FIGURE 119.1 The rostrocaudal progression of ischemia contemporarily known as coning.
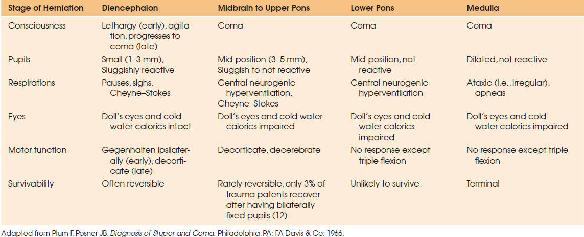
In concert with the previously described prebrain death physiology associated with severe brain injury, the process of brain death results in significant pathophysiologic changes in all organ systems with the cardiovascular system being most impacted. The rostrocaudal progression of ischemia, contemporarily known as coning is illustrated in Figure 119.1. Ischemia at the cerebral level produces vagal activation associated with a decreased heart rate, decreased cardiac output (CO), and decreased blood pressure. Although underappreciated, the first signs of incipient central herniation may simply be bradycardia in a severely brain-injured patient. Ischemia at the pons level produces the mixed vagal and sympathetic stimulation known as the Cushing response characterized by bradycardia and hypertension associated with irregular breathing. Further progression of the coning process to involve ischemia of the medulla oblongata is associated with a sympathetic stimulation termed the autonomic storm; Table 119.1 details the progression of central herniation. During this period, dramatic increases in catecholamines are reported with significant tachycardia and elevations in blood pressure. This represents the severely brain-injured patient’s attempt to maintain cerebral perfusion pressure (CPP) gradients in the face of elevated increased intracranial pressure (ICP) and evolving herniation. During this period, there is ischemic destruction of the hypothalamic pituitary axis resulting in thermoregulatory impairment and purported endocrine dysfunction. Further progression of ischemia results in spinal cord destruction with herniation and sympathetic deactivation characterized by bradycardia, vasodilatation, and a low CO state. Somatic death after clinical brain death will inevitably occur in the absence of aggressive support. In an era when brain death was not accepted, prolonged survivorship, with a mean duration of 23 days, was noted in a study that aggressively maintained brain-dead patients (10). Autopsy studies of patients that were declared brain dead revealed histopathologic evidence of necrosis and liquefaction (11).
| TABLE 119.1 Central Herniation |
 |
The catecholamine surge or autonomic storm produces multiple ECG and hemodynamic abnormalities along with biochemical and histologic changes in the cardiac system. In a series of sentinel observations and experiments, Novitzky (13–18) initially defined the cardiovascular pathophysiology associated with brain death. Catecholamines induce a sudden increase in cytosolic calcium, which jeopardizes ATP production and activates lipases, proteases, and endonucleases. Xanthine oxidase activation reportedly produces free radicals, further impairing organ function. Histopathologic changes reported in experimental animals reveal various degrees of focal myocyte necrosis located predominantly in the subendocardial area consisting of contraction bands and myocytolysis with mononuclear cell infiltrates precipitating edema proximate to the necrotic areas. Contraction bands were observed in the smooth muscle of coronary arteries, and electron microscopy revealed a hypercontractile state of the sarcomere visualizing mitochondrial deposition of electron-dense material and secondary lysosome containing injured mitochondrial. The loss of ATP production jeopardizes myocardial energy stores and mediates the transition from the aerobic to anaerobic metabolism compromising myocardial function.
Animal data and observations from human series have defined multiple abnormalities related to the catecholamine surge and brain death including impaired coronary endothelial dysfunction (19), selective expression of inflammatory molecules (20), downregulation of myocardial contractility (21), abnormalities in loading conditions and impaired coronary perfusion (22), abnormalities of left ventricular myocardial gene expression (23), and changes in myocardial beta-adrenergic receptor function and high-energy phosphates along with beta-adrenergic receptor deregulation (24,25). From animal models, it appears that a sudden rise in ICP is more provocative of the hyperdynamic–hemodynamic response, with significantly higher catecholamine levels, and is associated with greater histopathologic damage. A more gradual increase in ICP resulting in brain death is associated with a milder hyperdynamic response, less catecholamine release, and milder ischemic changes in the myocardium (26). Clinically, this has been correlated with the development of cardiac allograft vasculopathy in the recipient. The coronary artery vasoconstriction, subendocardial ischemia, and focal myocardial necrosis associated with the autonomic storm have been reported to be associated with a high incidence of intimal thickening of the transplanted heart coronary arteries, myocardial infarction, and the need for subsequent revascularization surgery (27).
The hemodynamic abnormalities and their impact are illustrated in a study comparing postbrain death cardiac function in a group of potential organ donors whose autonomic storm was attenuated with donors whose autonomic system storm was untreated. Using a definition of autonomic storm characterized by a systolic blood pressure ≥200 mmHg and tachycardia with heart rates >140 beats per minute, the authors treated this hemodynamic response in the study group, which was observed in 63% of donors for a mean duration of 1.2 hours, with beta-blockers. Treatment resulted in a significantly higher postbrain death left ventricular ejection fraction (LVEF) (63.9% vs. 49.0%), a higher rate of cardiac transplantation (91.7% vs. 41.2%), and better heart recipient survival at 2 months (100% vs. 43%). The investigators concluded that treatment of the autonomic storm enabled better cardiac function postbrain death, higher rates of cardiac transplantation, and better recipient outcomes (28). Recommendations regarding the treatment of the autonomic surge should be viewed with caution, as this physiologic compensatory mechanism represents the patient’s attempt to maintain cerebral perfusion in the face of herniation. Abolition of this response constitutes active intervention and donor management in patients who have not been declared brain dead which raises significant ethical concerns.
Globally, the intense systemic vasoconstriction of the autonomic storm compromises blood flow to various organs. Subsequently, with herniation/brain death and associated denervation with vasodilatation, there is reperfusion which forms the basis of the global IR injury, thought to contribute significantly to organ dysfunction in the donor, and facilitate the development of an immunologic continuum between the donor and recipient. In addition to the IR injury that occurs with the brain death process, IR may occur antecedent to the brain death event during resuscitation from the initial trauma, or may follow brain death during the periods of cold storage and transplantation. Ischemia precipitates the loss of aerobic oxidative metabolism, associated cellular energy loss along with changed ion gradients which promote calcium influx. With reperfusion of oxygen-rich blood, there is generation of oxygen radicals, lipid peroxidation, and further membrane permeability to calcium. IR activates the vascular endothelium and donor leukocytes with resultant cytokine expression. This precipitates local inflammation, which is thought to contribute to graft immunogenicity by producing major histocompatibility antigens and adhesion molecules.
In concert with the above, there is substantial animal and some human data to support that hypothalamic pituitary destruction produces an endocrinopathy of brain death which is additive to the above. Dominated by the thyroid and adrenal deficiencies, it is proposed that the absence of these key hormones contributes to cellular dysfunction, metabolic abnormalities, and hemodynamic deterioration. Deficiency of thyroid hormone is proposed to impair mitochondrial function and consequently diminishes energy substrate with the resultant transition from aerobic to anaerobic metabolism. Proponents of HRT propose that diminished cardiac contractility consequent to low thyroid hormone levels can be reversed with exogenous hormone supplementation. However, significant disparities exist related to hypothalamic–pituitary axis dysfunction when comparing animal and human studies. An abundance of animal data suggests that low-circulating thyroid hormone levels are responsible for abnormal energy sources, impaired cardiac function, and hemodynamic instability (15,16,29). Animal studies and some human reports suggest that there is a dramatic reversal of the anaerobic metabolism, improvement in cardiovascular stability, and normalization of laboratory parameters and EKG changes, as well as improved organ suitability for transplantation when the exogenous hormonal therapy is employed (15,16).
Nonetheless, several studies have failed to define the presence of endocrine dysfunction (30–32), show improvement with the addition of exogenous hormones (33,34), or correlate hemodynamic instability with hormone levels (31,32). Consequently, the use of HRT remains controversial; this is further discussed under cardiovascular management.
The impact of brain death upon graft function and transplanted organs was first recognized in the early 1980s. Cooper et al. (29) observed that hearts procured from healthy anesthetized baboons and stored for 48 hours, when subsequently transplanted functioned immediately with no evidence of cardiac dysfunction. However, hearts procured from brain-dead donors and stored in a similar fashion required several hours to achieve adequate function. These investigators recognized that the only difference between the two groups was brain death, and determined that the brain death process was a risk factor for poor outcomes after transplantation. These observations began to establish that the brain death process was not static, and that the graft not biologically inert. Tilney et al. (35,36) proposed the existence of an immunologic continuum between donors and recipients as a mechanism to understand the influence of brain death on recipient organ function. Utilizing this model, they hypothesize that IR events associated with brain death and pre/postbrain death events precipitate immunologic and nonimmunologic injuries that impact upon short- and long-term graft function. A major component of the immunologic continuum is the IR injury, proposed to initiate a significant inflammatory response, which triggers and amplifies the acute postimmunologic activity, impacting multiple organs and contributing to their dysfunction in the short and long terms.
It has been noted that increased plasma IL-6 levels in donors is associated with lower recipient hospital-free survival after cadaveric organ transplantation (37). Similarly, elevated plasma IL-6 levels in donors were associated with greater degrees of preload responsiveness that correlated with fewer organs transplanted (38). In cardiac donors, serum and myocardial levels of tumor necrosis factor-alpha (TNF-α) and IL-6 were elevated in all, but were more markedly elevated in the dysfunctional unused donor hearts (39). An intense inflammatory environment defined by elevated levels of IL-1, IL-6, TNF-α, C-reactive protein (CRP), and procalcitonin (PCT) has been reported in potential heart and lung donors. In this series, elevated PCT levels were correlated with worse cardiac function and, potentially, thought to attenuate any improvement in cardiac function gained by donor management (40). Similar elevations of inflammatory markers have been reported in liver transplantation. In a comparison study of hepatic tissue from brain-dead and living donors, investigators reported significant elevations in inflammatory cytokines in brain-dead, as compared to living, donors. Cellular infiltrates were appreciably increased in parallel to the cytokine levels; this correlated with elevated liver enzymes, bilirubin levels, and increased rates of rejection and primary graft nonfunction (41). Attenuation of the increased inflammatory response with methylprednisolone was shown to significantly decrease soluble interleukins and the inflammatory response, which significantly ameliorated IR injury in the posttransplant course, and was accompanied by a decreased incidence of acute rejection (42). In summary, there is appreciable evidence that brain death and the associated inflammatory response have a substantial impact upon the transplanted organs. Future strategies will likely seek to preserve not simply organs, but to attenuate the donor inflammatory response.
BRAIN DEATH DECLARATION
After the 1959 description of “Le coma depasse” by Mollarat and Goulon, the description and understanding of coma and death were forever changed (43). These authors presented 23 cases from their Paris hospital in which they described irreversible or “irretrievable coma.” This coma was associated with a lack of cognitive and vegetative functions, going beyond any coma previously described. This case series formed the basis of what is contemporarily recognized as brain death. The investigators noted the necessity of considering the circumstances of the injury, the role of the neurologic examination, the results of electroencephalography (EEG), and the consequence of brain death on other organs. They found that the majority of injuries to the brain were confined to trauma, SAH, meningitis, cerebral venous thrombosis, massive stroke, and brain death after craniotomy for posterior fossa tumor. The investigators detailed problems including deterioration of pulmonary function, polyuria, hyperglycemia, and tachycardia. It is intriguing that this paper, even though published in a relatively well-known European journal, took more than 15 years before it became known in the United States and Great Britain.
Interestingly, Mollart and Goulan’s paper was not the first description of brain death (43). Lofstedt and von Reis (44) described six mechanically ventilated patients with absent reflexes, apnea, hypotension, hypothermia, and polyuria associated with absent angiographic cerebral blood flow; death was declared when cardiac arrest occurred, between 2 and 26 days after the clinical examination. In 1963, Schwab and associates (45) reported EEG as an adjunct for determining death when cardiac activity was present. These authors proposed the following criteria to determine death: (1) absence of spontaneous respiration for 30 minutes; (2) no tendon reflexes of any type; (3) no pupillary reflexes; (4) absence of the occulocardiac reflex; and (5) 30 minutes of an isoelectric EEG.
This corpus of research, and the recommendations contained therein, generated substantial controversy in the organ transplant community, as some were uncomfortable procuring organs for transplantation from donors that were pronounced dead using brain death criteria. Nonetheless, in 1968, Harvard Anesthesiologist Henry Beecher chaired a committee at Harvard Medical School which attempted to define irreversible coma as new criteria for death. The committee defined death as the irreversible loss of all brain functions and proposed the criteria necessary to make that determination (46). The Harvard criteria included nonreceptivity and unresponsiveness, no movements or breathing, no reflexes, and a flat EEG. The committee suggested that the tests be repeated at 24 hours and, in the absence of hypothermia and central nervous system depressants and with no change in examination, the patient would fulfill criteria for the diagnosis of brain death.
Subsequently, concern regarding the relevance of EEG unfolded and, the Conference of the Royal Colleges and Faculties of the United Kingdom published the Diagnosis of Brain Death first in 1976 and again in 1995, altering the definition from brain death to brain stem death (47). They determined that if the brain stem was dead, the brain was dead, and if the brain was dead, the patient was dead. The conference required that the etiology of the condition that led to coma be established, and a search for reversible factors be undertaken. Examples of “reversible factors” included central nervous system depressant drugs, neuromuscular blocking agents, respiratory depressants, and metabolic or endocrine disturbances. A period of observation was recommended and the technique for apnea testing was described (47,48).
The Quality Standards Subcommittee of the American Academy of Neurology formally redefined brain death in 1993, utilizing an evidence-based approach. They defined criteria for evaluating brain death as the presence of coma and evidence for the cause of the coma, with the absence of the following confounding factors: hypothermia, intoxication, sedative drugs, neuromuscular blocking agents, severe electrolyte disturbances, severe acid–base abnormalities, endocrine crises. Fulfilling the preceding criteria, brainstem reflexes and motor responses needed to be absent, and a positive apnea test established the clinical diagnosis of brain death. An apnea test was finally established as a criterion and part of the examination to define brain death. The subcommittee recommended a repeat evaluation 6 hours after the initial evaluation, but recognized that the time was arbitrary and suggested that confirmatory studies should only be required when specific components of clinical testing could not be reliably evaluated (49). With the 2010 update, the subcommittee recognizes there is insufficient evidence for determination of the minimally acceptable time for an observation period (50).
The 1977 NIH-sponsored study (51) is the only prospective attempt to develop comprehensive guidelines for determination of brain death based on neurologic criteria. Enrollment required demonstration of cerebral unresponsiveness and apnea, and at least one isoelectric EEG. This group recommended examinations at least 6 hours after the onset of coma and apnea. The examination required demonstration of cerebral unresponsiveness, dilated pupils, absent brain stem reflexes, apnea, and an isoelectric EEG. The apnea examination, as defined in this study, only required that the patient not make any effort to breath over the ventilator. In the United States today, most institutional policies are modeled after the Quality Standards Subcommittee of the American Academy of Neurology (50).
Examination to Determine Brain Death
When the diagnosis of brain death is considered in the appropriate clinical context, a very careful physical examination must be performed. Brain death testing requires first, definitive evidence of an acute catastrophic event that involves both cerebral hemispheres or the brain stem in the appropriate clinical context so that irreversibility is assured; second, complicating medical conditions that could potentially compromise the clinical assessment must be ruled out. These include electrolyte, acid–base, and endocrine disturbances. There should be no evidence of drug intoxication, neuromuscular blocking agent use, poisoning, or any other agent that might compromise the clinical examination. Additionally, hypothermia needs to be corrected; ideally, the patient should have a core temperature between 35° and 38°C. Frequently, the computerized tomographic (CT) scan of the head will provide evidence for the magnitude of the brain injury. These injuries may include massive intraparenchymal hemorrhage or SAH, and/or epidural or subdural hemorrhage with mass effect. The CT scan may also appear slightly less dramatic after a cardiac arrest. Findings may be limited to the loss of sulci and the gray matter–white mater differentiation, effacement of the basilar cisterns, all of which reflect cerebral edema.
The patient must exhibit lack of consciousness. Determining unresponsiveness usually implies the administration of some painful stimuli. While there are multiple approaches—sternal rubbing, rubbing knuckles on ribs, twisting nipples, and pin prick—we consider these to be somewhat abusive. Perhaps more appropriate, although not accepted as the standard, is to utilize an instrument such as a pen, pencil, or the tip of a Kelly clamp to apply pressure at the lunula (junction of the cuticle and skin of the digit). This pressure will consistently elicit a response in patients with an intact nervous system and it is not construed as potentially “violent” as pin prick or nipple twisting. Furthermore, it does not leave bruising that nipple twisting does and will not cause skin abrasions in the fragile skin of the elderly.
When painful stimulation is applied, there should be no responses such as eye opening or withdrawal and grimacing, although there may be an occasional “spinal” reflex with this stimulus; this spinal reflex is neither reproducible nor purposeful. Spinal movements have been described by Wijdicks (52) as brief, slow movements in upper limbs, flexion of the finger, and arm lifting that is not a decerebrate or decorticate response; these movements are not persistent and usually not reproducible. The precise reflex pathway(s) is not understood; however, these are recognized as spinal reflexes. Moreover, triple flexion (flexion of foot, knee, and hip) should be carefully considered whether it is withdrawal, or just represents a spinal reflex. In the examination for brain death, there should be no uncertainty, so triple flexion can lead to diagnostic uncertainty.
