3 Management of Acute Pain in the Intensive Care Unit
Critically ill patients frequently experience acute pain, but assessment rates for pain remain below 40% in mechanically ventilated patients.1 Pain and discomfort can have multiple causes in the intensive care unit (ICU) setting, including surgical and posttraumatic wounds, the use of invasive monitoring devices and mechanical ventilators, prolonged immobilization, and routine nursing care (e.g., dressing changes, airway suctioning).1–3 Pain is defined by the International Association for the Study of Pain (IASP-1979) as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage.”4 The experience of pain differs among patients, but the physiologic sequelae of inadequately treated pain are relatively predictable and potentially deleterious. Some physiologic responses to acute pain and stress are mediated by neuroendocrine activation and increased sympathetic tone. As a consequence, patients develop tachycardia, increased myocardial oxygen consumption, immunosuppression, hypercoagulability, persistent catabolism, and numerous other metabolic alterations.5 Additional morbidity may be incurred by pain-related functional limitations such as impaired pulmonary mechanics6 or delayed ambulation.
 Acute Pain Assessment
Acute Pain Assessment
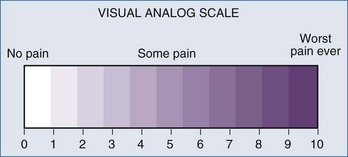
The past decade has seen an increase in the number of scales and assessment tools for the evaluation of sedation and analgesia in ICU patients. Several sedation scales—the Richmond Agitation Sedation Scale (RASS), Adaptation to the Intensive Care Environment (ATICE) tool, and the Minnesota Sedation Assessment Tool (MSAT)—as well as tools for assessment of analgesia in the ICU, such as the visual analog scale, the numeric rating scale, behavioral pain scale,7,8 and critical care pain observation scale, have been developed (Figure 3-1). The actual percentage of ICUs implementing formal sedation and analgesia protocols is approximately 50% in the United States. Unfortunately, many ICU patients cannot provide full (or even partial) information regarding their pain. However, the inability of ventilated, sedated ICU patients to report pain should not preclude pain management and does not rule out the possibility that the patients are experiencing pain.9 Caregivers sometimes must use signs of heightened sympathetic activity like hypertension, tachycardia, lacrimation, diaphoresis, and restlessness as surrogate indicators for the presence of pain. Trends in such signs provide a measure of the success of a given intervention.
 Options for Acute Pain Therapy
Options for Acute Pain Therapy
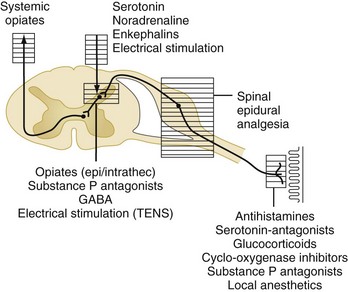
Acute pain is triggered by stimulation of peripheral nociceptors in the skin or deeper structures and is a complex process involving multiple mediators at various levels of the neuraxis (Figure 3-2).4 Different parts of the pain pathway can be targeted either individually or as part of a comprehensive strategy aimed at multiple sites for additive or synergistic effects. Thus, nociception can be influenced (1) peripherally by the use of nonsteroidal antiinflammatory drugs (NSAIDs) and nerve blocks, (2) at the spinal cord level by the use of epidural or intrathecal medications, and (3) centrally by the use of systemic medications.
Nonsteroidal Antiinflammatory Drugs
Drugs in this class inhibit cyclooxygenase (COX) enzymes, which are involved in synthesis of prostaglandins and related inflammatory mediators in response to injury. COX-1 is a constitutive enzyme that is present in most tissues and, through the production of prostaglandins E2 and I2, serves homeostatic and protective functions.10 COX-2 is an inducible enzyme that is expressed in response to inflammation. NSAIDs are commonly used in conjunction with other agents such as opioids to take advantage of different side-effect profiles and possible synergistic efficacy. As a class, NSAIDs can cause adverse effects that include nausea, gastrointestinal (GI) bleeding, inhibition of platelet function, operative site bleeding, renal insufficiency, and bronchospasm in aspirin-sensitive patients (triad of asthma, nasal polyposis, and aspirin allergy).2,4
Ketorolac tromethamine (Toradol) is the only parenteral NSAID available in the United States. It has been shown to reduce postoperative opioid requirements and does not cause respiratory depression.11 However, prolonged use has been associated with a significant incidence of the aforementioned side effects (primarily GI bleeding and renal failure)12; consequently, ketorolac therapy should be limited to a maximum of 5 days.2 In addition, ketorolac should be used at decreased dosages or avoided altogether in patients at higher risk of such complications (e.g., advanced age, hypovolemia, preexisting renal insufficiency). This caution also applies to enterally administered NSAIDs.
Selective COX-2 inhibitors like celecoxib (Celebrex) are available for enteral administration, and injectable COX-2 agents are being studied primarily for the management of acute postoperative pain.10 The main advantage of these agents over their nonselective relatives lies in the promise of decreased GI side effects.10 A joint meeting of the U.S. Food and Drug Administration (FDA) Arthritis Advisory Committee and the Drug Safety and Risk Management Advisory Committee reaffirmed that COX-2 inhibitors are important treatment options for pain management and that the preponderance of data demonstrate that the cardiovascular risk associated with celecoxib is similar to that associated with commonly used older nonspecific NSAIDs.13
Acetaminophen is a paraaminophenol derivative with analgesic and antipyretic properties similar to those of aspirin. The mechanism of action of acetaminophen is still poorly defined. Recent evidence has suggested that it may selectively act as an inhibitor of prostaglandin synthesis in the central nervous system (CNS) rather than in the periphery. A meta-analysis of randomized controlled trials of acetaminophen for postoperative pain revealed that this analgesic induced a morphine-sparing effect of 20% (9 mg) over the first 24 hours postoperatively but did not reduce the incidence of morphine-related adverse effects.14 It was concluded that acetaminophen may be a viable alternative to NSAIDs in high-risk patients because of the lower incidence of adverse effects. Therefore, it may be appropriate to administer acetaminophen with NSAIDs or COX-2 inhibitors, since the analgesics in these two classes may act additively or synergistically to improve analgesia.15
Opioid Analgesics
A number of opioids are available (Table 3-1), and this drug class remains the mainstay of ICU analgesia. Morphine, hydromorphone (Dilaudid), and fentanyl are commonly used in ICUs in the United States and have been recommended as first-line narcotic analgesics.2 Opioids bind to a variable degree with opioid receptor subtypes (µ, δ, κ) located in the brain, spinal cord, and peripheral sites and modulate the transmission and processing of nociceptive signals.4 The clinical and pharmacologic properties of opioids depend on several variables: chemical and solubility properties, dosing regimen, patient characteristics (Box 3-1), and presence of active metabolites. Drugs that are often thought of as short acting (e.g., fentanyl) actually have a markedly prolonged duration of action if given repeatedly or as an infusion (Figure 3-3).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree