Fig. 15.1
Colonoscopy showing multiple diverticula (white arrows) without evidence of diverticulitis
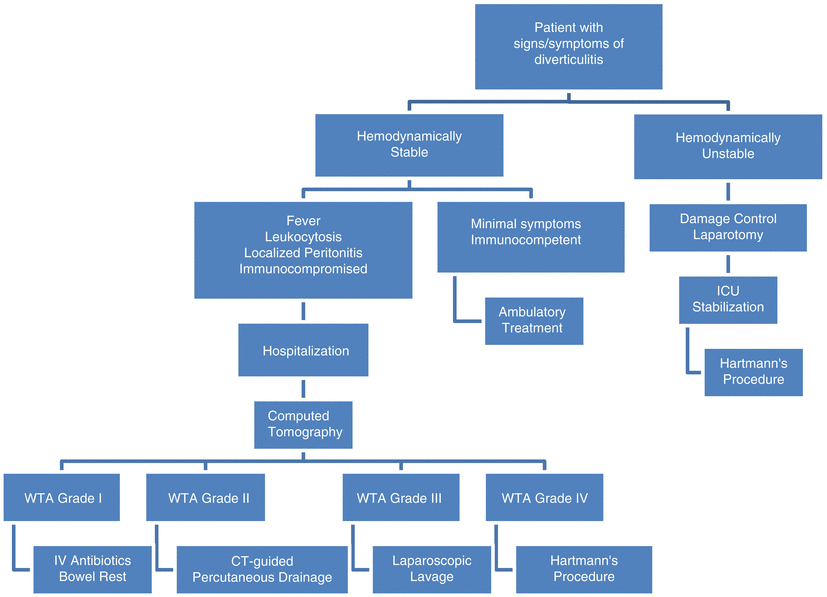
Complicated Diverticulitis
In contrast to uncomplicated diverticulitis, patients with acute, complicated diverticulitis typically present with localized or generalized peritonitis, fever, and leukocytosis. The diagnosis should be confirmed radiographically, with either computed tomography or magnetic resonance imaging. In 1978, Hinchey et al. published the first classification system to standardize the nomenclature describing the breadth of perforated diverticular disease [35]. This classification has been used since the 1970s, before the advent of computed tomography. It describes four stages of perforated disease. Stage I disease includes pericolic abscess or phlegmon. Stage II disease is a walled-off pelvic, intra-abdominal, or retroperitoneal abscess. Stage III disease describes generalized purulent peritonitis and stage IV disease is generalized feculent peritonitis. Since the advent of this classification system, few modifications have been introduced. The Western Trauma Association (WTA) consensus has modified this system to reflect contemporary management of complicated diverticular disease (Table 15.1).

Table 15.1
Western Trauma Association Complicated Diverticulitis Score

The optimal treatment of patients with complicated diverticulitis has undergone significant changes with the evolution of surgical techniques and nonoperative therapies. As late as the 1990s, the standard treatment for complicated diverticulitis was surgical resection and colostomy often performed in a multistage procedure, a technique described in the 1920s [36]. A paradigm shift has occurred mostly as the result of advances in antibiotics, interventional radiology, and critical care medicine.
Patients with WTA grade IA (phlegmon with no abscess (Fig. 15.2)) and grade IB (phlegmon with abscess <4 cm) should be treated with hospitalization, bowel rest, and intravenous antibiotics. Those who respond with resolution of pain, fever, and leukocytosis should be started on an oral diet and converted to oral antibiotics. Antibiotics should be continued for 14 days, from the initiation of intravenous antibiotics. Patients should undergo colonoscopy and be placed on a high-fiber diet following resolution of the acute inflammatory process. Those who fail conservative management should undergo definitive resection, described later in this chapter.
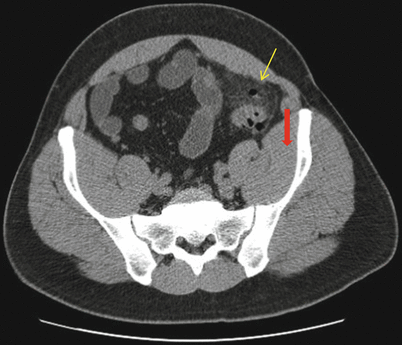

Fig. 15.2
Western Trauma Association grade IA complicated diverticulitis. Computed tomography shows sigmoid diverticulitis (red arrow) with pericolonic phlegmon and small focus of extraluminal air (yellow arrow)
Patients with WTA grade II disease (phlegmon with abscess >4 cm), or those with abscesses not responding to conservative therapy, should undergo CT-guided percutaneous drainage (PCD) [26, 37]. Seventy to ninety-three percent of patients amenable to CT-guided PCD can be successfully drained [38]. The preferred approach is transabdominal, either anterior or lateral. Other approaches include transgluteal, transperitoneal, transvaginal, or transanal. The potential advantage to performing CT-guided PCD is that it may avoid the need for urgent operation, allowing defervescence with resolution of some inflammation prior to a definitive operation. These admitted patients should be treated with intravenous antibiotics, while following their clinical exam for possible failure, such as increased abdominal pain, increasing leukocytosis, or hemodynamic instability. The reported failure rates for CT-guided PCD range from 15 to 30 %. Complications, including bleeding, perforation of a hollow viscus, and fistula formation, occur in up to 5 % of patients. Although some authors suggest CT-guided PCD may avoid the need for definitive resection altogether, the American Society of Colon and Rectal Surgeons does not endorse this concept currently.
In patients whom CT-guided PCD is not feasible, laparoscopic lavage and drainage may be an acceptable alternative. To perform the operation, pneumoperitoneum is established and a 12 mm umbilical trocar is placed. Two 5 mm operating trocars are then placed in the right lower quadrant and suprapubic positions to assist with manipulation and lavage (Fig. 15.3). The patient should be positioned in Trendelenburg with a left tilt, and the surgeon and assistant should be on the patient’s right side. The abdomen should be thoroughly inspected, and all omental, small bowel, pelvic structures, and abdominal wall attachments should be mobilized away from the inflamed sigmoid colon. However, vigorous manipulation of the diseased segment of colon should be avoided. If there is no evidence of free perforation of the colon, the four quadrants of the abdomen are lavaged until the effluent is clear. Should free perforation be identified, the diseased colon should be resected. A closed-suction drain is placed in the pelvis and externalized through the right lower quadrant trocar site. Multiple studies have reported the use of laparoscopic lavage in patients with Hinchey II–IV diverticulitis with low complication rates (<8 %) and without the need to convert to an open procedure [39–41]. Successful laparoscopic lavage may control sepsis and allow patients to be bridged for an elective one-stage operation. However, more recently two large multicenter, randomized trials have questioned the utility of laparoscopic lavage in perforated diverticulitis associated with purulent and feculent peritonitis. The laparoscopic lavage group of the Ladies trial sought to prove laparoscopic lavage for perforated diverticulitis would lead to a reduction in major morbidity and mortality [42]. During the 12-month follow-up, no significant difference was reported in the incidence of morbidity, including need for surgical re-intervention, abscess requiring drainage, facial dehiscence, or death. However, the combined 30-day morbidity and mortality rates were higher among the patients who underwent laparoscopic lavage (39 %) versus those who received a Hartmann’s procedure (HP) (19 %). This study, which was terminated early due to non-superiority, did not show a reduction of combined morbidity and mortality in patients who underwent laparoscopic lavage for perforated diverticulitis when compared with sigmoidectomy. The authors concluded that laparoscopic lavage for the treatment of diverticulitis is not superior to sigmoidectomy. The SCANDIV randomized clinical trial postulated that laparoscopic lavage was superior to colon resection in patients with perforated diverticulitis [43]. Primary outcomes included any complication that required re-intervention, life-threatening organ dysfunction, or death within 90 days. 101 patients were randomized to the lavage group, while 96 were randomized to the resection group. No statistically significant difference was found between the two groups. Among patients with fecal peritonitis, statistically significant differences were observed in the secondary outcomes. Considerably more patients in the lavage group required a secondary surgical procedure because of complications, including secondary peritonitis and intra-abdominal abscess formation. These findings led the authors to conclude that laparoscopic lavage should not be used in the treatment of perforated diverticulitis. The current role of laparoscopic lavage for perforated diverticulitis continues to evolve. Further studies are needed to determine the patient population in which this intervention may pose the most therapeutic benefit.