Each patient needs to be examined as prescribed by institutional and state standards which vary. Please be aware of your local standards. We give an accurate general description, but make no attempt to include local standards.
Brain Stem Reflexes
Pupillary Response
The pupillary response to light should be absent in both eyes. The pupils in brain-dead patients are most often dilated, mid-position, usually 4 to 6 mm. It is important to ensure that there is no pre-existing ocular abnormalities and that topical ocular agents have not been instilled. Wijdicks (52) suggests that neuromuscular blocking agents may cause a nonreactive light reflex, although in our experience this is quite rare. The cranial nerves (CrNs) evaluated in the pupil light response are CrN II and III.
Ocular Testing
In the presence of brain death, there should be no ocular movements either to brisk movement of the head from side to side (absence of dolls’ eyes) nor to instillation of cold water into the auditory canals. The nerves stimulated by these maneuvers include CrN VIII (efferent) with CrN III and VI (afferents). Prior to stimulating the oculocephalic reflex, one must ensure that the cervical spine is intact and the test should not be performed when there is known or suspected cervical spine injury. With the head in neutral position, the head is briskly moved, first to the left and held there for approximately 30 seconds; if the CrNs are intact, the eyes will move from the direct frontal gaze, to the left and then back toward the previous midline focus. The same is true when head is moved to the right, if the nerves are intact, the eyes will move from the direct frontal gaze to the right, and then back to the previous midline frontal gaze. In the presence of brain death, the eyes will remain in the direction the head is moved.
Even though cold water calorics test the same nerves, they also need to be tested on both sides. Prior to the instillation of iced saline into the auditory canal, one must ensure that the tympanic membranes are intact and that there is no occlusion of the auditory canal. Approximately 50 mL of iced saline is instilled into the auditory cannel. The cold stimulus results in sedimentation of the endolymph and stimulation of hair cells in the vestibular apparatus; the response in a comatose patient with an intact neurologic system is a slow deviation of the eyes toward the cold stimulus. In the presence of brain death, the eyes stay fixed in midline position. Drugs such as aminoglycosides, tricyclic antidepressants, anticholinergic agents, any antiepileptic drug, and some chemotherapeutic agents may ablate or abolish this caloric response in the presence of an intact brain stem (52). Basilar fracture may abrogate the response unilaterally on the side of the fracture. Nurses appreciate placement of towels to capture the water which spills out. Cold water caloric testing is often performed immediately prior to apnea testing.
Corneal Reflexes
Corneal reflexes should be evaluated by carefully using a sterile cotton-tipped swab. Blinking requires an intact brain stem, but care must be taken so that the eyelashes are not stimulated. The CrNs involved are V (afferent) and VII (efferent). Blinking that occurs with stimulation of the cornea is not compatible with brain death. Severe facial and ocular trauma can compromise the interpretation of these findings.
Pharyngeal and Tracheal Reflexes
In the intact brainstem, pharyngeal and tracheal reflexes—cough, gag—may be stimulated by passing a catheter through the endotracheal tube into the trachea and suctioning for several seconds. The CrNs involved are IX and X; CrN IX is the afferent to the trachea and CrN X is the efferent from the brain stem back to the trachea. The presence of a cough reflex is not compatible with brain death; the gag response may be difficult to interpret and is unreliable in an intubated patient (52).
Apnea Study
The apnea study is usually the final portion of the clinical examination to determine brain death. In principle, the arterial partial pressure of CO2 (PaCO2) must rise to at least 60 or 20 mmHg greater than the patient’s baseline. This relatively rapid rise in PaCO2 results in a decrease in the cerebral spinal fluid pH, which is sensed by the medullary respiratory center. When the respiratory center is functional, respiratory efforts will result; in the presence of brain death, there will be no respiratory effort.
Initially, one ensures that the patient’s core temperature is between 35° and 38°C, preferably normothermic. The patient is denitrogenated (“pre-oxygenated”) and stabilized, ensuring correction of any hemodynamic or electrolyte abnormalities. This is especially true when the technique used for the apnea study is the removal of the patient from the ventilator with no continuous positive airway pressure (CPAP). Denitrogenation usually requires 10 minutes of breathing an FiO2 of 1.0. Prior to initiation of the procedure, an arterial blood gas (ABG) analysis must be obtained both to ensure adequate oxygenation and to define a baseline arterial CO2 value. With the baseline arterial CO2 value, one can calculate the apnea time required for the PaCO2 to rise to 60 mmHg.
The technique is as follows: The measured PaCO2 value from the ABG is subtracted from 60 mmHg (delta-CO2). It is recognized that PaCO2 will climb approximately 3 mmHg, in the first minute of apnea and thereafter, it will climb by approximately 2 mmHg/min. Therefore, dividing the delta-CO2 by the lower value of 2 mmHg increase per minute will ensure an adequate apnea time, allowing the PaCO2 to achieve the minimal value of 60 mmHg in the presence of brain death–associated apnea, or a 20 mmHg rise from baseline.
Once the time required to achieve the delta value is determined, there are three techniques that may be used for the apnea study. These include (1) simply removing the patient from mechanical ventilation, and placing a catheter through the ETT while insufflating O2 at approximate 6 L/min. This will most often ensure adequate apneic oxygenation. (2) Set the mechanical ventilator to spontaneous mode with no backup apneic mode. With this approach, the patient can be maintained on a low level of CPAP to preserve oxygenation. The monitoring modalities of the mechanical ventilator can be used to visualize respiratory efforts if these were to occur. (3) The patient may be taken off mechanical ventilation, and connected to a Mapleson D circuit with a Wright spirometer placed in line. With fresh O2 flow of 6 to 10 L/min, one can partially close the Mapleson D circuit pop-off valve, ensure that there is some CPAP, and then watch both the bag of the circuit and the Wright spirometer for respiratory efforts.
Whichever technique is used, the patient is kept off mechanical ventilation for the calculated time to achieve the delta CO2 value; pulse oximetric saturation is followed to ensure that desaturation does not occur. Desaturations, hemodynamic instability, or significant rhythm disturbances necessitate immediate replacement of full mechanical ventilation. Ideally, an ABG should be drawn at the onset of instability and used for assessment. A PaCO2 >60 mmHg is consistent with a failed apnea test. Failure to achieve a PCO2 ≥60 mmHg, in the absence of any respiratory efforts, suggests inadequate time for CO2 production to achieve threshold. In this case, the test may be reperformed after correcting metabolic/physiologic abnormalities or moving directly to a confirmatory study. At the end of the newly calculated time period, an ABG is obtained. If the PaCO2 value is ≥60 mmHg, or has increased >20 mmHg above the patients known baseline value, and there have been no respiratory efforts, the result is compatible with brain death.
After the second ABG is drawn, the patient is reconnected to the mechanical ventilator and, if the PaCO2 from the previous sample is ≥60 mmHg, the family is notified that the examination is consistent with brain death. With a “failed” apnea study, the patient is pronounced clinically brain dead.
Common complications of the apnea study are hypotension and cardiac dysrhythmias. If one is unable to adequately perform the apnea study because of these complications or because of hypoxia, confirmatory tests, such as a radionuclear cerebral blood flow study or a four-vessel angiogram, will be required. Taylor et al. (53), in a meta-analysis of CT angiography as a confirmatory study in the setting of clinical brain death, found that based upon the extant data, CTA had a sensitivity, after the clinical determination of brain death, of only 85%. They suggest that this study not be used as a confirmatory study as approximately 15% of patients clinically dead will not have this diagnosis confirmed; other studies—as above—are more appropriate. Finally, a single apnea examination will suffice (43) for the declaration of brain death; when a repeat clinical examination is performed, a repeat apnea study is not an absolute requirement, although it is imperative to ensure that institution and state regulations are followed.
EXCLUSIONS AND CONTRAINDICATIONS
Given the shortage of organs available for donation, exclusions and contraindications should be viewed as absolutely relative or relatively absolute (54). Consequently, all cases should be reviewed in conjunction with the OPO coordinator to determine suitability. Successful procurement has been undertaken in a broad array of cases that were previously deemed unsuitable including patients with sepsis and bacterial meningitis, provided appropriate anti-infective treatment is undertaken. However, organs should not be procured from potential donors when the etiology of the purported infection has not been determined. An evolving literature suggests that organ procurement from patients with known meningitis that has been appropriately treated has not resulted in significant transmission of the infectious agent nor organ compromise in the recipient (55). In a retrospective study performed over 10 years in 39 patients undergoing heart and lung transplantation, undertaken with organs from cadaveric donors with bacterial meningitis defined by either positive blood or cerebral spinal fluid cultures and associated clinical signs and symptoms, no contraindications could be defined because none of the recipients died of infection-related causes. Common organisms in the donor were reported to be Neisseria meningitides, 53.8%; Streptococcus pneumonia, 41%; and Haemophilus influenza, 5.2%. Importantly, adequate antibiotic therapy was initiated before organ retrieval and continued after transplantation (56). Similarly, Satoi (57) reported that liver transplantation from donors with bacterial meningitis was safe, provided the donor and recipient received adequate antimicrobial therapy. In this study of 34 recipients, there were no infectious complications caused by the meningeal pathogens. Although recommendations are difficult to establish, treatment of the donor for 24 to 48 hours and a minimum 7 to 10 days for the recipient appears to be adequate.
Frequently, potential organ donors in the ICU are bacteremic; similar to the above literature for donors with meningitis, procurement of organs from bacteremic patients has been successfully undertaken and the presence of bacteremia should not preclude donor evaluation. In a study that reviewed heart transplantation from donors that expired from community-acquired infections with severe septic shock, meningitis, and/or pneumonia, no evidence of donor-associated infection and sepsis or rejection was observed in the recipient (58). In a report of transplantation from bacteremic donors with gram-negative septic shock, all recipients were alive with good graft function 60 days following transplantation with no infectious complications. It is recommended that appropriate antibiotics be given for at least 48 hours prior to organ retrieval and that recipients receive 7 days of culture specific antibiotics posttransplantation (59).
Patients with human immunodeficiency virus (HIV) represent an absolute contraindication to donation. However, the occasional patient engaging in high-risk social behavior, but remaining HIV seronegative, may be considered for possible organ donation. In this circumstance, there should be an extensive review of the medical record, interviews with the family, and active communication with OPO. It is recommended, however, that information about the donor’s high-risk behavior be conveyed to the transplant center, which ought to inform the potential recipient of the risks and benefits of donation from this individual.
Donor malignancy represents another area of concern that warrants careful evaluation when a donor is considered for procurement. Any active noncentral nervous system malignancy is viewed as an absolute contraindication to donation. A previous history of choriocarcinoma, lung cancer, melanoma, and patients with previous colon, breast, or kidney cancer are, similarly, precluded. Donors with a previous history of nonmelanoma skin cancers and a select group of patients with cancer defined in situ or with very low-grade levels of malignancy may be considered as can be patients with a history of previous curative therapy; these cases should be discussed on an individual basis with the OPO and the transplant center. Central nervous system malignancies are not uncommon in the potential organ donor population. Given their rare metastasis and low incidence of development in the recipient, procurement is frequently undertaken. Potential donors with a low-grade tumor, absent craniotomy, and no ventricular shunts are better candidates than those donors with previously defined high-grade malignancy, craniotomy, and shunt placement.
CONSENT
Approaching and obtaining consent from the potential donor’s family is an absolute requirement for organ donation. In the case of previously defined, first-person consent, in which an individual firmly establishes their desire to donate via a driver’s license or donor card, it is imperative that the first-person consent be honored and recognized as the basis for consent. In 1998, the CMS established several parameters governing the organ donation process through the Federal Conditions of Participation. A change in the Conditions of Participation required timely notification of the OPO when death is imminent to ensure that families were provided the opportunity to discuss the option of organ and tissue donation. Similarly, the Conditions of Participation mandated that individuals specially trained in requesting, termed “designated requestors” be responsible for making the request, and required that all individuals discussing organ donation with families receive the appropriate training.∗ The intent of this mandate was to ensure that individuals approaching families and discussing organ donation were trained and sensitive to the family situation. Initially, this was interpreted by some to imply that physicians would be excluded from the request process and only OPO-designated requestors could approach the family. Subsequent discussion and policy recommendations adopted by the American Medical Association suggested that the designated requestor contacts the attending physician before organ donation requests and includes the attending physician in the discussion with the family. It is important to appreciate that OPO coordinators, physicians, and nurses may be defined as designated requestors, provided they undertake the appropriate training (60).
Family characteristics and the approach to the consent process have been shown to significantly impact upon the decision to donate. Nondonor families tend to be less satisfied with the quality of care, have a lesser degree of understanding of brain death, and remain under the impression that brain-dead patients could survive. These families felt that there was insufficient time and privacy during the request process and that the requestor was not sensitive to their needs. Alternatively, consenting families had a much clearer understanding of brain death and were more satisfied with the overall consent process and their decision-making (61). Siminoff (62) evaluated the roles of prerequest factors and decision process variables in the consent process in a large study of 11,555 deaths; of 741 potential donors and a family request rate of 80%, the final consent rate was 48%. Decisions were made early, with 55% of families making their decision during the initial request. Of those families with an initial favorable view of consent (58%), 81% went on to complete the consent with consent not obtained in 19%. In families who initially had an unfavorable view of the donation process (25%), consent was eventually obtained in 9% and no consent was sustained in the remaining 91%. In the 17% of families that were undecided during the initial donation request, 47% went on to consent and consent was not obtained in 53%. The initial response predicted a final donation decision in 70% of families. Prerequest factors that were associated with successful consent included patient characteristics of young, white males dying from trauma and family beliefs in donation, prior knowledge of organ donation, the presence of a donor card, explicit discussions, and a belief that the patient would have wished to donate and that the information provided was adequate and the health care provider was comfortable with questions. There was no association noted between family education and income levels, hospital environmental factors, health care practitioner–associated demographics or the health care practitioner’s attitude toward donation. Decision process variables that correlated with donation were the correct initial assessment by the health care provider, instances when the family raised the donation issue, conversations and time spent with the OPO coordinator, and clear, unambiguous discussions related to cost, funeral homes, and choices. Decision process variables that had a negative correlation with donation included perceptions that the health care provider was not caring, surprise by the family when the request was made, or feeling harassed and pressured to make a decision. No correlation was found with the overall satisfaction of care, the timing of the request, or the belief that the patient was alive after the declaration of brain death. Hazard ratios for factors that directly related to donation included prerequest characteristics (7.68), optimal request pattern with the health care provider being a nonphysician and the OPO coordinator (2.96), OPO-related factors (3.08), and the topics discussed (5.22).
The request for organ donation has undergone an evolutionary process from random or inconsistent requesting to the use of designated requestors to the use of an effective requestor to the currently recommended process of effective requesting. Key elements of the requesting process recommended by the Institute of Medicine (IOM) include a focus on the family and the continuation of compassionate care with an acknowledgement of the uniqueness of each family and avoiding scripted statements. A determination of the most appropriate requestor and the timing of the request should be individualized and done on a case-by-case basis. Families of patients with protracted ICU stays frequently develop close associations with specific physicians or nurses, and may be willing to accept discussions related to impending death and donation at times earlier in the course than families with an acute crisis. The IOM panel recommended that donation be discussed as an opportunity, utilizing language that emphasizes the benefit to the transplant recipient and the potential of healing for the donor family. Importantly, the panel recommended that excellent EOL care be continued for the family, independent of the donation decision (63).
Although there is some variability, it has generally been accepted that decoupling or separating the request for donation from the declaration of brain death notification be used as the model for requests (64,65). In this model, the notification of brain death is both temporally and geographically segregated from the organ donation request. This provides the opportunity for the family to process the notification of brain death before the request is made for consent, although it has been suggested that consent may occur after the family has accepted that further care is futile (66). In conjunction with the decoupling process, factors that have been associated with a successful consent rate include making the request in a private setting, and ensuring the engagement of the OPO Transplant Coordinator. When all three elements are present, the consent rate was reported to be 2.5 times greater than when none of the elements were present (64). The Council on Scientific Affairs for the American Medical Association recommended that the process focuses upon supporting the family of all potential donors, be consistent with quality EOL principles, decouple discussions of brain death from the organ donation requests, ensure that the opportunity to donate is presented to all families, and do so in a private setting. Ensuring that the OPO Transplant Coordinator is involved and assists with coordinating the efforts in the ICU was strongly recommended; for those wishing to participate in the request process, special training should be undertaken, and certification as a designated requestor obtained.
MEDICAL MANAGEMENT
Hemodynamic and cardiovascular management form the cornerstone of potential organ donor management (ODM). A standardized and structured approach to hemodynamic management ensures that the donor somatically survives for procurement and maintains the remainder of potential organs in the best possible condition. Similar to the care of any critical patient, a collaborative approach utilizing the skills of physicians, nursing, respiratory therapists, and the OPO coordinator is pivotal for optimum management. Standardization of donor management from the referral process through consent, followed by management and recovery has been shown to significantly increase the number of organs recovered and organs transplanted. A 10.3% increase in organs recovered per 100 donors and a 3.3% increase in total organs transplanted per 100 donors was reported by Rosendale in a study that emphasized standardization of general medical management, eliminating variability in laboratory and diagnostic studies, along with standardization of respiratory therapy, and IV fluids and medications (67). The Surgical Trauma Group at the University of Southern California has been instrumental in pioneering the standardized approach to ODM. The development of an aggressive ODM program was reported to significantly increase the number or organs available for transplantation. Employing a critical care team that accepted potential organ donors for management utilizing pulmonary artery catheters (PAC), fluid resuscitation and use of vasopressors, prevention and treatment of complications associated with brain death, and liberal use of thyroid hormone in unstable donors, resulted in a 57% increase in total referrals, a 19% increase in potential donors, an 82% increase in the number of actual donors and an 87% decrease in the number of donors lost to hemodynamic instability. Overall, the implementation of this aggressive donor management team resulted in a 71% increase in the number of organs recovered (68). In a follow-up study by the same group utilizing an aggressive approach to ODM, the authors evaluated the impact of the complications of brain death upon organ retrieval. They hypothesized that brain death–related complications would have no significant impact on the number of organs donated, provided there was an aggressive ODM protocol in place. With complications defined as the requirement for vasoactive support which occurred in 97.1%, coagulopathy in 55.1%, thrombocytopenia in 53.6%, diabetes insipidus in 46.4%, cardiac ischemia in 30.4%, lactic acidosis in 24%, renal failure in 20.3%, and adult respiratory distress syndrome noted in 13%, there was no appreciable impact of the complications on the average number of organs procured. Additional benefits included a dramatic diminution in the number of donors lost to cardiovascular collapse, and improvement in conversion rates (69). In a comparison with other Level I trauma centers that did not utilize an aggressive donor management protocol, dramatic benefits were similarly reported which included a significant decrease in the incidence of cardiovascular collapse, and the number of organs procured per potential donor (69).
Although the traditional management of the potential organ donor has been taken by the OPO transplant coordinator, there has been an evolution toward a collaborative approach between the Intensivist/critical care community and the OPO, as reflected in the above-mentioned studies. Intensivist-led management of potential organ donors has been reported to increase the organs recovered for transplantation. In a study that evaluated the implementation of an Intensivist-led donor management program, the overall number of organs recovered for transplantation increased significantly (44% vs. 31%), which was largely reflective of an increase in the number of lungs procured and transplanted (24% vs. 11%). No appreciable change occurred in the number of hearts and livers recovered for transplantation. This study is reflective of the enormous impact that a collaborative and partnered approach between Intensivists and OPO coordinators can have upon donor management (70).
Although no clear current consensus exists, the traditional approach to ODM was to minimize the time between brain death and procurement because of the perception that prolonged management was detrimental to the donor organs and bed utilization in busy ICUs necessitated admission for salvageable patients. However, this concept has recently been challenged as evolving literature reports that a longer period of donor management may be beneficial (71). In a retrospective study with a mean time from brain death to procurement of 35 hours, it was reported that there was no decrease in the procurement to consented ratio with increasing time after brain death. When individual organs were analyzed separately, heart and pancreas procurement improved with an increased management period after brain death and some organs were successfully procured greater than 60 hours after brain death (72). Similarly, in a study of 100 consecutive organ donors whose mean donor management time was 23 hours, it was reported that donors managed in excess of 20 hours resulted in significantly more heart and lung procurements, more organs procured per donor (4.2 vs. 3.2) and more organs transplanted per donor (3.7 vs. 2.6). Interestingly, there was no significant difference noted in the obtainment of donor management goals (73).
Specific donor management goals during the donor management period have evolved as a standard. Attainment of these management goals has resulted in a significant increase in the number of organs procured and transplanted per donor. In an initial report by Hagan, consensus was developed for six specific donor management goals among six OPO organizations. The following management goals were derived: mean arterial pressure greater than 60 mmHg, CVP less than 10 mmHg (or serum osmolarity 285 to 295 mOsm/kg), sodium less than 155 mMol/L, pH 7.25 to 7.5, pressors (1 or none—1 pressor plus vasopressin for diabetes insipidus was deemed appropriate), and PaO2 greater than 300 mmHg while on 100% oxygen. These donor management goals were considered a bundle with compliance defined as achieving a minimum of five goals. The number of organs transplanted per donor was 4.87 in those meeting goals and 3.19 in those donors failing to meet the bundle goals for standard criteria donors. No significant change was noted for extended criteria donors (74). In a similar consensus-driven study, eight common goals were defined: mean airway pressure, CVP, pH, PaO2, sodium, glucose, vasopressor use, and urine output. Throughout the study period, there was a dramatic increase in the compliance with donor management goals, which was associated with a significant improvement in organs transplanted per donor. The authors reported that the success of transplantation was predominantly associated with limitations in vasopressor use and achieving adequate PaO2. Thoracic organs were most sensitive to the donor management goals as there was a dramatic increase in lung transplantation with higher levels of PaO2. Interestingly, mean arterial pressure, CVP, pH, sodium, and urine output had little effect on the transplantation rate. The authors concluded that goals and standardization of endpoints of donor management are associated with increased rates of transplantation. However, it was evident that not all standard goals are necessary with the most significant parameters being the low use of vasopressors and ensuring adequate oxygenation, which should form the focus of donor management (75). Similarly, in a study that evaluated 10 donor management goals and defined success as the achievement of eight goals, the authors used binary logistic regression to determine the independent predictors of more than four organs transplanted per donor. The authors reported that donors meeting donor management goals had more organs transplanted per donor (4.4 vs. 3.3). Independent predictors of transplanting more than four organs were age, serum creatinine, thyroid hormone, and meeting donor management goals. Among the individual donor management goals, odds ratios (ORs) were higher for CVP 4 to 10 mmHg (OR = 1.9), EF greater than 50% (OR = 4.0), PaO2/FiO2 greater than 300 (OR = 4.6), and a serum sodium 135 to 168 mEq/L (OR = 3.4) (75,76). The impact of a structured and standardized approach to ODM has similarly been reported to dramatically increase the retrieval rate of lungs and hearts for transplantation. In a study where potential lung donors were aggressively managed through protocol-guided optimization of ventilatory and hemodynamic strategies that consisted of measurements of extravascular lung water, bronchoscopy, and invasive monitoring, a dramatic increase in the rate of lung procurement was reported (40% vs. 27%) (77). Similarly, an aggressive and structured approach to the management of potential heart donors reported a significant increase in the numbers of hearts procured with a standardized approach using invasive monitoring and critical care management techniques (78). It appears that over the past decade, there has been an overall increase in available donors and organs, as well as a moderate increase in the mean number of organs per TBI donor. Additionally, the increased use of hormone replacement therapy (HRT) appears to be key to the successful conversion of marginal donors and enhanced recovery from certain subsets leading to decreased transplant wait times (79). For example, in the decade studied by Callahan and colleagues (79) using the Organ Procurement and Transplant Network’s (OPTN) organ donor and thoracic recipient dataset (from July 1, 2001 to June 30, 2012), the most common causes of donor death were cerebrovascular disease (31,804 cases, 42.9%) and TBI (28,142 cases, 37.9%). A slow but significant increase in the raw number of donors per year, from 5,857 in 2002 to 6,945 in 2012 (p < 0.001) was noted, as was an increase in the raw number of total organs procured, from 20,558 to 24,308 (p < 0.001). These increases coincide with the increased use of HRT in donor management, from 25.1% to 72.3% (p < 0.001); high-yield donors showed a similar increase in the use of HRT, from 33% in 2002 to 76.6% in 2012 (p < 0.001) (79).
Finally, recent data (80) suggest that elevated glucose is common in patients who donate after determination of neurologic death. Glucose levels above 180 mg/dL are associated with lower organ transplantation rates and worse graft outcomes. It is recommended that serum glucose levels be targeted to a glucose of 180 mg/dL or less; in our practice, we aim for 150 to 180 mg/dL.
Cardiovascular and Fluid Goals
An algorithmic approach to the cardiovascular and hemodynamic management of the potential organ donor is suggested (Fig. 119.2). As depicted in the figure, age plays a major role in the initial cardiac evaluation. Traditionally, cardiac catheterization has been required for potential organ donors over 40 years of age. Given the significant myocardial stress associated with brain death, echocardiography should not be performed immediately after brain death declaration as this may provide misleading information related to cardiac function. Initial attempts at stabilization should include normalizing blood pressure, metabolic abnormalities, and electrolyte disturbances. Transthoracic echocardiography (TTE) should be performed in all patients to evaluate structural abnormalities that may preclude cardiac donation and evaluate the LVEF. Since first reported in the evaluation of potential organ donors in 1988, TTE has proven invaluable for evaluation of cardiac function, particularly in circumstances where clinical events might have precluded cardiac utilization. In the original study, 29% of donor hearts that would have been previously excluded on clinical criteria were procured and successfully transplanted (81). Echocardiographic abnormalities are reported to be responsible for 26% of nonused hearts with an OR of 1.48 per 5% decrease in EF (82). Although instrumental in the evaluation of cardiac function in the potential organ donor, several issues warrant consideration regarding echocardiographic evaluation. These include the difficulty in securing the test, technical challenges with imaging in critically ill patients, and the accuracy and impact of the echocardiographic interpretation. Similarly, it is important to appreciate that LVEF is a load-dependent measure of contractility with variance noted when there are changes in preload and afterload (83). It is also important to recognize that temporal changes occur in left ventricular systolic function. In a study that evaluated sequential echocardiograms in potential organ donors, with EFs less than 50% or regional wall motion abnormalities on the initial cardiogram, 12 of 13 patients improved after donor management. Utilizing a strategy that employed high-dose corticosteroids and dopamine without the use of thyroid hormone, these 12 donor hearts were transplanted with a survival rate of 92% with an average follow-up of 16 months (84). In a series that evaluated clinical characteristics, echocardiographic and pathologic findings of myocardial dysfunction in potential donors, echocardiographic evidence of systolic dysfunction was appreciated in 42% of potential organ donors; this was not predicted by clinical findings, the EKG findings, or the type of neurologic injury. Histopathologically, there was a very limited correlation to the area of echocardiographic dysfunction in the histopathology of hearts that were not procured, suggesting, again, that brain death is associated with significant myocardial dysfunction and the potential for reversibility needs to be appreciated. Consequently, no heart should be rejected on the basis of a first initial abnormal echocardiogram. In instances where TTE evaluation is difficult, consideration should be given to the use of transesophageal echocardiography (TEE); limited literature comparing TTE with TEE suggests that the TTE assessment may be inadequate in almost one-third of the patients. A substantial increase in the number of abnormalities was detected with TEE, although no outcome difference was established (85). Recent literature suggests that more than 50% of the hearts with initial abnormal function may attain hemodynamic transplantation criteria with aggressive donor management. In this prospective study of 66 potential organ donors, an initial normal LVEF independently predicted end assessment hemodynamic suitability for transplantation. An initial abnormal left ventricular systolic function was identified in almost half of donor hearts, of which 58% achieved hemodynamic stability criteria for procurement (86). Although there is limited data from small case series, low-dose dobutamine stress tests may be able to detect myocardium that appears dysfunctional in the donor and may be capable of recrudescing function in the recipient (87).

FIGURE 119.2 Algorithmic approach to the cardiovascular and hemodynamic management of the potential organ donor.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








