Fig. 16.1
Cerebral lobes depicted using a 3 dimensional (3D) reconstruction of functional brain MRI (fMRI)
Multiple functional areas in the cortex interact to produce conscious behavior, which includes the abilities to process sensation, communicate, retain memory, and coordinate movements. The sensory cortices allow for awareness of the environment, while the motor cortices control voluntary movement. Each of the primary sensory and motor cortices have secondary and tertiary cortices nearby to connect to association areas. These association areas integrate complex information for purposeful action.
16.2 Sensory Cortices
The main sensory cortices include the somatosensory, visual, auditory, gustatory, vestibular, and olfactory cortices, along with Wernicke’s (receptive speech) area (◘ Fig. 16.2). These primary cortices receive direct input from the peripheral sensory receptors. The sensory information is then transmitted from the primary cortices to the secondary cortices to perform higher order functions.
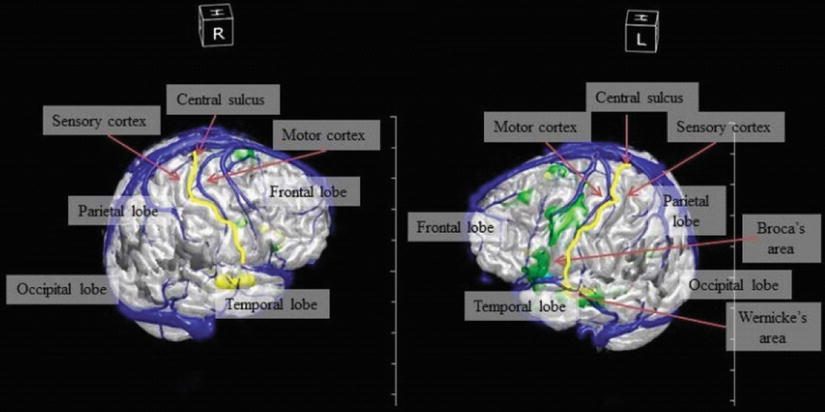

Fig. 16.2
Cerebral lobes and eloquent cortical areas depicted using a 3D reconstruction of functional brain MRI (fMRI)
16.2.1 Somatosensory Cortex
Located in the postcentral gyrus of the parietal lobe, the somatosensory cortex and adjacent somatosensory association cortex are able to process information from cutaneous stimulation, proprioceptors, and tactile receptors.
16.2.2 Visual Cortex
The visual cortex is located in the occipital lobe. Damage to the occipital lobe (or to the subcortical optic radiations and/or the thalamic lateral geniculate nucleus) can cause cortical blindness, visual field deficits, and visual hallucinations.
16.2.3 Auditory Cortex
The auditory cortex is located bilaterally in the temporal lobes. Unilateral damage to the auditory cortex does not cause bilateral deafness due to bilateral sound representation from set neuronal pathways.
16.2.4 Gustatory, Vestibular, and Olfactory Cortices
The gustatory and vestibular cortices are located in the insular region of the parietal lobe and receive sensory input from the taste buds of the tongue and the ears, respectively. The olfactory cortex is located in the temporal lobes and receives sensory input from the nose.
16.2.5 Wernicke’s Area
Wernicke’s area, located in the language-dominant temporal-parietal lobe, is the receptive language area that is responsible for processing spoken and written language. Damage to this area results in impaired language comprehension and receptive aphasia. Preoperative testing before neurosurgical resection of a tumor in this area involves functional magnetic resonance imaging (fMRI) to identify this area in the individual patient (see the areas colored in green in ◘ Fig. 16.2 which are actual functional brain regions from a patient).
16.3 Motor Cortices
The motor cortices receive input from the sensory cortices and their respective association areas to allow for planning and execution of purposeful movement (◘ Fig. 16.2).
16.3.1 Primary Motor and Premotor Cortices
The primary motor and premotor cortices are located in the precentral gyrus and areas anteriorly adjacent in the frontal lobe. The corticospinal tracts originate in the primary motor cortex and travel caudally through the brainstem (decussate in the caudal medulla) and spinal cord to innervate skeletal muscle on the contralateral side. Stimulation of the primary motor cortex results in execution of a specific movement. Most of the synapses from the premotor cortex interface with the primary motor cortex to coordinate more complex movements. Stimulation of the premotor cortex results in a plan for a movement that can only be executed when the signal travels to the primary motor cortex.
16.3.2 Frontal Eye Fields
Projections travel from the visual association cortex in the occipital lobes to the frontal eye fields to produce eye movements. The frontal eye fields are on the lateral superior frontal lobes and drive the eyes (ie, gaze) to the opposite side. Frontal lobe seizures classically cause gaze deviation to the opposite side along with clonic motor activity on the opposite side of the body.
16.3.3 Broca’s Area
Broca’s area is the expressive language area in the language-dominant frontal lobe. Damage to this area causes an expressive aphasia with difficulty speaking or writing.
The cortical areas serving motor or language function are termed eloquent cortex. Tumors or lesions in these areas often have to be removed during awake cranial surgery in order to continuously test the patient during resection of the lesion.
16.4 Association Areas
There are multiple association areas that allow for higher level cognition such as memory, judgment, emotion, language; with coordination of sensori-motor data based on these interpretations. Perhaps the most important association area is the prefrontal cortex. This cortex plays a critical role in executive control of goal-directed behavior, attention, and personality.
16.4.1 Subcortical Areas
The subcortical areas are the parts of the brain beneath and deep to the cortex. These areas include the basal ganglia, hippocampus, internal capsule (which includes the descending corticospinal tracts), cerebellum, brain stem, and the reticular activating system (◘ Fig. 16.3).
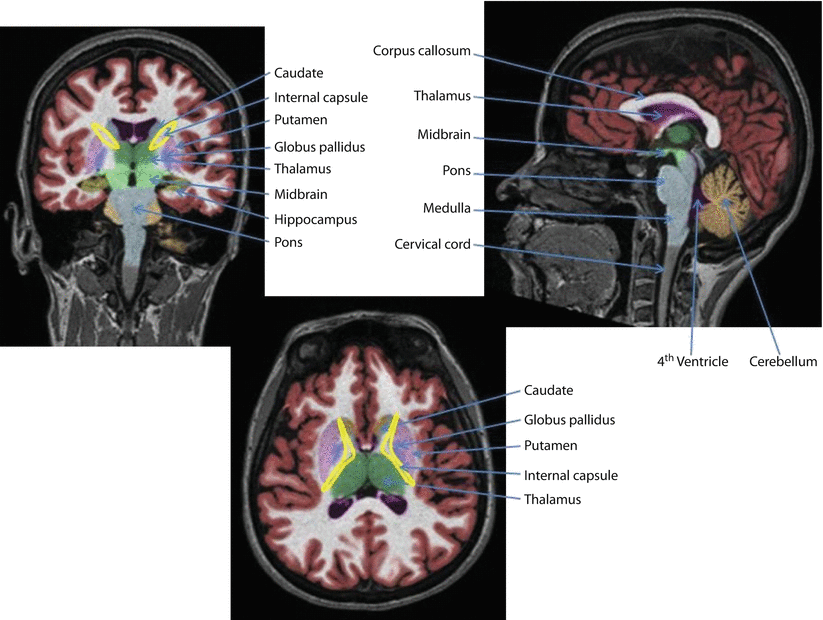

Fig. 16.3
Subcortical structures
16.5 Basal Ganglia
The basal ganglia are a group of subcortical nuclei that regulate movement and posture by integrating motor processes associated with the cerebral cortex. The main components of the basal ganglia include the caudate nucleus, putamen, globus pallidus, subthalamic nucleus, and substantia nigra. The basal ganglia receive input from the cerebral cortex and send neuronal output to the thalamus. From the thalamus, the neurons travel to the frontal cortex, brainstem, and spinal cord to regulate movement. Damage to the basal ganglia results in dyskinesias and movement disorders, such as Parkinson’s and Huntington’s diseases.
Dopaminergic pathways are integral to the regulation of movement. Anti-dopaminergic therapies given in the perioperative period—such as metoclopramide, promethazine, and droperidol—can have dystonic side effects or exacerbate dopamine-deficient conditions such as Parkinson’s disease.
16.6 Hippocampus
Located in the temporal lobe, the hippocampus regulates short-term memory. A neuronal circuit connects the hippocampus and the rest of the limbic system, including the amygdala, to the hypothalamus and autonomic nervous system for cortical control of emotional behavior.
Lesions of the hippocampus and anterior temporal lobe (such as mesial temporal sclerosis) are particularly high risk for causing seizures.
16.7 Internal Capsule
The internal capsule contains the input and output pathways between the cerebral cortex and the rest of the central nervous system (CNS). These include the sensory pathways that run from the thalamus to the cortex and the motor pathways that run from the primary motor cortex to the brainstem and spinal cord.
Patients with old strokes causing hemiparesis have damage to the corticospinal tracts. These patients are prone to hyperkalemia with depolarizing neuromuscular blockade (succinylcholine). When monitoring twitch count intraoperatively to titrate neuromuscular blockers, the monitor should be placed on the normal (non-paretic) limb as the paretic limb tends to be more resistant to non-depolarizing neuromuscular blockers (and leads you to give excessive doses).
16.8 Cerebellum
The cerebellum is located in the posterior cranial fossa (inferior to the occipital lobe of the cortex and dorsal to the brainstem). It is separated from the cortex by the tentorium cerebelli. It plays a vital role in motor coordination, posture, and balance. Functionally, the cerebellum can be divided into three components: cerebrocerebellum, vestibulocerebellum, and spinocerebellum.
The cerebrocerebellum includes the lateral aspects of the cerebellum that receive input from the cerebral cortex. It is involved in the planning of complex motor actions. The vestibulocerebellum includes the flocculonodular lobe of the cerebellum, and it receives input from the vestibular system. Its purpose is to control equilibrium. The spinocerebellum includes the vermis and intermediate hemispheres. The vermis receives somatosensory input from the head and proximal parts of the body to maintain posture, muscle tone, and control of the proximal muscles of the body and limbs. The intermediate parts of the hemispheres receive somatosensory input from the limbs to control the distal muscles of the limbs.
Note that damage to the cerebellum results in movement and balance problems ipsilateral to the site of the lesion. A cerebellar hematoma or edema can impose mass effect on the brainstem due to its close proximity to this structure. Moreover, cerebellar edema may compress the 4th ventricle, causing rapidly fatal obstructive hydrocephalus unless relieved with external ventricular drainage (catheter placed into the lateral ventricle through the skull and frontal lobe).
16.9 Brain Stem
The brain stem is divided into the midbrain, pons, and medulla and is responsible for controlling some of the most critical functions in the body (◘ Fig. 16.3). The brainstem maintains consciousness through the reticular activating system (discussed later). Moreover, the brainstem contains respiratory control centers (located in the medulla and pons) that are responsible for generating a respiratory rhythm and processing signals coming from the peripheral and central chemoreceptors. Damage to this section of the brainstem can lead to respiratory arrest.
The medulla contains the origin of the body’s parasympathetic nervous system. The dorsal vagal nucleus and nucleus ambiguous are the cell bodies of the vagus nerve (cranial nerve X) that contributes parasympathetic innervation to much of the body. Sympathetic pathways that begin in the diencephalon (hypothalamus) travel through the lateral medulla to the spinal cord. Through the autonomic nervous system, the medulla is vital in controlling the functions critical to life, including maintenance of heart rate, respiratory rate, and blood pressure. The specific sensory, motor, and autonomic pathways that course through the brainstem are described later in this chapter in the Spinal Cord Tracts section.
16.10 Cranial Nerves
Cranial nerves I and II arise from the cerebrum and cranial nerves III-XII exit the brain stem. The cranial nerves control functions critical for survival and contain the sensory, motor, and autonomic pathways that travel between the cerebral cortex, cerebellum, and the rest of the body (◘ Table 16.1).
Table 16.1
Cranial nerves
Cranial nerve | Origin | Function |
|---|---|---|
CN I (Olfactory) | Cerebrum | Smell |
CN II (Optic) | Cerebrum | Vision |
CN III (Oculomotor) | Midbrain | Motor to eye and eyelid; Pupil constriction |
CN IV (Trochlear) | Midbrain | Motor to eye |
CN V (Trigeminal) | Pons | Sensation of touch, taste; Motor to lower jaw |
CN VI (Abducens) | Pons | Motor to eye |
CN VII (Facial) | Pons/Pontomedullary junction | Sensation of taste; motor to muscles of facial expression; tearing; salivation |
CN VIII (Vestibulocochlear) | Pontomedullary junction/Medulla | Hearing, balance, and equilibrium |
CN IX (Glossopharyngeal) | Medulla | Sensation of taste, salivation; Motor to swallowing muscles |
CN X (Vagus) | Medulla | Sensation of taste; sensory, motor, and autonomic functions of viscera (gastric secretion and movement, cardiac reflex, respiratory reflex) |
CN XI (Accessory) | Medulla | Motor to trapezius and sternocleidomastoid |
CN XII (Hypoglossal) | Medulla | Motor control of tongue |
16.11 Reticular Activating System
The reticular activating system (RAS) is a complex group of nuclei in the brainstem that regulates arousal, consciousness, and sleep. The RAS projects to the cortex directly as well as indirectly through the thalamus. The neurotransmitters involved in these processes are predominantly serotonin and norepinephrine. Stimuli come from somatosensory, auditory, visual, and visceral sensory systems.
Damage to the RAS can lead to a comatose state. It is important to understand that in order to produce a coma, there must be bi-hemispheric dysfunction (such as from sedative agents, anoxic injury, hypoglycemia, etc.); or damage to the RAS in the upper brainstem (upper pons and midbrain) or diencephalon (hypothalamus or thalamus); such as from acute basilar artery occlusion.
16.11.1 Cerebral Blood Flow
Cerebral blood flow (CBF) varies in different parts of the brain depending on regional differences in metabolic activity because of a coupling mechanism between flow and metabolism. There is an intrinsic capability of the brain to deliver more blood flow to regions that have increased metabolic activity to match the heightened demand. Overall, the total CBF average is ~50 mL/100 g brain tissue/minute. CBF comprises approximately 15–20% of the cardiac output.
Decreased CBF rates are associated with cerebral impairment (20–25 mL/100 g brain tissue/min), isoelectric electroencephalogram (EEG) (15–20 mL/100 g brain tissue/min), and irreversible brain damage (CBF <10 mL/100 g brain tissue/min).
CBF is affected by the following parameters: cerebral metabolic rate, cerebral perfusion pressure, CO2 reactivity (PaCO2), O2 reactivity (PaO2), autoregulation, temperature, and anesthetic medications. Cerebral oximetry and EEG monitored over the frontal lobes (forehead; ◘ Fig. 16.4) are used as surrogates for CBF, which cannot be measured directly in the operating room with current technology. EEG is also processed to derive depth of anesthesia data.The electroencephalogram (EEG) detects brain electrical activity recorded at the scalp, while electrocorticography (eCOG; used for example in epilepsy surgery) measures brain electrical activity recorded directly from the surface of the brain. ◘ Figures 16.5, 16.6, 16.7, 16.8, 16.9, and 16.10 show various examples of EEG and eCOG. Cerebral ischemia and anesthetics both cause slowing of the EEG and must be interpreted in the context of the clinical situation.
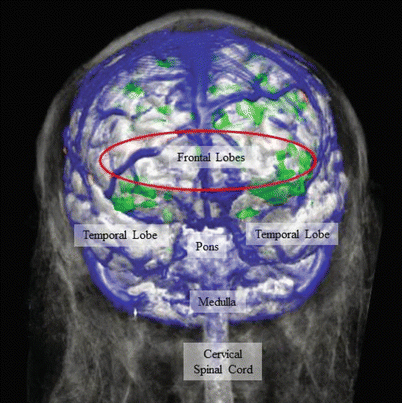
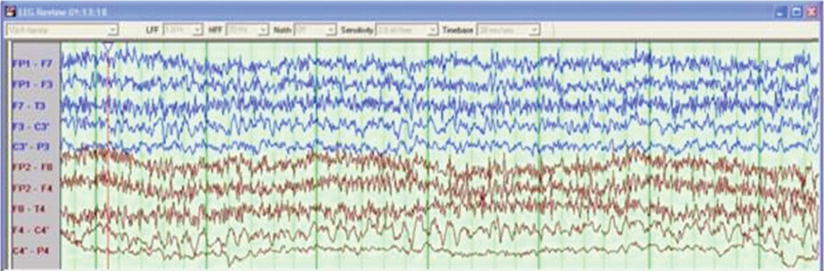
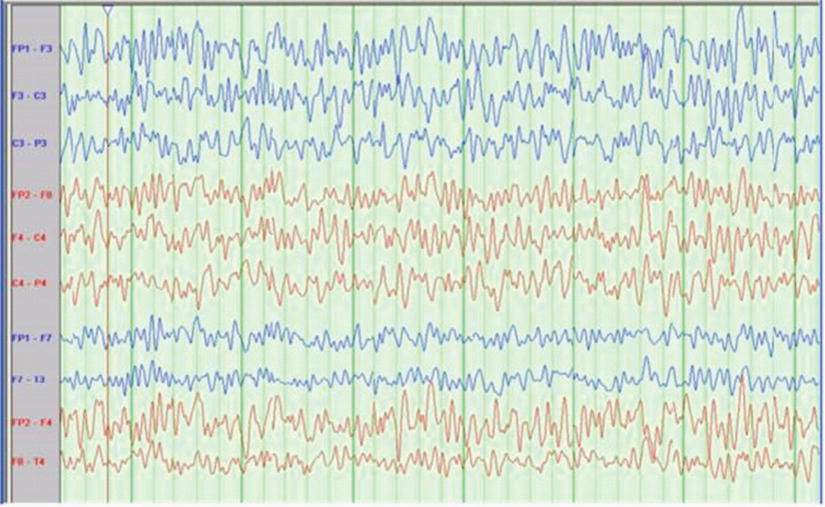
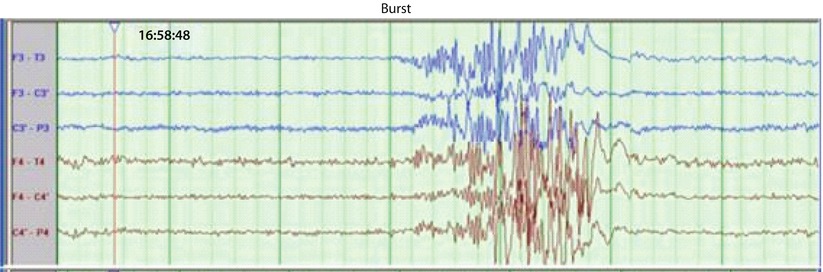
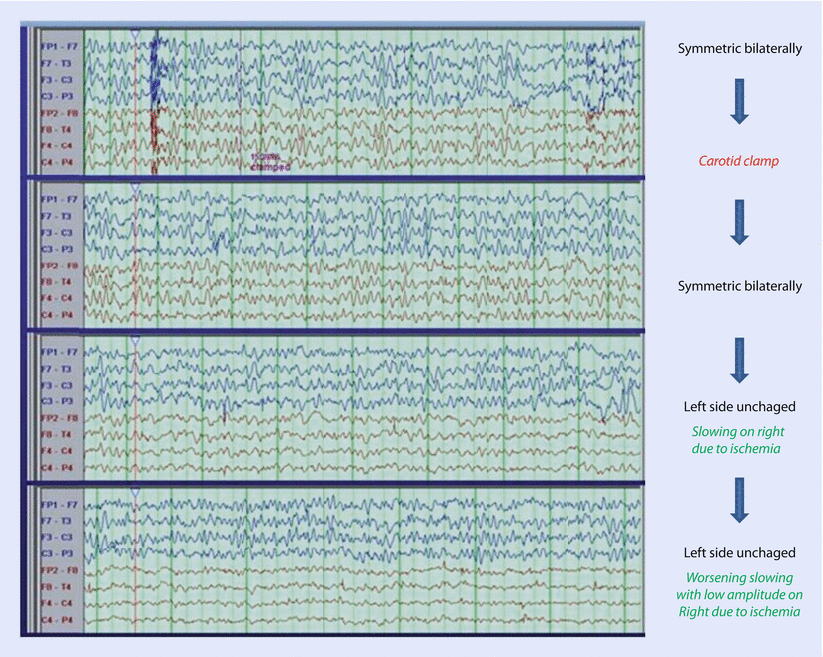
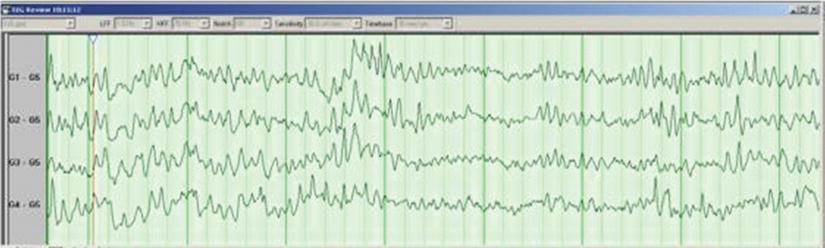
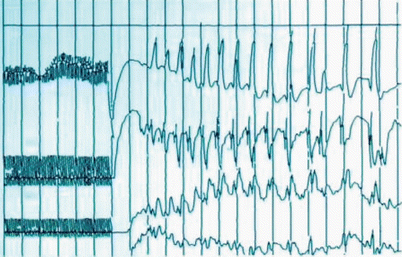

Fig. 16.4
Red ellipse notes the area covered by processed electroencephalogram (EEG) and cerebral oximetry monitors. The major cerebral veins and sinuses appear blue in the this fMRI image. Green areas represent areas of increased blood flow (ie, increased metabolic activity causing coupled increases in blood flow) during functional MRI testing

Fig. 16.5
Scalp electroencephalography (EEG) obscured by noise (usually due to muscle/EMG or electrocautery) (Courtesy of Biotronic Neuronetworks)

Fig. 16.6
Scalp electroencephalography (EEG) – normal (Courtesy of Biotronic Neuronetworks)

Fig. 16.7
Scalp electroencephalography (EEG) showing 6 channels (3 left and 3 right). Burst suppression pattern is seen (Courtesy of Biotronic Neuronetworks)

Fig. 16.8
Scalp electroencephalography (EEG) at 4 time periods following R carotid occlusion during endarterectomy. EEG from the left is in purple (1st 4 channels) and EEG from the right is in red (2nd 4 channels) (Courtesy of Biotronic Neuronetworks)

Fig. 16.9
Electrocorticography (ECoG) is EEG recorded directly from the surface of the brain. Here you see 4 channels showing normal brain waves (Courtesy of Biotronic Neuronetworks)

Fig. 16.10
Electrocorticography (ECoG). Here you see 4 channels with initial stimulation artifact followed by “after discharges” (electrically induced seizures) (Courtesy of Biotronic Neuronetworks)
16.12 Effects of Perfusion Pressure, pH, PaCO2, PaO2, Cerebral Metabolic Rate for O2
16.12.1 Perfusion Pressure
CBF is equal to the cerebral perfusion pressure (CPP) divided by the cerebral vascular resistance (CVR). CPP is the mean arterial pressure (MAP) minus the intracranial pressure (ICP), or central venous pressure (CVP), whichever is greater (◘ Fig. 16.11; ◘ Box 16.1). Normal CPP is 80-100 mm Hg and is primarily dependent on MAP given the generally low values of ICP and CVP. Normal adult ICP values range from 0 to 15 mm Hg; ICP is zero or negative in the normal patient in a sitting or standing position. Generally, 20 mm Hg is the threshold for treating pathologically elevated ICP.
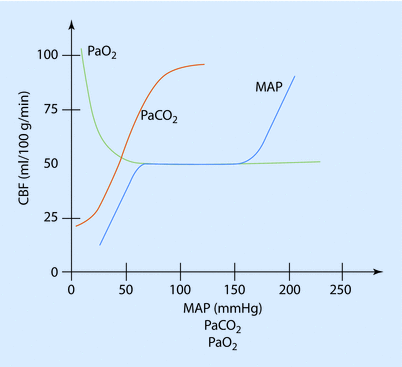

Fig. 16.11
The relationship betwe en CBF, PaO2, PaCO2, and MAP
CPP below the lower limit of cerebral autoregulation can lead to cerebral ischemia. This is generally thought to be <60 mm Hg, whether due to decreased MAP or increased ICP. However, it is essential to remember that there is considerable inter-individual variability and that blood pressure goals intraoperatively should be chosen after consideration of the patient’s baseline blood pressure. For patients with pathologically elevated ICP, CPP should generally be kept ≥60 mm Hg. Within normotensive blood pressure variations, CBF is maintained due to autoregulation of the CVR.
◘ Box 16.1
CBF = CPP/CVR
CPP = MAP−ICP (or CVP)
CBF: cerebral blood flow
CPP: cerebral perfusion pressure
CVR: cerebral vascular resistance
MAP: mean arterial pressure
ICP: intracranial pressure
CVP: central venous pressure
16.12.2 PaCO2 and pH
PaCO2 is the most significant extrinsic factor affecting CBF (◘ Fig. 16.11). Changes in pH and PaCO2 can quickly alter CBF by changing CVR: Hypercapnia/acidosis increases CBF and hypocapnia/alkalosis decreases CBF. CBF changes 1–2 ml/100 g brain tissue/min per mm Hg PaCO2 change between PaCO2 20–80 mm Hg. Hyperventilation is a very effective measure to help reduce cerebral blood volume in a patient with elevated ICP. However, excessive hyperventilation can lead to critically low CBF and cause cerebral ischemia. The effect of hyperventilation wanes with time so it is best used as an acute rescue modality for patients with elevated ICP. In operative neurosurgery, hyperventilation is used to control brain bulk (target PaCO2 ~ 30 mm Hg) as needed and normocapnia is restored subsequently.
During acute metabolic acidosis, the increased hydrogen ions (H+) cannot readily cross the blood-brain barrier (BBB), so it does not acutely affect CSF pH. However, with acute respiratory acidosis, an increase in PaCO2 can quickly impact the CSF pH since the CO2 can readily diffuse across the BBB. CSF bicarbonate takes up to 6 h to equilibrate across the blood-brain barrier to compensate for the pH change from the PaCO2 changes. Once CSF bicarbonate equilibrates, the CSF pH normalizes and CBF returns to the previous rate (ie, the effect of hyperventilation wanes).
16.12.3 PaO2
Extremes in PaO2 can alter CBF (◘ Fig. 16.11). Hypoxemia (PaO2 <50 mm Hg) significantly increases CBF, however hyperoxia (PaO2 >300 mm Hg) only slightly decreases CBF. The neurohumoral mechanisms for these effects are not well understood.
16.12.4 Cerebral Metabolic Rate for O2
The concept of flow-metabolism coupling in the brain reflects the relationship between CBF and the cerebral metabolic rate for O2 (CMRO2) (◘ Fig. 16.11). The average CMRO2 is 3–3.5 ml O2/100 g brain/min. Elevated regional brain activity leads to local increases in CMR that increase regional cerebral blood flow. CMR decreases during sleep, comatose states, and hypothermia; CMR increases with seizure activity and hyperthermia. Hypothermia reduces CMR and decreases CBF by ~7% for every 1 °C reduction in body temperature. The effects of anesthetic agents on CMR and CBF are discussed later.
16.13 Cerebral Steal and Inverse Steal Phenomena
The cerebral steal phenomenon takes place when vasodilation occurs in normal regions of the brain, shunting blood away from pathologic regions with impaired vasoreactivity that are already vulnerable to ischemia (ie, the reverse Robin Hood effect). This can be relevant with hypercapnia and/or high concentrations (>1 MAC) of volatile anesthetics. Inverse steal, also known as the Robin Hood effect, is the phenomenon where cerebral perfusion to pathologic regions increases. Hypocapnia can induce vasoconstriction in normal tissue thereby increasing blood flow to the otherwise ischemic pathologic regions. The clinical relevance of both phenomena are unclear due to the inability to continuously measure regional cerebral blood flow.
16.14 Gray versus White Matter
While the average CBF is ~50 ml/100 g brain tissue/min for the entire brain, gray matter has 4 times more CMR and CBF than white matter. Due to the inherent coupling mechanism between flow and metabolism, the CBF in the cortical gray matter is about 80 ml/100 g brain tissue/min while the CBF in the subcortical white matter is about 20 ml/100 g brain tissue/min (◘ Box 16.2).
◘ Box 16.2
Average CBF = 50 ml/100 g brain tissue/min
Gray matter CBF = 80 ml/100 g brain tissue/min
White matter CBF = 20 ml/100 g brain tissue/min
16.15 Autoregulation
16.15.1 Normal
Several organ systems in the body have autoregulatory capabilities, such as the brain, heart, kidneys, and spinal cord. This is an inherent, protective mechanism where an organ can adjust its vascular resistance to maintain a normal blood flow despite changing perfusion pressure. However, this only takes place when in a specific blood pressure range (◘ Fig. 16.12). For instance, in a normal brain, autoregulation takes place when within certain blood pressure (BP) parameters (for example, MAP 60–150 mm Hg). While in this BP range, the CVR will automatically adjust, by vasodilating or vasoconstricting cerebral arterioles, in order to maintain a consistent CBF. The BP range for autoregulation has classically been designated as being between 50 and 150 mm Hg; however, this BP range in normal patients is now being challenged with consideration being given to raising the average lower limit of autoregulation to a higher value (60–80 mm Hg).
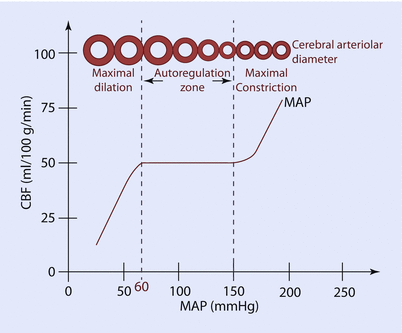

Fig. 16.12




Cerebreal blood flow (CBF) autoregulation normally occurs between mean arterial pressure (MAP) ~60–150 mm Hg. Within these blood pressure limits, the cerebral vascular resistance will automatically adjust by vasodilating or vasoconstricting cerebral arterioles in order to maintain a consistent CBF. Cerebral vascular resistance is maximally dilated below the lower limit of the CBF autoregulation and maximally constricted above the upper limit of CBF autoregulation
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






