heart rate, and blood pressure that occur with painful uterine contractions and “bearing-down” efforts.40 In reducing maternal secretion of catecholamines, epidural analgesia may convert a previously dysfunctional labor pattern to normal. Maternal analgesia may also benefit the fetus by eliminating maternal hyperventilation, which often leads to reduced fetal arterial oxygen tension because of a leftward shift of the maternal oxygen–hemoglobin dissociation curve.
Nonpharmacologic Methods of Labor Analgesia
Nonpharmacologic methods to relieve the pain of childbirth include childbirth education, emotional support, massage, aromatherapy, audiotherapy, and therapeutic use of hot and cold. More specialized techniques that require specialized training or equipment include hydrotherapy, intradermal water injections, biofeedback, transcutaneous electrical nerve stimulation, acupuncture or acupressure, and hypnosis. Conclusions regarding the efficacy of most of these techniques are not possible, as the techniques have been inadequately studied because of methodologic flaws in many study designs.42
Prepared Childbirth and Psychoprophylaxis
The philosophy of prepared childbirth maintains that lack of knowledge, misinformation, fear, and anxiety can heighten a patient’s response to pain and consequently increase the need for analgesics. The most popular method of prepared childbirth was introduced by Lamaze. It provides an educational program on the physiology of parturition and attempts to diminish cortical pain perception by encouraging responses such as specific patterns of breathing and focused attention on a fixed object.43 Scientific data as to whether childbirth education and psychoprophylaxis are effective in reducing childbirth pain are inconsistent and lack scientific rigor. Education, intense motivation, and cultural influences can influence the affective and behavioral responses to pain, although they probably minimally affect actual pain sensation.
Other Nonpharmacologic Methods
Continuous labor support refers to the presence during labor of nonmedical support by a trained person. Prospective, controlled trials and several systematic analyses have concluded that women who receive continuous labor support have shorter labors, fewer operative deliveries, fewer analgesic interventions, and overall satisfaction.44 Systematic reviews of randomized controlled trials of hydrotherapy (water baths) have concluded that women experience less pain and use less analgesia, without change in the duration of labor, rate of operative delivery, or neonatal outcome.45 Intradermal water injection consists of the injection of 0.05 to 0.1 mL of sterile water at four sites on the lower back to treat back pain during labor. Randomized controlled trials have found that the technique is effective in reducing severe back pain during labor, without any known side effects to the mother and the fetus.46 Hypnosis requires prenatal training of the mother by a trained hypnotherapist. A meta-analysis of five randomized controlled trials concluded that women randomized to hypnosis used pharmacologic analgesia methods at a lower rate compared with women in the control groups.42,47 The results of studies using transcutaneous electrical nerve stimulation are inconsistent, but in general, labor pain does not appear to be lessened, nor does it lower the use of other analgesic modalities.47 In a meta-analysis including 13 trials, women who were randomized to receive acupuncture or acupressure versus control (no or “false” acupuncture) had modestly lower pain scores.48 Similarly, relaxation techniques may also reduce pain intensity and satisfaction with pain relief compared to standard care.49
Systemic Medication
The advantages of systemic analgesics include ease of administration and patient acceptability. However, the drug, dose, time, and method of administration must be chosen carefully to avoid maternal or neonatal depression. Opioids are used most commonly, although tranquilizers and ketamine are used occasionally.
Opioids
Systemic opioids are commonly administered for labor analgesia, although existing data suggest that they provide little significant analgesia50,51 (see Chapter 19). Meperidine has historically been the most commonly used systemic analgesic for the treatment of labor pain. However, in the past decade, because of concerns of the lack of efficacy and the presence of side effects, there has been a move away from its use for both labor pain and other pain conditions.52 Meperidine can be administered by intravenous injection (effective analgesia in 5 to 10 minutes) or intramuscularly (peak effect in 40 to 50 minutes). The major side effects are a high incidence of nausea and vomiting, maternal sedation, dose-related depression of ventilation, orthostatic hypotension, and the
potential for neonatal depression. Meperidine may cause transient alterations of the FHR, such as decreased beat-to-beat variability and mild tachycardia. Among other factors, the risk of neonatal depression is related to the interval from the last drug injection to delivery. The placental transfer of an active metabolite, normeperidine, which has a long elimination half-life in the neonate (62 hours), has also been implicated in contributing to neonatal depression and subtle neonatal neurobehavioral dysfunction.
potential for neonatal depression. Meperidine may cause transient alterations of the FHR, such as decreased beat-to-beat variability and mild tachycardia. Among other factors, the risk of neonatal depression is related to the interval from the last drug injection to delivery. The placental transfer of an active metabolite, normeperidine, which has a long elimination half-life in the neonate (62 hours), has also been implicated in contributing to neonatal depression and subtle neonatal neurobehavioral dysfunction.
Synthetic opioids such as fentanyl, alfentanil, and remifentanil are more potent than meperidine; however, their use during labor is limited by their short duration of action. These drugs offer an advantage when analgesia of rapid onset but short duration is necessary (e.g., with forceps application). For more prolonged analgesia, fentanyl or remifentanil can be administered with patient-controlled delivery devices.53 Patient-controlled analgesia administration of opioids does carry with it the potential for drug accumulation and the risk of neonatal depression. Remifentanil has the theoretical advantage of rapid onset and offset compared with the other opioids. Bolus doses ranging from 0.2 to 1 μg/kg with lockout intervals from 1 to 5 minutes and background infusion rates from 0 to 0.1 μg/kg/min54 have been described. However, as with other systemic opioid techniques, it is unclear whether remifentanil patient-controlled analgesia can provide satisfactory analgesia without an unacceptably high incidence of maternal, fetal, and neonatal side effects.54
Opioid agonists–antagonists, such as butorphanol and nalbuphine, have also been used for obstetric analgesia. These drugs have the proposed benefits of a lower incidence of nausea, vomiting, and dysphoria, as well as a “ceiling effect” on depression of ventilation. Butorphanol, 1 to 2 mg, or nalbuphine, 10 mg by intravenous or intramuscular injection, is probably the most popular. Unlike meperidine, these are biotransformed into inactive metabolites and have a ceiling effect on depression of ventilation.
Naloxone, a pure opioid antagonist, should not be administered to the mother shortly before delivery to prevent neonatal ventilatory depression because it reverses maternal analgesia at a time when it is most needed. In addition, in some instances, it has caused maternal pulmonary edema and even cardiac arrest. If necessary, the drug should be given directly to the newborn intramuscularly (0.1 mg/kg).
Ketamine
Ketamine is a potent analgesic. However, it may also induce unacceptable amnesia that may interfere with the mother’s recollection of the birth. Nonetheless, ketamine is a useful adjuvant to inadequate regional analgesia during vaginal delivery or for obstetric manipulations. In low doses (0.2 to 0.4 mg/kg), ketamine provides adequate analgesia without causing neonatal depression. Constant communication is required with the patient to ensure that she is awake and able to protect her airway.
Regional Analgesia
Regional techniques provide excellent analgesia with minimal depressant effects on the mother and the fetus. The regional techniques most commonly used in obstetric anesthesia include central neuraxial blocks (spinal, epidural, and combined spinal/epidural [CSE]), paracervical and pudendal blocks, and, less frequently, lumbar sympathetic blocks (LSBs). Hypotension resulting from sympathectomy is the most frequent complication of central neuraxial blockade. Therefore, maternal blood pressure should be monitored at regular intervals, typically every 2 to 5 minutes for approximately 15 to 20 minutes after the initiation of the block and at routine intervals thereafter. The use of regional analgesia may be contraindicated in the presence of coagulopathy, acute hypovolemia, or infection at the site of needle insertion. Chorioamnionitis without frank sepsis is not a contraindication to central neuraxial blockade in obstetrics, provided antibiotics have been administered.
