KEY POINTS
1. An aortic dissection usually occurs when blood penetrates the aortic intima, forming either an expanding hematoma within the aortic wall or simply a false channel for blood flow between the medial layer.
2. An aortic aneurysm involves dilation of all three layers of the aortic wall.
3. The term dissecting aneurysm, although commonly used, is often a misnomer because the aorta may not be dilated.
4. Nitroprusside, a potent arterial dilator, and a β-blocker to decrease LV ejection velocity are critical to prevent further propagation of aortic dissection, rupture of a torn aorta, or leaking thoracic aortic aneurysm.
5. Aortic valve repair or replacement is often needed with repair of aortic dissections or aneurysms. Which procedure is used depends on involvement of the sinus of Valsalva and the aortic annulus.
6. Management of left heart bypass (LHB) can be very challenging for the cardiac anesthesiologist while repairing descending thoracic aneurysms or aortic tears (see Table 25.12). TEE is very useful to guide volume management, as native cardiac output must be preserved to provide adequate blood flow to the brain.
7. Cerebral spinal drainage has been shown to significantly decrease postoperative paraplegia and paraparesis in a randomized controlled trial in patients undergoing descending thoracic surgery.

ANESTHESIOLOGISTS CARING FOR THORACIC AORTIC surgical patients encounter considerable variation between patients with regard to the cause and location of aortic disease. It is vital that anesthesiologists understand the implications of and the challenges related to these variations in providing optimal perioperative care. Members of both the American Society of Anesthesiologists and the Society of Cardiovascular Anesthesiologists were involved in the multidisciplinary development of the 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM Guidelines for the Diagnosis and Management of Patients with Thoracic Aortic Disease [1]. This chapter gives a concise overview of the pathophysiology of thoracic aortic surgery, a review of its surgical approaches and results, and a rational approach to managing patients undergoing thoracic aortic surgery. Surgery on the thoracic aorta and, in particular, the descending and thoracoabdominal aorta may obviously involve complicated surgical and anesthetic management. Therefore, each team member must have a clear understanding of what is being planned. Often, all that is required is a brief preoperative conversation between the surgeon and the anesthesiologist as to the exact requirements for a particular procedure.
1
I. Classification and natural history
A. Dissections. An aortic dissection usually occurs when blood penetrates the aortic intima, forming either an expanding hematoma within the aortic wall or simply a false channel for flow between the medial layers. (This expanding hematoma may be called a dissecting hematoma.) The true lumen of the dissecting aorta is generally not dilated; rather, it is often compressed by the dissection. Because the dissection does not necessarily involve the entire circumference of the aorta, branching vessels may be not affected, they may be occluded, or they may arise from the false lumen. In contrast, an aortic aneurysm involves dilation of all three layers of the aortic wall and has different pathophysiology and management concerns. The term dissecting aneurysm, although commonly used, is often a misnomer because the aorta may not be dilated.
2
1. Incidence and pathophysiology
3
a. Incidence. The incidence of dissection in the United States is unclear mainly because of under-reporting; however, European studies have reported an incidence of 3.2 dissections per 100,000 autopsies, with increasing numbers over time. Also, dissections resulted in more deaths than did aneurysm rupture [2].
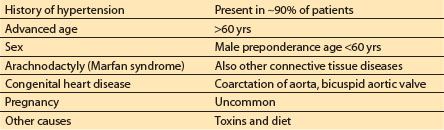
b. Predisposing conditions. The medical conditions predisposing to aortic dissection are listed in Table 25.1 in their order of importance. Interestingly, atherosclerosis by itself may not contribute to the risk of subsequent aortic dissection.
Table 25.1 Conditions predisposing to aortic dissections
c. Inciting event. The onset of aortic dissections has been associated with increased physical activity or emotional stress. Dissections also have been associated with blunt trauma to the chest; however, the temporal relationship of blunt trauma and subsequent dissections has not been well established. Dissections can occur without any physical activity. They may also occur during cannulation for cardiopulmonary bypass (CPB), either antegrade from the ascending aorta or retrograde from the femoral artery.
d. Mechanism of aortic tear. An intimal tear is the initial event in aortic dissection. The intimal tear of aortic dissections usually occurs in the presence of a weakened aortic wall, predominantly involving the middle and outer layers of the media. In this area of weakening, the aortic wall is more susceptible to shear forces produced by pulsatile blood flow in the aorta. The most frequent locations of intimal tears are the areas experiencing the greatest mechanical shear forces, as listed in Table 25.2. The ascending and isthmic (just distal to the left subclavian artery) segments of the aorta are relatively fixed and thus subject the aortic wall to the greatest amount of mechanical shear stress. This explains the high incidence of intimal tears in these areas.
Table 25.2 Sites of primary intimal tears in acute dissections

In large autopsy series, however, up to 4% of dissections had no identifiable intimal disruption. In these cases, rupture of the vasa vasorum, the vessels that supply blood to the aortic wall, has been implicated as an alternative cause of dissections. The thin-walled vasa vasorum are located in the outer third of the aortic wall, and their rupture would cause the formation of a medial hematoma and propagation of a dissection in the presence of an already diseased vessel, without formation of an intimal tear.
e. Propagation. Propagation of an aortic dissection can occur within seconds. The factors that contribute to propagation are the hemodynamic forces inherent in pulsatile flow: pulse pressure and ejection velocity of blood.
f. Exit points. Exit points of dissections are found in a relatively small percentage of cases. Exit point tears usually occur distal to the intimal tear and represent points at which blood from the false lumen re-enters the true lumen. The presence or absence of an exit point does not appear to have an impact on the clinical course.
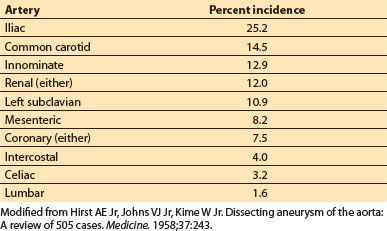
g. Involvement of arterial branches. The origins of the major branches of the aorta, including the coronary arteries, may be involved in aortic dissections. Their involvement ranges from branch vessel occlusion via mechanical compression by the false lumen or from propagation of the dissecting hematoma into the arterial branch. The incidence of involvement of arterial branches gathered from a large autopsy series is listed in Table 25.3 [3].
Table 25.3 Involvement of major arterial branches in aortic dissections
2. DeBakey classification of dissections (Fig. 25.1). This classification consists of three different types based upon the location of the intimal tear and which section of the aorta is involved.
Figure 25.1 DeBakey classification of aortic dissections by location: Type I, with intimal tear in the ascending portion and dissection extending to descending aorta; Type II, ascending intimal tear and dissection limited to ascending aorta; Type III, intimal tear distal to left subclavian, but dissection extending for a variable distance, either to the diaphragm (a) or to the iliac artery (b). (From DeBakey ME, Henly WS, Cooley DA, et al. Surgical management of dissecting aneurysms of the aorta. J Thorac Cardiovasc Surg. 1965;49:131, with permission.)
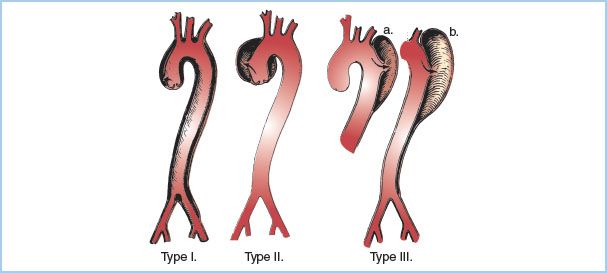
a. Type I. The intimal tear is located in the ascending portion, but the dissection involves all portions (ascending, arch, and descending) of the thoracic aorta.
b. Type II. The intimal tear is in the ascending aorta, but the dissection involves the ascending aorta only, stopping before the takeoff of the innominate artery.
c. Type III. The intimal tear is located in the descending segment. If the dissection involves the descending portion of the thoracic aorta only, starting distal to the origin of the left subclavian artery and ending above the diaphragm, it is a Type III A. If the dissection propagates below the diaphragm, it is a Type III B. By definition, Type III dissections can extend proximally into the arch, but this is rare.
3. Stanford (Daily) classification of dissections (Fig. 25.2). This classification is simpler than DeBakey’s and has more clinical relevance.
Figure 25.2 Stanford (Daily) classification of aortic dissections. Type A describes a dissection involving the ascending aorta regardless of site of intimal tear (1, ascending; 2, arch; 3, descending). In Type B, both the intimal tear and the extension are distal to the left subclavian. (From Miller DC, Stinson EB, Oyer PE, et al. Operative treatment of aortic dissections. Experience with 125 patients over a sixteen-year period. J Thorac Cardiovasc Surg. 1979;78:367, with permission.)

a. Type A. Type A dissections are those that have any involvement of the ascending aorta, regardless of where the intimal tear is located and regardless of how far the dissection propagates. Clinically, Type A dissections run a more virulent course and are generally considered urgent or emergent cases.
b. Type B. Type B dissections are those that involve the aorta distal to the origin of the left subclavian artery.
4. Natural history
a. Mortality—untreated. The survival rate of untreated patients with ascending aortic dissections is dismal, with a 2-day mortality of up to 50% in some series and 3-month mortality approaching 90% [3]. The usual cause of death is rupture of the false lumen into the pleural space or pericardium. Mortality is lower with DeBakey Type III or Stanford B dissections. Other causes of death include progressive cardiac failure (aortic valve involvement), myocardial infarction (coronary artery involvement), stroke (occlusion of cerebral vessels), and bowel gangrene (mesenteric artery occlusion).
b. Surgical mortality. Overall mortality ranges from 3% to 24% and varies with the section of aorta that is affected. Dissections involving the aortic arch carry the highest mortality, while those confined to the descending thoracic aorta carry the lowest [2].
B. Aneurysms
1. Incidence. The European studies cited above report an incidence of thoracic aneurysms in approximately 460 autopsies per 100,000. In one study, 45% of thoracic aneurysms involved the ascending aorta, 10% the arch, 35% the descending aorta, and 10% the thoracoabdominal aorta [4].
2. Classification by location and cause. In general, the causes and pathophysiology of aortic aneurysms are site dependent. Most commonly, medial degeneration affects the ascending aorta, while degenerative conditions associated with atherosclerosis affect the descending and thoracoabdominal portions of the aorta. Other causes are listed in Table 25.4.
Table 25.4 Causes of aneurysms based on location in the aorta

3. Classification by shape
a. Fusiform. Fusiform aneurysmal dilation involves the entire circumference of the aortic wall.
b. Saccular. Saccular aneurysms involve only part of the circumference of the aortic wall. Isolated aortic arch aneurysms are commonly saccular.
4. Natural history. The natural history of aortic aneurysms is one of progressive dilation, with more than half of aortic aneurysms eventually rupturing. The untreated, 5-yr rate of survival for patients with thoracic aortic aneurysms ranges from 13% to 39% [2]. Other complications of thoracic aortic aneurysms include mycotic infection, atheroembolism to peripheral vessels, and dissection. This last complication is rare, probably occurring in fewer than 10% of cases. Some predictors of poor prognosis are large size (greater than 10 cm maximum transverse diameter), presence of symptoms, and associated cardiovascular disease, especially coronary artery disease, myocardial infarction, or cerebrovascular accident.
C. Thoracic aortic rupture (tear)
1. Etiology. The overwhelming majority of thoracic aortic ruptures occur after trauma and almost always involve a deceleration injury from a motor vehicle accident. Sudden deceleration places large mechanical shear stress on points of the aortic wall that are relatively immobile. While aortic rupture leads to immediate exsanguination and death in many patients, approximately 10% to 15% of these patients maintain integrity of the adventitial covering of the aortic lumen and survive to emergency care. Surgical treatment of these survivors is often successful.
2. Location. Most thoracic aortic ruptures occur just distal to the origin of the left subclavian artery (isthmus) because of the relative fixation of the aorta at this point by the ligamentum arteriosum (Fig. 25.3). The second most common site of aortic rupture is in the ascending aorta, just distal to the aortic valve.
Figure 25.3 The heart and great vessels are relatively mobile in the pericardium, whereas the descending aorta is relatively fixed by its anatomic relations. The attachment of the ligamentum arteriosum enhances this immobility and increases the risk of aortic tear due to deceleration injury. (From Cooley DA, ed. Surgical Treatment of Aortic Aneurysms. Philadelphia, PA: WB Saunders; 1986:186, with permission.)

II. Diagnosis
A. Clinical signs and symptoms (Table 25.5)
Table 25.5 Presenting clinical signs and symptoms by location and type of aortic pathology



1. Dissections. Aortic dissections usually present with a dramatic onset and a fulminant course. Clinical presentation of Stanford Types A and B are listed in Table 25.5.
2. Aneurysms. Aneurysms of the ascending, arch, or descending thoracic aorta are often asymptomatic until late in their course. In many circumstances, the presence of an aneurysm is not diagnosed until medical evaluation is conducted for an unrelated problem or for a problem related to a complication of the aneurysm.
3. Traumatic rupture. Ruptures most commonly occur just distal to the left subclavian artery. If the patient survives the initial trauma, signs and symptoms are similar to those seen with aneurysms of the descending thoracic aorta.
B. Diagnostic tests
1. Electrocardiogram (ECG). Many patients with aortic disease will have evidence of left ventricular (LV) hypertrophy on ECG, secondary to the high incidence of hypertension in these patients. In the setting of aortic dissection, the ECG may show ischemic changes caused by coronary artery involvement or evidence of pericarditis from hemopericardium.