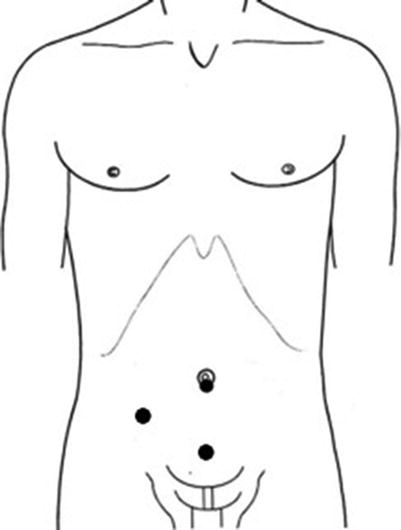

Fig. 15.3
Trocar placement for laparoscopic lavage. A 12 mm trocar is inserted at the umbilicus and two 5 mm trocars are placed in the right lower quadrant and suprapubic positions
Diverticulitis Associated with Free Perforation
Free perforation associated with acute diverticulitis is classified as WTA grade III (purulent/Hinchey III) or grade IV (feculent/Hinchey IV) peritonitis. Lidor et al. demonstrated that of more than 53,000 elderly patients studied, patients that underwent emergency surgery for complicated diverticulitis were older (76.8 vs. 73.9 years of age), had increased inhospital mortality, had a larger rate of intestinal diversion, and had higher 30-day readmission rates [44]. This population presents unique challenges, but the surgical management remains unchanged. Patients who present with florid sepsis, hypotension, and significant metabolic derangements represent a surgical emergency that requires rapid resuscitation, broad-spectrum antibiotics, and prompt operative therapy. Depending on the hemodynamic stability, a truncated or damage control laparotomy may be performed. In the unstable patient, a limited resection of the inflamed colon and temporary abdominal closure should be performed. The patient is then returned to the intensive care unit for further resuscitation. Once the physiologic abnormalities are corrected, the patient is returned to the operating room for peritoneal lavage, completion sigmoid colectomy, and end colostomy formation.
In patients who present with perforation, but whom are otherwise hemodynamically stable, a definitive resection may be undertaken. Ureteral stents should be used selectively in those patients with abscesses or excessive inflammation in the pelvis, which may aid in the identification of the ureters and protection from injury. The sigmoid colon is then mobilized, extending proximally to an area of noninflamed descending colon. The distal dissection should extend to the upper rectum. It should be noted that it is not necessary to resect all diverticula. The current standard of care therapy is Hartmann’s procedure, whereby the diseased colon is resected and a temporary end colostomy is performed. This procedure requires a second operation for reversal of the colostomy. However, there is currently much debate as whether to perform a primary resection with anastomosis (PRA) or a Hartmann’s procedure (HP). Primary resection and anastomosis of the affected colon segment has been used in an attempt to minimize the morbidity and mortality associated with the reversal of Hartmann’s procedure, which has a documented morbidity as high as 50 %. Therefore, the choice of operation should be influenced by factors such as patient age, comorbidities, degree of contamination, and mode of presentation. In a meta-analysis of 15 studies comparing PRA to HP, PRA was shown to have reduced morbidity and mortality, even when used in an emergency setting [45]. However, all studies were limited by significant patient selection bias, where patients with less comorbidities underwent PRA. Currently PRA should be reserved for patients with more favorable comorbid criteria, a lesser degree of contamination, and no preoperative hemodynamic instability.
Summary
Diverticular disease is commonly seen in the elderly population. Thorough history and physical examination combined with computer tomography will diagnose and grade nearly all patients. Treatment algorithms (see Fig. 15.4) will be based upon the patient’s preexisting medical comorbidities, physiologic status, and grade of diverticulitis. Newer concepts, including laparoscopic lavage and single-stage operations, are gaining acceptance and may become more routine as more data becomes available.