Because of ethical considerations and methodologic difficulties, it is difficult to design clinical studies to examine the effects of neuraxial analgesia on the progress of labor and mode of delivery. Randomized controlled trials have found no difference in the rate of cesarean delivery in women who received neuraxial compared with systemic opioid labor analgesia.55 Meta-analyses suggests that neuraxial analgesia does not prolong the first stage of labor, although the data are heterogeneous.55 There has been concern that early initiation of epidural analgesia during the latent phase of labor (<4 cm cervical dilation) in nulliparous women may result in a higher incidence of dystocia and cesarean delivery. Recent large randomized trials and a meta-analysis of these trials found no difference in the rate of cesarean delivery in women randomized to early neuraxial compared with systemic opioid analgesia.56 Neuraxial analgesia is, however, associated with prolongation of the second stage of labor in nulliparous women, possibly owing to a decrease in expulsive forces or malposition of the vertex.55 Thus, the American College of Obstetricians and Gynecologists has redefined an abnormally prolonged second stage of labor as >3 hours in nulliparous and 2 hours in multiparous women with epidural analgesia. Prolongation of the second stage may be minimized by the use of dilute local anesthetic solutions in combination with opioid.57
Epidural Analgesia
Epidural analgesia may be used for pain relief during labor and vaginal delivery, and if necessary, converted to anesthesia for cesarean delivery. Effective analgesia during the first stage of labor may be achieved by blocking the T10 to L1 dermatomes with low concentrations of local anesthetic, usually combined with lipid-soluble opioids. Combining drugs allows the use of lower doses of both drugs, thus minimizing side effects and complications of each. For the second stage of labor and delivery, the nerve block should be extended to include the S2 to S4 segments in order to block pain for vaginal and perineal distension and trauma.
Long-acting amides such as bupivacaine or ropivacaine are most frequently used because they produce excellent sensory analgesia while sparing motor function, particularly at low concentrations (<0.1%). Although some studies have found that ropivacaine is associated with less motor blockade than equipotent doses of bupivacaine, there was no difference in the rate of instrumental vaginal delivery among women randomized to receive epidural levobupivacaine, bupivacaine, or ropivacaine for maintenance of labor analgesia.58
Analgesia for the first stage of labor may be achieved with 5 to 10 mL of bupivacaine or ropivacaine (0.125%) combined with fentanyl (50 to 100 μg) or sufentanil (5 to 10 μg). There is controversy regarding the need for a test dose when using dilute solutions of local anesthetic. Because catheter aspiration is not always diagnostic, particularly when using single-orifice epidural catheters, some experts believe that a test dose should be administered to improve detection of an intrathecally or intravascularly placed catheter.59
Analgesia may be maintained with a continuous infusion (8 to 12 mL/hr) of bupivacaine (0.0625% to 0.1%) or ropivacaine (0.08% to 0.15%). The addition of fentanyl (1 to 2 μg/mL) or sufentanil (0.3 to 0.5 μg/mL) is often required and will allow for more dilute local anesthetic solutions to be administered. Alternatively, analgesia may be maintained with patient-controlled epidural analgesia (PCEA) with similar solutions of local anesthetic and
opioid. PCEA resulted in greater patient satisfaction, a lower average hourly dose of bupivacaine (and therefore less motor block), and less need for physician intervention60,61 compared with a continuous epidural infusion. Protocols for PCEA vary widely. Data are conflicting as to whether a background infusion improves analgesia; however, a background infusion may be helpful in selected parturients (e.g., nulliparas with long labors).61 Common PCEA parameters include a parturient administered bolus dose of 5 to 10 mL, a lock-out interval of 10 to 20 minutes, and a background infusion of 0 to 10 mL/hr. Thirty percent to 50% of the hourly dose is often administered as a background infusion.
opioid. PCEA resulted in greater patient satisfaction, a lower average hourly dose of bupivacaine (and therefore less motor block), and less need for physician intervention60,61 compared with a continuous epidural infusion. Protocols for PCEA vary widely. Data are conflicting as to whether a background infusion improves analgesia; however, a background infusion may be helpful in selected parturients (e.g., nulliparas with long labors).61 Common PCEA parameters include a parturient administered bolus dose of 5 to 10 mL, a lock-out interval of 10 to 20 minutes, and a background infusion of 0 to 10 mL/hr. Thirty percent to 50% of the hourly dose is often administered as a background infusion.