2. Chest X-ray film. A widened mediastinum is a classic X-ray finding with thoracic aortic pathology. Widening of the aortic knob is often seen, with disparate ascending-to-descending aortic diameter. A double shadow has been described in the setting of aortic dissection, secondary to visualization of the false lumen.
3. Serum laboratories. There are no laboratory findings specifically found with asymptomatic aortic aneurysms. Aortic dissections or ruptures will decrease hemoglobin. Dissections may cause elevated cardiac enzymes from coronary artery occlusion, may increase blood urea nitrogen and creatine from renal artery involvement, and may lead to metabolic acidosis from low cardiac output or ischemic bowel. Fibrinogen may decrease in patients experiencing associated disseminated intravascular coagulation.
4. Computed tomographic (CT) scans and magnetic resonance imaging. CT is a useful tool for ascertaining aneurysm size and location and has replaced angiography in many instances. It is also useful for following the progression of aortic disease. Digital images can be manipulated into a three-dimensional form, which may make it easier to assess the lesion and plan repair. Magnetic resonance imaging is extremely sensitive and specific in identifying the entry tear location, presence of false lumen, aortic regurgitation, and pericardial effusion accompanying aortic dissections [5].
5. Angiography. This technique remains useful for determining the severity and extent of aortic aneurysms and dissections. With dissections it can be used to locate the site of an intimal tear, to assess aortic valve integrity, and to identify the distal and proximal spread. It can especially delineate involvement of the coronary arteries, as well as the presence of significant coronary artery disease in patients with ascending aortic pathology. Patients with disease of the thoracic aorta often have concurrent coronary artery disease, and bypassing significant lesions helps prevent perioperative myocardial infarction and improves ventricular function when weaning from CPB. Aortography can also determine if other major branch arteries off the aorta are involved. Unfortunately, aortography only rarely identifies those intercostal vessels that are critical for providing blood supply to the spinal cord (see Section IV.G).
6. Transesophageal echocardiography (TEE). TEE has been found to be highly sensitive and specific for diagnosing aortic dissection. In many cases, pulsed-wave and color-flow Doppler imaging can aid in defining the presence, extent, and type of dissection. Identification of a mobile intimal flap provides a prompt bedside diagnosis that can be lifesaving. In addition, entry and re-entry tears can be defined; aortic regurgitation can be identified and quantified; assessment of LV function and wall motion abnormalities can be made; presence of pericardial effusion with possible associated cardiac tamponade can be identified; and follow-up studies of the false lumen can be made after therapeutic intervention.
TEE can also be used to assess many thoracic aortic aneurysms. For ascending or descending thoracic aortic aneurysms in particular, the location, diameter, and extent of the aneurysm, as well as whether the aneurysm contains significant atheroma, can often be well described. TEE can also be used to determine if the aneurysm is saccular or fusiform in shape. TEE can be rarely used to define definitive aneurysmal involvement of the aortic arch and distal ascending aorta. This is because the trachea obscures the latter section from transesophageal ultrasound imaging and the entire arch is not usually visible on TEE. If the surgeon desires detailed preoperative imaging of the aortic arch, he or she should confer with a radiologist about magnetic resonance or CT imaging.
Because traumatic aortic transection generally occurs just distal to the left subclavian takeoff, it is often easily and rapidly identified by TEE imaging. In addition, aortic transection operations are usually emergencies, so TEE imaging is advantageous because it can be conducted quickly and in most locations.
7. Recommendation for diagnostic strategies. In hemodynamically stable patients, Nienaber and colleagues recommend diagnosis of thoracic aortic dissection noninvasively via magnetic resonance imaging because of its high degree of sensitivity (98.3%) and specificity (97.8%) [6]. For patients too unstable for this rather lengthy procedure (40 to 45 min), TEE, which has an average duration of about 15 min and a sensitivity and specificity of 97.7% and 76.9%, respectively, is recommended. Because of its general inability to provide additional information to that provided by more noninvasive methods and its higher incidence of complications, aortography is only useful in very select cases.
C. Indications for surgical correction
1. Ascending aorta
a. Dissections. Acute Type A dissections should be surgically corrected, given their virulent course and high mortality if not surgically treated.
b. Aneurysms. Indications for surgical resection include the following:
(1) Presence of persistent pain despite a small aneurysm
(2) Aortic valve involvement producing aortic insufficiency
(3) Presence of angina from LV strain secondary to aortic valve involvement or from coronary artery involvement by the aneurysm
(4) Rapidly expanding aneurysm or an aneurysm greater than 5 to 5.5 cm in diameter, because the chance of aortic rupture increases as the aneurysm’s size increases
2. Aortic arch
a. Dissections. Acute dissection limited to the aortic arch is rare but is an indication for surgery.
b. Aneurysms. Since even elective surgical treatment of arch aneurysms is more difficult than surgery for other aortic aneurysms and is associated with a higher morbidity and mortality, management may be more conservative. However, arch involvement is often seen with ascending aneurysms (less so with descending aneurysms) and is dealt with during surgical repair of these lesions. Surgical indications include the following:
(1) Persistent symptoms
(2) Aneurysm greater than 5.5 to 6 cm in transverse diameter
(3) Progressive aneurysmal expansion
3. Descending aorta
a. Dissection. Some controversy remains concerning the best treatment for an acute Type B dissection. Because of similar in-hospital mortality statistics for medical versus surgical interventions [7], Type B dissections are often treated medically in the acute phase, especially if the patient’s comorbidities make the chance of surgical mortality prohibitively high. However, patients with Type B dissections who have the following complications are treated surgically:
(1) Failure to control hypertension medically
(2) Continued pain (indicating progression of the dissection)
(3) Enlargement on chest X-ray film, CT scan, or angiogram
(4) Development of a neurologic deficit
(5) Evidence of renal or gastrointestinal ischemia
(6) Development of aortic insufficiency
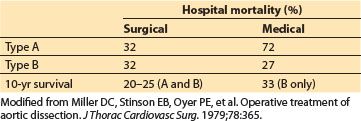
Note, as shown in Table 25.6, that the 10-yr survival of patients receiving medical management only for Type B dissections is slightly better than the combined survival of patients treated surgically for Type A and B dissections [7].
Table 25.6 Surgical versus medical therapy for aortic dissections
b. Aneurysm. Indications for surgical repair of descending thoracic aneurysms include the following:
(1) Aneurysm greater than 5 to 6 cm in diameter
(2) Aneurysm expanding
(3) Aneurysm leaking (more fulminant symptoms)
(4) Chronic aneurysm causing persistent pain or other symptoms
III. Preoperative management of patients requiring surgery of the thoracic aorta.
Emergency preoperative management of aortic dissections is discussed below. However, emergency preoperative management is similar for a leaking thoracic aortic aneurysm or a contained thoracic aortic rupture .
A. Prioritizing: Making the diagnosis versus controlling blood pressure (BP). In the setting of a suspected aortic dissection, aortic tear, or leaking aortic aneurysm, the first priority is always to control the BP and ventricular ejection velocity, as these propagate aortic dissection or rupture. If dissection is strongly suspected, making a definitive diagnosis with radiographic studies should occur after proper monitoring, intravenous (IV) access, hemodynamic stability, and heart rate and BP control have been established (if possible). During diagnostic procedures, the patient should be monitored closely, with a physician present as clinically indicated. An anesthesiologist should become involved as early as possible, if needed, to lend expertise in monitoring and airway and hemodynamic management in cases where clinical deterioration occurs before the patient reaches the operating room. Rapid diagnosis using TEE may save critical minutes in initiating definitive surgical treatment in patients with suspected thoracic aortic dissection or rupture.
4
B. Achieving hemodynamic stability and control. The ideal drug to control BP is administered by IV and is rapidly acting with a short half-life and few, if any, side effects. Systolic and diastolic BPs and LV ejection velocity should be reduced, because all these factors can propagate aortic dissection.
1. Monitoring. Patients must have an ECG for detection of ischemia and dysrhythmias, two large-bore IV catheters for volume resuscitation, an arterial line in the appropriate location (discussed below), and, if time permits, a central venous catheter or pulmonary artery (PA) catheter for monitoring filling pressures and infusing drugs centrally.
2. BP-lowering agents
a. Vasodilators
(1) Nitroprusside has emerged as a useful agent for controlling BP in patients with critical aortic lesions, because its rapid onset and offset make it quickly effective and easily regulated. A vasodilator that relaxes both arterial and venous smooth muscle, it is given as an IV infusion, and while central administration is probably optimal, it can be administered through a peripheral vein with good effect. The usual starting dose is 0.5 to 1 μg/kg/min, titrated to effect. Doses of 8 to 10 μg/kg/min have been associated with cyanide toxicity (see Chapter 2).
(2) Nitroglycerin is a less potent vasodilator than sodium nitroprusside, and it causes more venous than arterial dilation. It can be useful in settings where ascending aortic pathology is coupled with myocardial ischemia, as it can improve coronary blood flow via coronary artery vasodilation. Infusion dosage usually ranges from 1 to 4 μg/kg/min.
(3) Fenoldopam is a rapidly acting vasodilator that is a selective D1 dopamine receptor agonist. It has little affinity for the D2, α1, or β adrenoreceptors. Fenoldopam causes vasodilation in many vascular beds, but it increases renal blood flow to a significant degree. Therefore, it may have some renal protective effects while also being used to treat acute hypertension. Dosing starts at 0.05 to 0.1 μg/kg/min and can be increased incrementally to a maximum dose of 0.8 μg/kg/min.
(4) Nicardipine is a calcium-channel blocker that inhibits calcium influx into vascular smooth muscle and the myocardium. It may be used as a single 0.5 to 2 mg IV “push” or as a 5 to 15 mg/hr infusion titrated to the desired effect.
b. b1-antagonists
Decreasing LV ejection velocity is important for decreasing risk of propagating aortic dissection. Medications to lower heart rate may be particularly useful for attenuating reflex tachycardia and increased ventricular contractility that can occur with use of sodium nitroprusside. Nitroprusside can increase LV ejection velocity by increasing dP/dt and heart rate. For this reason, β -adrenergic blockade should be used with nitroprusside to decrease both tachycardia and contractility (see Chapter 2).
(1) Propranolol, a nonselective β -antagonist, has been used for many years as first-line therapy for this role and can be administered as an IV bolus of 1 mg, but doses of 4 to 8 mg may be required for adequate heart rate control. Propranolol has been somewhat supplanted by selective β1-antagonists.
(2) Labetalol is a combined α– and β-blocker and offers an alternative to the nitroprusside–propranolol combination. It should be given initially as a 5 to 10 mg loading bolus; once the effect has been assessed, the dose is doubled, allowing a few minutes for onset of effect. This process should be repeated until target BP or a total dose of 300 mg is reached. Once target BP and heart rate are achieved via the loading dose, a continuous infusion may be started at 1 mg/min, or a small bolus dose can be repeated every 10 to 30 min to maintain BP control.
(3) Esmolol is a β-blocking agent with a short half-life that may be useful in this setting. It is administered as a bolus loading dose of 500 μg/kg over 1 min and then continued as an infusion starting at 50 μg/kg/min and titrated to effect to a maximum dose of 300 μg/kg/min. This drug is particularly useful in patients with obstructive lung disease because it is β1 selective and its action can be terminated quickly if β2-mediated respiratory symptoms ensue.
(4) Metoprolol, another β1-selective agent, is used in doses of 2.5 to 5 mg titrated to effect over a few minutes to a maximum dose of 15 to 20 mg. It provides a longer effect, which may be useful.
3. Desired endpoints. In order to decrease the chance of propagating aortic dissection or rupture, systolic BP should usually be lowered to approximately 100 to 120 mm Hg or to a mean pressure of 70 to 90 mm Hg. Heart rate should be 60 to 80 beats/min. If a PA catheter is in place, the cardiac index may be lowered to a range of 2 to 2.5 L/min/m2 to reduce ejection velocity from a hyperdynamic LV.
C. Bleeding and transfusion. Coagulopathy is frequently encountered in the thoracic aortic surgical patient. Many of these patients require left heart or full CPB during surgery to help maintain sufficient end-organ perfusion during aortic repair; thus, they also require heparinization. CPB may cause a consumptive coagulopathy and enhanced fibrinolysis, thus increasing blood loss [8,9]. Patients requiring deep hypothermic circulatory arrest (DHCA) for aortic arch surgery also may experience substantial platelet dysfunction secondary to extreme hypothermia. Platelet consumption has also been noted in the abdominal aortic surgical population [10]. In patients undergoing thoracoabdominal aortic aneurysm repairs, “back-bleeding” through intercostal vessels increases blood loss, and very large losses necessitating transfusion of multiple units of blood products can occur [11].
1. A total of 8 to 10 units of packed red blood cells should be typed and cross-matched before surgery.
2. Use of blood scavenging/reprocessing devices decreases the amount of banked blood transfused, but extensive bleeding and the logistics of effectively scavenging autologous blood during these operations may frequently require transfusion of packed cells and procoagulant blood products.
3. Antifibrinolytic therapy during aortic surgery is controversial but commonly used. Few adequately powered trials have examined this surgical population, so it is unclear if significant benefit is derived from antifibrinolytics, particularly in patients in whom left heart bypass (LHB) is used and full heparinization is unnecessary [12].
a. Tranexamic acid or -aminocaproic acid. A retrospective study of 72 patients who underwent descending thoracic aortic surgery with LHB and tranexamic acid or -aminocaproic acid infusion versus no antifibrinolytic therapy found no difference in incidence of transfusion or chest tube output; however, all of these patients also received intraoperative methylprednisolone and platelet-rich plasmapheresis before aortic repair [12]. These authors did find that intraoperative hypothermia independently predicted chest tube output and that preoperative hemoglobin, older age, and duration of cross-clamp time independently predicted transfusion. Casati and colleagues conducted a single-institution, double-blind, randomized, controlled study of 60 consecutive elective thoracic aortic surgical patients, half of whom received perioperative tranexamic acid and the other half received a normal saline placebo [13]. This study found that the tranexamic acid recipients required significantly fewer packed red blood cell transfusions, as well as overall allogeneic transfusions, than the placebo recipients. No differences in perioperative thrombotic complications were noted between the tranexamic acid intervention group and the placebo group. However, this was a small study that was not statistically powered to evaluate the occurrence of adverse perioperative thrombotic events. Larger prospective randomized studies are needed to determine whether tranexamic acid or -aminocaproic acid effectively reduce bleeding and do not cause thrombotic complications in thoracic aortic operations, because there are no other data regarding the risk/benefit of these agents in thoracoabdominal aortic surgical patients.
b. On the basis of available data, the authors can neither recommend nor advise against the use of antifibrinolytic therapies in thoracic aortic surgery. Potential thrombotic risks, including neurocognitive and renal dysfunction, are of concern, and the clinician should weigh these risks against the benefits of potential decreases in transfusion.
D. Assessment of other organ systems
1. Neurologic. Preoperatively the patient should be monitored closely for change in neurologic status, as this is an indication for immediate surgical intervention. Involvement of the artery of Adamkiewicz may lead to lower extremity paralysis, while propagation of a dissection into a cerebral vessel may lead to a change in mental status or stroke symptoms.
2. Renal function. Urine output should be monitored, as development of anuria or oliguria in the euvolemic setting is an indication for immediate surgical intervention.
3. Gastrointestinal. Serial abdominal examinations should be performed, and blood gas analysis should be done routinely to assess changes in acid–base status. Ischemic bowel can cause significant metabolic acidosis.
E. Use of pain medications. Patients with aortic dissections may be anxious and in severe pain. Not only is pain relief important for patient comfort, but it is beneficial in controlling BP and heart rate. Oversedation should be avoided so that ongoing patient assessments may occur. In addition to neurologic or abdominal symptoms, worsening of back pain may indicate aneurysm expansion or further aortic dissection and is regarded by many surgeons as an emergent situation.
IV. Surgical and anesthetic considerations
A. Goal of surgical therapy (for aortic dissections, aneurysms, or rupture). The foremost goal in treating acute aortic disruption is to control hemorrhage. Once control is achieved, the objectives of management of both acute and chronic lesions are to repair the diseased aorta and restore its relationships with major arterial branches.
Thoracic aortic aneurysm repair is usually conducted by replacing the diseased segment of the aorta with a synthetic graft and then reimplanting major arterial branches into the graft. In contrast, when repairing an aortic dissection the goal is to resect the segment of the aorta that contains the intimal tear. When this segment is removed, it may be possible to obliterate the false lumen and interpose graft material. It is usually not possible or necessary to replace the entire dissection portion of aorta, because, if the origin of dissection is controlled, re-expansion of the true lumen usually compresses and obliterates the false lumen. With contained aortic rupture, the objective is to resect the area of the aorta that ruptured and either reanastomose the natural aorta to itself in an end-to-end fashion or interpose graft material for the repair.
B. Overview of intraoperative anesthetic management (for aortic dissections, aneurysms, or rupture)
1. Key principles
a. Managing BP. BP control should be sought during the transition from the preoperative to the intraoperative period. Such control is important in light of the surgical and anesthetic manipulations that will profoundly affect BP.
b. Monitoring of organ ischemia. If possible, the central nervous system, heart, kidneys, and lungs should be monitored for adequacy of perfusion. The liver and gut cannot be monitored continuously, but their metabolic functions can be checked periodically.
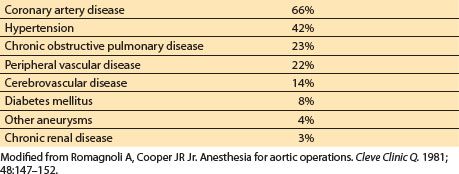
c. Treating coexisting disease. Patients with aortic pathology often have associated cardiovascular and systemic diseases, as outlined in Table 25.7.
Table 25.7 Incidence of coexisting diseases in patients with aortic pathology
d. Controlling bleeding. Patients undergoing aortic surgery often experience an inflammatory response to foreign graft material and cardiopulmonary or LHB. This inflammation can interact with the coagulation cascade and lead to significant perioperative coagulopathy. Furthermore, patients with acute dissection and lower fibrinogen and platelet counts may already have a consumptive process from the clotting that often occurs in the false lumen. The challenges of coagulation abnormalities and their treatment are discussed in Chapter 19.
2. Induction and anesthetic agents. Many thoracic aortic operations are emergent procedures that require aspiration precautions when securing the airway. However, rapid sequence induction and intubation typically done for patients with full stomachs may not be appropriate for the patient with thoracic aortic pathology, as wide swings in hemodynamics may occur. A compromise in this situation is a smooth, controlled IV induction, with gentle manual ventilations and cricoid pressures held. This “modified” rapid sequence induction allows not only some airway protection but also titration of anesthetic induction drugs that control BP with laryngoscopy. Use of nonparticulate antacids, H2-blockers, and metoclopramide should be considered before induction of anesthesia. Other anesthetic considerations and agents are described more fully in Section IV.D. Despite precautions, marked changes in hemodynamics are common when securing the patient’s airway, and vasoactive drugs (nitroglycerin, esmolol, or others) should be available to immediately treat an undesirable hemodynamic response to intubation [14].
3. Importance of site of lesion (Table 25.8). Although the principles of anesthetic induction and maintenance are similar for all aortic lesions, the location of the thoracic aortic lesion is also important for intraoperative management.
Table 25.8 Anesthetic and surgical management for thoracic aortic surgery

C. Ascending aortic surgery
1. Surgical approach. Ascending aortic surgery is conducted through a midline sternotomy.
2. CPB. CPB is required because of proximal aortic involvement.
a. If the aneurysm ends in the proximal portion or midportion of the ascending aorta, the arterial cannula for CPB can be placed in the upper ascending aorta or proximal arch.
b. If the entire ascending aorta is involved, the femoral artery may be cannulated, because an aortic cannula cannot be placed distal to the lesion without jeopardizing perfusion to the great vessels. Arterial flow on CPB in this case is retrograde from the femoral artery toward the great vessels. Another, newer approach is to cannulate the right axillary, the innominate, or occasionally the right carotid artery, allowing retrograde perfusion into the innominate artery and then into the aorta in an antegrade manner.
c. Venous cannulation is usually through the right atrium; however, femoral venous cannulation may be necessary if the aneurysm is very large and obscures the atrium.
3. Aortic valve involvement. Aortic valvuloplasty or valve replacement is often needed with repair of ascending aortic dissections or aneurysms. Which procedure is used depends on the degree of involvement of the sinus of Valsalva and the aortic annulus.
5
4. Coronary artery involvement. Ascending aortic dissections or aneurysms may involve the coronary arteries. Aortic dissections may cause coronary occlusion by compression of the coronary ostia by an expanding false lumen; such occlusion will require surgical coronary artery bypass grafting to restore myocardial blood flow. Displacement of the coronary arteries from their normal position distal to the aortic annulus with proximal aortic aneurysms usually requires coronary artery reimplantation into the reconstructed aortic tube graft or coronary artery bypass grafting.
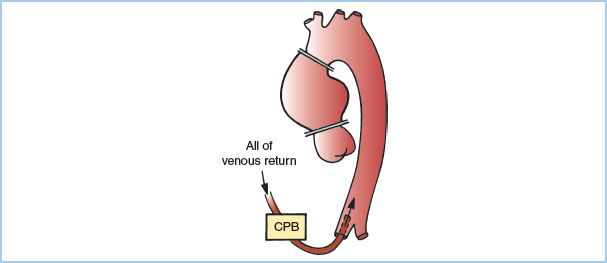
5. Surgical techniques. An example of the usual cross-clamp placement used in surgery of the ascending aorta is shown in Figure 25.4. Note that placement of the distal clamp is more distal than when cross-clamping for coronary artery bypass surgery and might include a part of the innominate artery. If aortic insufficiency is present, a large portion of the cardioplegic solution infused into the aortic root will flow through the incompetent aortic valve and into the LV instead of the coronary arteries. This can cause distention of the LV with increased myocardial oxygen utilization and diminished myocardial protection from reduced distribution of cardioplegia. For these reasons, an immediate aortotomy is often performed after aortic cross-clamping with direct infusion of cardioplegia into individual coronary arteries. Many centers also use retrograde coronary perfusion for cardioplegia administration as an alternative or in addition to an antegrade technique.
Figure 25.4 Circulatory support and clamp placement for surgery of the ascending aorta if femoral arterial cannulation is used; the distal clamp must be distal to the diseased segment. This may be the only clamp required. CPB, cardiopulmonary bypass. (From Benumof JL. Intraoperative considerations for special thoracic surgery cases. In: Benumof JL, ed. Anesthesia for Thoracic Surgery. Philadelphia, PA: WB Saunders; 1987:384, with permission.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