Women with hemodynamic stability and preserved motor function who do not require continuous fetal monitoring may ambulate with the assistance of a partner during the first stage of labor. Before ambulation, women should be observed for 30 minutes after initiation of neuraxial blockade to assess maternal and fetal well-being.
During delivery, the sacral dermatomes may be blocked with 10 mL of bupivacaine (0.25% to 0.5%), lidocaine (1.0%), or 2-chloroprocaine (2% to 3%). Many parturients have adequate analgesia for delivery without an additional bolus dose, particularly if epidural analgesia has been maintained for a long interval (hours). However, instrumental vaginal delivery may require a more dense block than that obtained with dilute local anesthetic solutions.
Spinal Analgesia
A single subarachnoid injection for labor analgesia has the advantage of fast and reliable onset of neural blockade, and it is technically easier to initiate compared with epidural analgesia. However, repeated intrathecal injections may be required for a long labor, thus increasing the risk of postdural puncture headache (PDPH). Spinal analgesia with fentanyl (15 to 25 μg) or sufentanil (2 to 5 μg) in combination with plain bupivacaine (1.25 to 2.5 mg) may be appropriate in the multiparous patient whose anticipated course of labor does not warrant a catheter technique (duration, 1.5 hours). A potential disadvantage of single-shot spinal analgesia is that the duration of labor, even in a rapidly progressing multiparous woman, may be longer than anticipated. Furthermore, if the woman requires an urgent cesarean delivery, a new anesthetic will need to be initiated. However, spinal anesthesia (a “saddle block”) is a safe and effective alternative to general anesthesia or pudendal nerve block for instrumental delivery in parturients without pre-existing epidural analgesia.
Combined Spinal/Epidural Analgesia
CSE analgesia is an ideal analgesic technique for use during labor. CSE combines the rapid, reliable onset of profound analgesia resulting from spinal injection with the flexibility and longer duration associated with a continuous epidural technique. After identification of the epidural space using a conventional (or specialized) epidural needle, a longer (127 mm), pencil-point spinal needle is advanced into the subarachnoid space through the epidural needle. After intrathecal injection, the spinal needle is removed and an epidural catheter is inserted. Intrathecal injection of fentanyl (10 to 25 μg) or sufentanil (2.5 to 5 μg) alone or more commonly in combination with bupivacaine (1.25 to 2.5 mg) produces profound analgesia lasting for 90 to 120 minutes with minimal motor block. Spinal opioid alone provides complete analgesia for the early latent phase of labor. However, the addition of bupivacaine is necessary for satisfactory analgesia during advanced labor. Continuous epidural analgesia or PCEA may be initiated following the spinal injection.
The most common side effects of intrathecal opioids are pruritus, nausea, vomiting, and urinary retention. The incidence of pruritus is lower if opioid is coadministered with local anesthetic.62 Rostral spread resulting in delayed respiratory depression is rare with fentanyl and sufentanil, and usually occurs within 30 minutes of injection. Transient nonreassuring FHR patterns may occur after initiation of both epidural and spinal analgesia, with and without opioids; however, the incidence may be higher after CSE compared to epidural analgesia.63 Presumably, uterine hypertonus and decreased uteroplacental perfusion occur as a result of rapid decrease in circulating maternal epinephrine levels after initiation of analgesia or as a result of hypotension after sympatholysis. The incidence of emergency cesarean delivery, however, is no greater after CSE than after conventional epidural analgesia.63,64
Mothers in early labor, or with preload-dependent medical conditions (e.g., aortic stenosis), may particularly benefit from opioid-only CSE. Spinal opioid provides complete analgesia without the need for local anesthetic in early labor, thus avoiding an acute decrease in preload, and almost always allowing motivated women to ambulate because there is no motor block. Multiparous women with advanced cervical dilation also benefit from CSE analgesia in which both intrathecal opioid and local anesthetic are injected. The onset of sacral analgesia is accomplished significantly faster with much less drug than initiation of lumbar epidural analgesia. However, because the epidural component of a CSE is not initially tested, CSE analgesia should be used with caution in women who may require urgent cesarean delivery or are at increased risk from general anesthesia (e.g., morbidly obese or anticipated difficult airway).
Paracervical Block
Bilateral paracervical block interrupts transmission of nerve impulses from the uterus and cervix during the first stage of labor. Five to 10 milliliters of dilute local anesthetic solution is injected submucosally via a needle guide in the vagina into the left and right lateral vaginal fornices. Although paracervical block effectively relieves pain during the first stage of labor, the technique has fallen out of favor during childbirth because it is associated with a high incidence of fetal asphyxia and poor neonatal outcome, particularly with the use of bupivacaine. Performing the block with dilute local anesthetic solutions, allowing 5 to 10 minutes to elapse between injections on the left and right sides, and limiting the block to women with <8 cm cervical dilation, may decrease the incidence of complications.
Paravertebral Lumbar Sympathetic Block
Paravertebral LSB is a reasonable alternative when contraindications exist to central neuraxial techniques. LSB interrupts the painful transmission of cervical and uterine impulses during the first stage of labor.65 Although there is less risk of fetal bradycardia with LSB compared with paracervical blockade, unfamiliarity and technical difficulties associated with the performance of the block and risks of intravascular injection have decreased its use in standard practice.
Pudendal Nerve Block
Inhalation Analgesia and General Anesthesia
Inhalation labor analgesia is rare in the United States, although its use is more common in other parts of the world (see Inhaled Anesthetics, Chapter 17). Nitrous oxide, 50% by volume, is the most commonly used inhalation agent for analgesia during labor, and the mother is trained to intermittently self-administer the gas at the onset of a contraction. Studies are conflicting as to whether nitrous oxide provides benefit to the parturient66; however, its use appears safe for the fetus and the neonate. A major disadvantage of inhalation analgesia is the need for a waste gas scavenging system.
General anesthesia is rarely used for vaginal delivery, and precautions against gastric aspiration must always be observed (see “General Anesthesia” under “Anesthesia for Cesarean Delivery”). General anesthesia may be required when time constraints prevent induction of regional anesthesia. Potent inhalation drugs (1.5 to 2 MAC for short periods) can provide uterine relaxation for obstetric maneuvers such as second twin delivery, breech presentation, or postpartum manual removal of a retained placenta. However, in current practice, intravenous nitroglycerin (50 to 250 μg) has largely replaced the need for general anesthesia for uterine relaxation.
Anesthesia for Cesarean Delivery
The most common indications for cesarean delivery include arrest of dilation, nonreassuring fetal status, cephalopelvic disproportion, malpresentation, prematurity, prior cesarean delivery, and prior uterine surgery involving the corpus. The choice of anesthesia depends on the urgency of the procedure, the condition of the mother and the fetus, and the mother’s wishes.
A 2001 survey of obstetric anesthesia practices in the United States revealed that most patients undergoing cesarean delivery do so under spinal or epidural anesthesia.67 Neuraxial techniques have several advantages, such as:
Help to avoid the necessity of airway manipulation
Lessen the risk of gastric aspiration
Avoid the use of depressant anesthetic drugs
Allow the mother to remain awake during delivery
May be associated with less operative blood loss
Compared with general anesthesia, there is less immediate neonatal depression after neuraxial compared with general anesthesia.
Neuraxial Anesthesia
Blockade to the T4 dermatome is necessary to perform cesarean delivery without maternal discomfort. The most common complication of neuraxial anesthesia is hypotension and the attendant risk of decreased uteroplacental perfusion (see “Hypotension” under “Anesthetic Complications”). Measures to decrease the incidence and severity of hypotension include left uterine displacement, intravenous fluid administration, and the liberal use of vasopressors to prevent and treat hypotension.
Most anesthesiologists administer a nonparticulate antacid before induction of anesthesia for pulmonary aspiration prophylaxis. Some practitioners also administer an H2 receptor antagonist and metoclopramide. Sedative premedication is usually not necessary. Intraoperative monitoring mimics that for all anesthetics, although blood pressure should be measured frequently (every several minutes) for the first 20 minutes after initiation of anesthesia. Although supplemental oxygen is frequently administered, there is no evidence of benefit to the mother, the fetus, or the neonate.68,69
Multimodal analgesia, including systemic nonsteroidal anti-inflammatory drugs and neuraxial opioids and/or local anesthetics, is optimal for postoperative analgesia. Abdominal wall nerve block techniques (transversus abdominis plane [TAP] block) have also been described after cesarean delivery.70 Although postcesarean delivery analgesia should take the nursing infant into account, very small amounts of drugs administered to the mother actually cross into breast milk, and even smaller amounts are absorbed from the neonatal gut. Prolonged (12 to 24 hours) postoperative pain relief in the postpartum patient can be provided by intrathecal morphine (100 to 150 μg)71 or epidural morphine (3.5 to 4.0 mg).72 Intrathecal morphine provides superior and longer-lasting analgesia compared to bilateral TAP block.73 PCEA with a dilute solution of local anesthetic and lipid-soluble opioid is another option after epidural anesthesia. Side effects of neuraxial morphine include nausea, vomiting, and pruritus. Delayed respiratory depression is a rare but potentially devastating complication; therefore, the patient must be monitored carefully in the postoperative period.74 Morbidly obese women may be at higher risk for respiratory depression.
Spinal Anesthesia
Despite an adequate dermatomal level for surgery, women may experience varying degrees of visceral discomfort and nausea and vomiting, particularly during exteriorization of the uterus and traction on abdominal viscera. Improved perioperative anesthesia and analgesia can be provided with the addition of fentanyl (10 to 20 μg), sufentanil (2.5 to 5 μg), or morphine (0.1 to 0.15 mg) to the local anesthetic solution. Fentanyl has a rapid onset, but is short acting and provides little additional postoperative analgesia. In contrast, morphine has a longer latency than fentanyl, but will also provide anesthesia for 12 to 18 hours after delivery.
Lumbar Epidural Anesthesia
In contrast to spinal anesthesia, epidural anesthesia is associated with a slower onset of action and a larger drug requirement to establish adequate sensory block. The major advantages of epidural compared with spinal anesthesia are the ability to titrate the extent and duration of anesthesia. To avoid inadvertent intrathecal or intravascular injection, correct placement of the epidural needle and catheter is essential. This is especially true because epidural anesthesia for cesarean delivery necessitates the administration of large doses of local anesthetic.
Aspiration of the epidural catheter for blood or cerebrospinal fluid is not reliable for detection of catheter misplacement, particularly with single-orifice catheters. Thus, most anesthesiologists administer a test dose before the initiation of surgical anesthesia. A small dose of local anesthetic (e.g., lidocaine, 45 mg, or bupivacaine, 5 mg) readily produces identifiable sensory and motor blocks if injected intrathecally. Addition of epinephrine (15 μg) with careful hemodynamic monitoring may signal intravascular injection if followed by a transient increase in heart rate and blood pressure. The use of an epinephrine test dose (15 μg) in obstetrics is controversial because false-positive results do occur
(10% increase in heart rate), especially in laboring women. In addition, epinephrine may reduce uteroplacental perfusion. Rapid injection of 1 mL of air with simultaneous precordial Doppler monitoring appears to be a reliable indicator of intravascular catheter placement.75 Fentanyl, 100 μg, has also been used to test epidural catheter placement (significant reduction in pain).76 A negative test, although reassuring, does not eliminate the need for incremental administration of local anesthetic.
(10% increase in heart rate), especially in laboring women. In addition, epinephrine may reduce uteroplacental perfusion. Rapid injection of 1 mL of air with simultaneous precordial Doppler monitoring appears to be a reliable indicator of intravascular catheter placement.75 Fentanyl, 100 μg, has also been used to test epidural catheter placement (significant reduction in pain).76 A negative test, although reassuring, does not eliminate the need for incremental administration of local anesthetic.
The most commonly used agents for obstetric epidural anesthesia are 2% lidocaine with epinephrine, 5 μg/mL (1:200,000) and 3% 2-chloroprocaine. Adequate anesthesia is usually achieved with 15 to 25 mL of local anesthetic solution, administered in divided doses over 5 to 10 minutes. 2-Chloroprocaine provides rapid onset of a reliable block with minimal risk of systemic toxicity because of its extremely high rate of metabolism in maternal and fetal plasma. However, 2% lidocaine with epinephrine and sodium bicarbonate (1 mEq/10 mL lidocaine) may also be used when the rapid conversion of pre-existing epidural labor analgesia to surgical anesthesia is required for urgent cesarean delivery. Lidocaine has an onset and duration intermediate to those of 2-chloroprocaine and bupivacaine. Lidocaine should be administered with epinephrine, as lidocaine without epinephrine does not consistently provide satisfactory surgical anesthesia. Bupivacaine is no longer commonly used for obstetric epidural anesthesia, as it is associated with a greater risk of cardiac toxicity compared with other amide local anesthetics. Unintentional intravascular injection of bupivacaine is associated with a high incidence of maternal mortality.77 Ropivacaine 0.5% combined with fentanyl may be used for surgical anesthesia, as the risk of toxicity is less than that of bupivacaine. The duration of motor block is shorter after ropivacaine compared with bupivacaine, but there are no differences in latency, quality of anesthesia, and duration of block.78,79
Combined Spinal/Epidural Anesthesia
Advantages of CSE anesthesia for cesarean delivery include the rapid onset of a dense block with a low anesthetic dose, and the ability to extend the duration of anesthesia, and perhaps to provide continuous postoperative analgesia. There is a lower incidence of breakthrough pain and intraoperative shivering, and maternal satisfaction was higher after CSE compared with epidural anesthesia for cesarean delivery.80 Several variations of the CSE technique have been described. The standard technique uses the same spinal dose of local anesthetic as one would use for standard spinal anesthesia. In sequential CSE anesthesia, a smaller spinal dose is expected to result in inadequate anesthesia for some patients. After 15 minutes, if anesthesia is inadequate, the block is extended by injecting supplemental local anesthetic via the epidural catheter.81 Although the incidence of hypotension is lower with this technique compared with full-dose spinal anesthesia, the induction to incision time is prolonged. A third technique is also associated with a lower incidence of hypotension without prolonging onset time. A small dose of spinal local anesthetic is followed by the routine injection of additional anesthetic through the epidural catheter approximately 5 minutes after the intrathecal dose.82 Bupivacaine doses from 6 to 12 mg have been described for CSE anesthesia.
General Anesthesia
General anesthesia may be necessary when contraindications or relative contraindications exist to neuraxial anesthesia (e.g., coagulopathy, or moderate or severe aortic stenosis), or when the need for emergency delivery precludes central neuraxial blockade. General anesthesia should be used cautiously in women with asthma, upper respiratory tract infection, obesity, or a history of difficult tracheal intubation. Preoperative airway evaluation is particularly important in pregnant women because the inability to intubate the trachea and provide effective ventilation is the leading cause of maternal death related to anesthesia.83 Equipment to manage the difficult airway equipment should be immediately available.86 Mallampati classification scores worsen during labor in some parturients.18 If airway difficulties are anticipated, a neuraxial anesthetic technique should be considered or an awake tracheal intubation performed. Pulmonary aspiration prophylaxis should be administered and the patient should be positioned with left lateral tilt to prevent aortocaval compression. Monitoring mimics that for all anesthetics.
To minimize the risk of hypoxemia during induction, denitrogenation for 3 to 5 minutes with a tight-fitting mask is essential. In an emergency, four deep breaths with 100% oxygen may suffice. A “defasciculating” dose of a nondepolarizing muscle relaxant is not necessary. Although somewhat controversial,84 a rapid-sequence induction is usually performed. Induction with a sedative–hypnotic (e.g., propofol [2 mg/kg], ketamine [1 mg/kg], or etomidate [0.2 to 0.3 mg/kg]) is followed by succinylcholine (1 to 1.5 mg/kg) to facilitate tracheal intubation. Succinylcholine is the preferred muscle relaxant; however, when its use is contraindicated, rocuronium (0.6 mg/kg) is an acceptable alternative. A trained assistant applies cricoid pressure until the airway is properly secured with a cuffed endotracheal tube. Once correct placement of the endotracheal tube is confirmed with capnography and auscultation, the obstetrician may proceed with incision.
If there is difficulty in securing the airway, the mother should be ventilated with 100% oxygen before a subsequent attempt at tracheal intubation is made. Although some experts advise attempting to maintain cricoid pressure throughout, this practice may actually make visualization of the glottis and mask ventilation more difficult in some patients. The American Society of Anesthesiologists difficult airway algorithm85 should be modified to include assessment of fetal status and the need for immediate delivery (Fig. 40-2). It may be safer for the mother to allow her to awaken and to reassess the method of induction and intubation, rather than to persist with traumatic efforts at tracheal intubation. However, if the fetus is in extremis, airway management with a mask or supraglottic airway device may be an acceptable alternative.86
In the interval between intubation and delivery, anesthesia is maintained with a 50:50 mixture of nitrous oxide in oxygen and a volatile anesthetic agent. In the past, it was common to limit the volatile agent concentration to 0.5 MAC to limit fetal exposure before delivery and to limit uterine relaxation after delivery. However, the incidence of intraoperative awareness appears to be unacceptably high with this technique.87 Indeed, a significant number of women had bispectral index values >60 during general anesthesia with sevoflurane 1% in nitrous oxide 50%.88 Therefore, higher concentrations of volatile agent should be used before delivery. After delivery, the nitrous oxide concentration can be increased and/or an intravenous amnestic (e.g., midazolam) and opioids can be administered.
General anesthesia for cesarean delivery is associated with lower neonatal Apgar scores at 1 minute compared with neuraxial anesthesia89; however, the Apgar scores at 5 minutes are comparable. Therefore, an individual trained in neonatal resuscitation should be present at delivery of the infant. After delivery, intravenous oxytocin is administered to decrease the risk of uterine atony and anesthesia is deepened with an opioid and benzodiazepine, as necessary. At the end of the procedure, the mother’s trachea is extubated once she is awake and extubation criteria have been met. The usual blood loss at a cesarean delivery is 750 to 1,000 mL; transfusion is rarely necessary.
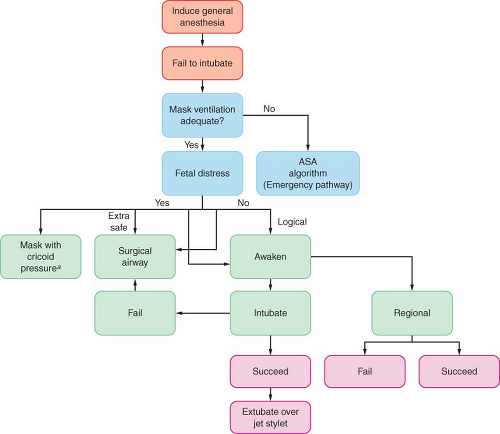 Figure 40.2. Management of the difficult airway in pregnancy with special reference to the presence or absence of fetal distress. When mask ventilation is not possible, the clinician is referred to the American Society of Anesthesiologists (ASA) algorithm for the emergency airway management found in Chapter 27. (Reprinted from: Kuczkowski KM, Reisner LS, Benumof JL. The difficult airway: Risk, prophylaxis, and management, Obstetric Anesthesia: Principles and Practice, 3rd ed. Chestnut DH, ed. St. Louis: Elsevier-Mosby; 2004:550, with permission.) aConventional face mask or laryngeal mask airway. |
Anesthetic Complications
Maternal Mortality
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree