1. Organized chronic cardiac thrombus from atrial fibrillation
2. Subacute thrombus from ischemia endocardium of acute MI
3. Iatrogenic catheter induced thrombus
4. Paradoxical embolism from DVT, multiple PE with patent foramen ovale
5. Peripheral artery aneurysm – congenital or atherosclerotic origins
6. Atherosclerotic aortic plaque
7. Aortic aneurysm
Peripheral arterial aneurysms are an uncommon but important source of distal emboli. At the time of presentation, there is usually evidence of multiple prior episodes of small emboli followed by the current complete thrombosis and acute distal limb ischemia [16]. These aneurysms can be of both an atherosclerotic and, less commonly, a congenital origin. Popliteal artery atherosclerotic aneurysms are the most common peripheral aneurysms and are associated with bilateral lesions in 50 % and abdominal aortic aneurysms in 60 % of patients [17, 18]. Congenital popliteal entrapment and thoracic outlet syndrome with arterial compression can lead to aneurysms with embolic and thrombotic complications [19, 20]. Rupture of peripheral aneurysms is rare [17]. If left untreated, these rare congenital aneurysms are associated with a significant risk of limb loss because of chronic small distal emboli, acute thrombosis, and delay in diagnosis [19–21].
The increasing use of the extremity arteries for diagnostic and therapeutic endovascular techniques has resulted in a rise in iatrogenic acute limb ischemia [9, 22, 23]. Catheterization site arterial occlusion may result from an intimal dissection, accumulation of thrombus along indwelling catheter sheaths with subsequent thrombosis or distal embolism, and intravascular occlusion from misplaced closure devices. Diagnosis of this source of acute ischemia is also frequently delayed. Similarly, the outcome is dependent upon prompt recognition and treatment.
The ischemic pattern in the extremity with the classic “five Ps” of arterial occlusion occurs in the extremity one level distal to the area of occlusion [5, 13]. If complete, this ischemia results in skeletal muscle and nerve tissue death at approximately 6 h [13]. This has given rise to the concept of a “golden period” of 6 h. This is the period of time between the onset of ischemia and the successful restoration of flow to salvage the limb from permanent loss of muscle and nerve tissue (Table 20.2).
Table 20.2
Acute and chronic signs and symptoms of extremity arterial occlusive disease
Acute ischemia |
Sudden onset of the “5 Ps” |
Pain |
Pulselessness |
Pallor |
Paresthesias |
Paralysis |
Absent or monophasic Doppler tones at the ankle or wrist |
Chronic arterial occlusion |
Mild: |
Diminished distal pulses |
Mild claudication of the legs or exercise-induced muscle pain arm relieved by rest |
Ankle or wrist pressure index 0.6–0.75 |
Moderate: |
Absent distal pulses |
Severe exercise-induced extremity pain |
Extremity pressure index <0.6 |
Severe: |
Night pain of the forefoot or numbness of the hand |
No exercise tolerance |
Extremity pressure index <0.5 |
Limb threat: |
Rest pain in the extremity |
Nonhealing ulcer digits, heel, or palm |
Dependent rubor |
Blanching on elevation |
Absent or monophasic distal Doppler tones |
Iatrogenic arterial occlusion is an increasingly common cause of acute limb ischemia [9]. The use of the femoral artery for catheter access for endovascular cardiac and peripheral interventions places a large number of patients at risk for acute ischemia from access site occlusions [22, 23]. Orthopedic surgical procedures in the hip and knee also put the femoral artery and the popliteal artery at risk for acute occlusion [25].
Acute massive venous thrombosis and outflow occlusion can cause acute limb ischemia. This entity is known as phlegmasia cerulean dolens or painful blue edema [26]. The venous obstruction and engorgement with desaturated blood is associated with significant pain and resulting arterial vasoconstriction. Left untreated, limb threatening ischemia results from the accumulation of desaturated venous blood, decreased arterial flow, and compartment syndrome [26].
Clinical Presentation and Diagnosis
Acute limb ischemia presents with sudden onset pain followed quickly by numbness and weakness. In patients with pre-existing occlusive disease, these symptoms may be less distinct [5, 13]. The most common clinical setting of acute limb ischemia includes a history of atrial fibrillation [5, 16]. Pre-existing lower extremity occlusive arterial disease with thrombosis is the next most common setting. These acute on chronic limb ischemia patients will have a history of claudication or past extremity angioplasty with stent placement or arterial bypass surgery. The third most common clinical setting is a history of either acute or chronic ischemic heart disease with thromboembolism when mural thrombus dislodges from the damaged endocardium [16]. There is rising incidence of catheterization site occlusion in patients undergoing cardiac endovascular procedures [9, 22]. These iatrogenic lesions also occur in the setting of associated cardiac disease.
History and physical examination quickly reveal the most likely source of the acute occlusion in the vast majority of patients. Cardiac source emboli usually occur in the setting of atrial fibrillation or acute myocardial infarction. Less commonly, emboli result from paradoxical embolism of venous thrombus through a patent foramen ovale in patients with lower extremity deep venous thrombosis and multiple pulmonary emboli [15]. The resulting pulmonary hypertension with opening of an incompletely closed foramen ovale allows passage of a venous embolism from the right atrium through the foramen ovale into the left atrium and into the systemic circulation. Rarely, emboli from atherosclerotic arterial plaque or a peripheral aneurysm present as acute limb ischemia [8, 16]. These lesions more commonly cause digital ischemia. Thrombosis of a pre-existing bypass graft or a stent is an important source of acute ischemia in patients with previous surgical or endovascular management of chronic disease [14]. This group of patients usually has a progression of distal occlusive disease and, less commonly, occlusion of a patent graft in the absence of identifiable progression of disease. These patients must undergo detailed catheter arteriography and often are candidates for thrombolytic therapy [14]. Consultation with a vascular surgeon is usually required in this setting.
Most patients with acute limb ischemia from embolism do not have a history of claudication and have normal pulses in the contralateral, non-affected extremity [5, 8, 16]. Saddle embolism to the aortic bifurcation may result in an absence of palpable pulses in both legs [27]. Acute thrombosis of pre-existing arterial occlusive disease usually occurs in the setting of pre-existing claudication. Physical exam usually reveals diminished pulses in the contralateral extremity and signs of chronic ischemia in the affected extremity. Ankle brachial indices may also reveal contralateral occlusive disease (Table 20.2).
Complete thrombosis of the infrarenal aorta is a rare and usually catastrophic event [28]. Practically all of these patients have significant cardiac disease with poor cardiac output which leads to thrombosis of chronic occlusive disease of the distal aorta and iliac bifurcation. The clinical presentation may include buttock and leg muscle weakness or paraplegia from distal spinal cord and lumbar plexus ischemia. The diagnosis is often delayed because of the baffling constellation of clinical findings despite the fact that physical examination reveals the bilateral absence of pulses from the femoral arteries distally with lower body and leg mottling. The mortality rate in these patients exceeds 50 % [28].
Phlegmasia cerulean dolens presents in the setting of massive lower extremity DVT complicated by arterial vasoconstriction and dehydration [26]. This lower extremity and, rarely, upper extremity syndrome is striking in its appearance with a swollen, blue, and cool to the touch limb [26]. Severe pain is always present. The compelling nature of these findings usually prompts venous duplex studies which confirm the diagnosis.
Doppler assessment of arterial flow in the extremity is an essential adjunctive measure to add to physical examination. Although the experienced examiner can assess flow based on the character of the audible Doppler signals, the best way to use the Doppler device is in conjunction with an extremity systolic blood pressure determination. The manual blood pressure cuff is placed at the wrist or ankle and the probe placed over the distal vessel. The cuff is slowly inflated, and the cessation of signals indicates the systolic blood pressure at the level of the cuff. Normal ankle brachial index (ABI) is 1.1 [6]. Less than 0.8 is abnormal. In general, claudication begins to be significant at that level of reduced flow. Below an ABI 0.5 or an ankle systolic pressure of 60 torr, potentially limb-threatening ischemia begins [6] (Table 20.2).
Once the diagnosis of acute limb ischemia due to a cardiac source embolism is made, there is an important decision to be considered. If the patient with distal limb ischemia has atrial fibrillation, no history of prior claudication or arm exercise intolerance, an otherwise normal peripheral vascular examination with a normal groin or axillary pulse proximal to the ischemic area, and normal contralateral limb pulses, immediate operative exploration for embolectomy is indicated [8]. Prompt CT angiography is indicated if the distribution of emboli is unclear. However, if the patient has pre-existing renal insufficiency, the risk of contrast nephropathy must be carefully considered when considering CT angiography.
Although thrombolytic therapy has been widely advocated in this setting, it does not offer the same prompt removal of the obstructing embolic debris that occurs with immediate embolectomy. Emboli from the left heart usually have a fibrous component that is not fully removed by thrombolysis and suction embolectomy catheters. Fogarty catheter embolectomy may be more effective and expedient. There is a tendency to want to avoid the morbidity of a general anesthetic in elderly patients. This may lead to days of infusing TPA in an ICU setting and, in reality, may involve overall increased morbidity including increased rates of limb loss. The choice of endovascular vs. open revascularization must be carefully made to optimize limb salvage and limit morbidity and mortality [29–34].
Preoperative CT or catheter arteriography is essential in patients who do not have a clearly identifiable cardiac source embolism or an obvious level of arterial occlusion. Acute on chronic occlusion needs to be delineated with detailed arteriography [13]. Although CT angiography may be adequate, catheter arteriography is preferred. In the setting of pre-existing chronic occlusive disease, catheter arteriography also allows for endovascular techniques when appropriate [35, 36].
The most important factor to consider in obtaining arteriography is the time it takes and the potential delay to the operating room. Remembering the “golden period,” there must be a rapid workup if arterial flow is to be successfully restored within 6 h. Therefore, if endovascular therapy is not an option, a high-resolution CT angiography should be obtained immediately.
Compartment syndrome should be anticipated in patients with acute limb ischemia [37]. All muscle compartments in the extremities are vulnerable to reperfusion intra-compartmental hypertension that can lead to muscle necrosis after revascularization [38]. Compartment syndrome results from post-ischemia swelling of muscle in the confined space created by the muscle fascia in the extremities. This swelling increases the tissue pressure within the compartment compressing the first lymphatic and then venous outflow with eventual occlusion of arteriolar inflow [28]. Ultimately the tissue perfusion pressure threshold of 25 mm Hg is exceeded, and ischemic neurolysis and myonecrosis occurs. It may take hours to occur after restoration of flow, and therefore serial examinations are essential to prevent delaying diagnosis and treatment. Compartment pressures should be measured with the Stryker device or by other devices (Fig. 20.1). Compartment syndrome occurs most commonly in the muscle compartments of the calf and is relatively uncommon in nontraumatic arterial occlusion in the upper extremity [37].
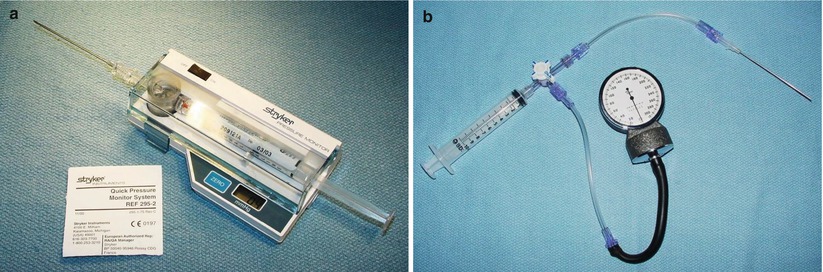

Fig. 20.1
(a) Stryker™ pressure-monitoring device for muscle compartment pressure measurement. (b) Alternative pressure monitoring device for muscle compartment pressure measurement. The syringe and half of the line to the manometer is filled with saline
Endovascular vs. Open Surgical Management
The integrated approach with both endovascular and open vascular surgical technical abilities is essential to the successful management of acute limb ischemia [29, 39]. The decision to choose one over the other method of relieving limb ischemia is dependent not only on the presence of acute vs. chronic vascular occlusive disease but also the status of the ischemic limb. Acute limb-threatening ischemia demands prompt revascularization if amputation is to be avoided. The likelihood and promptness of successful revascularization by either an endovascular or an open approach must guide the decision to go to the angiography suite or the operating room. Although hybrid suites offer both capabilities, decisive action must occur by choosing one or the other modalities and promptly intervening. If endovascular therapy is chosen, the conversion to an open approach must remain an option if further limb-threatening ischemia continues to the point of certain limb loss.
The most common cause of preventable limb loss after successful revascularization in both endovascular and open surgical procedures is failing to perform fasciotomy in the presence of calf compartment syndrome. There is an increased likelihood of failing to diagnose compartment syndrome with endovascular treatment of acute limb ischemia. Careful monitoring of calf compartment pressures and prompt fasciotomy must follow all endovascular procedures for acute limb ischemia.
Open Surgical Management
The performance of emergency vascular surgery should be limited to those surgeons who are capable and qualified. That does not limit these procedures to those who are board certified in vascular surgery. There are many general surgeons who are very skilled in vascular technique by virtue of their interest and experience. There should be a designated call panel for appropriate vascular surgical backup at all times for an acute care surgery service.
The use of checklists to manage acute surgical emergencies is strongly recommended. These are best prepared in advance and should be printed and posted in the operating room. Examples of checklists are provided below, and I recommend each surgeon to create their own version to include their choices for operative management (Figs. 20.2 and 20.3).



Fig. 20.2
Checklist for femoral thrombectomy for cardiac source embolism and ischemia leg

Fig. 20.3
Checklist for four-compartment calf fasciotomy
The patient should be widely prepped and draped with generous inclusion of the entire upper or lower extremity and the shoulder or lower abdomen. One leg should also be prepped and draped from inguinal region to toes to allow for saphenous vein harvest. Adjunctive measures such as bolus intravenous systemic heparinization, the administration of a continuous infusion of low molecular weight Dextran™, and administration of intravenous antibiotics should be considered and utilized where appropriate. Preparation for surgery should also include appropriate management of associated cardiopulmonary disease by the anesthesiologist.
Surgical exposure requires appropriately placed incisions that provide for exploration and reconstruction. In the upper extremity, the axillary artery is exposed by making a transverse infraclavicular incision over the delto-pectoral groove. A muscle-splitting incision is carried down through the pectoralis major muscle. The pectoralis minor muscle is divided close to the coracoid process and the axillary artery and vein exposed where they traverse just below the plane of the muscle. There are cords of the brachial plexus, nerves to the pectoral muscles, and large muscular branches of the artery in this area. Proximal and distal exposure of the artery should be carefully obtained avoiding damage to the brachial plexus and the axillary vein. Silastic vessel loops should be double passed proximally and distally and used to gently occlude the vessel. The artery is relatively fragile, and pulling too vigorously on the vessel loops may fracture the arterial intima causing a dissection. A transverse arteriotomy is performed, and proximal and distal thrombectomy with appropriately sized Fogarty catheters is carried out. A gentle heparinized saline flush (10 units heparin per ml) proximally and distally is performed taking care to not flush air or residual thrombus back up the vertebral arteries or the common carotid artery on the right side which can cause cerebral emboli and stroke.
The brachial artery is best exposed through a longitudinal incision along the medial aspect of the upper arm over the groove between the triceps and biceps muscles. The incision can be extended distally with an “S”-shaped extension across the antecubital fossa from ulnar to radial aspect and onto the forearm to expose the origins of the forearm vessels when the occluding thrombus is located at that level. (See below in Case Presentation 1.)
The location of incisions for acute lower extremity ischemia is determined by the level of the embolic occlusion. If the acute ischemic episode is due to a thrombosis of severe pre-existing occlusive disease, a bypass graft, or a stented arterial segment, a vascular surgery colleague should be consulted to appropriately manage this complex problem. Iliac or femoral artery occlusion from an embolism is best approached through a longitudinal incision over the common femoral artery in the groin. The common, superficial, and profunda femoral arteries are controlled with Silastic vessel loops. If the common femoral is soft and free of significant atherosclerotic plaque, a transverse arteriotomy is made. If it is not and chronic atherosclerotic changes are present, a longitudinal arteriotomy is the safest approach. Proximal and distal Fogarty catheter thrombectomy/embolectomy is carried out followed by gentle flushing with heparinized saline. The transverse arteriotomy is closed primarily with either a running or interrupted Prolene™ suture. A longitudinal arteriotomy should be closed with a patch angioplasty. Distal flow is assessed by pulse and Doppler examination or, if needed, completion arteriogram. If distal thrombus is present and needs to be removed, a medial longitudinal incision along the posterior aspect of the tibia below the knee provides access to the distal popliteal artery. The exposure of the proximal tibial vessels may be required for distal control and should be carefully performed to avoid injury to the popliteal vein and the tibial nerve (Fig. 20.4). A checklist for management of lower extremity acute ischemia due to an embolism originating from the heart is helpful for even the most experienced surgeon in the management of this problem (Fig. 20.2).
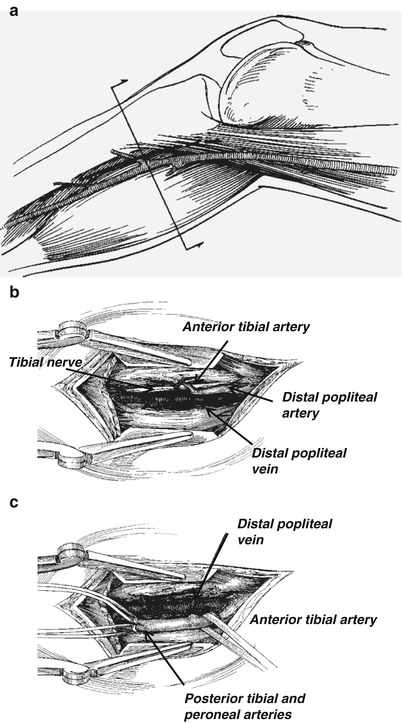

Fig. 20.4
(a) Incision for exposure vessels in the upper calf. (b) Dissection to expose distal popliteal artery. (c). Dissection to expose origins of tibial vessels
The treatment of phlegmasia cerulean dolens is based on prompt anticoagulation and catheter-directed thrombolytic therapy [26]. Pain management and hydration are also important. An inferior vena cava filter should be placed at the initiation of thrombolysis because of the risk of pulmonary embolism [40]. Open thrombectomy of the common femoral and iliac vein is very infrequently required [40]. At the time of opening the common femoral vein, careful use of a large Fogarty catheter in the iliac vein will retrieve distal thrombus. Wrapping the leg firmly with a sterile elastic bandage will milk out proximal thrombus. Retrograde Fogarty catheter passage down the veins of the leg results in significant damage to the valves and worsens the risk of recurrent thrombosis.
Fasciotomy
Failure to perform an adequate fasciotomy when indicated after revascularization of an acutely ischemic limb is the most common cause of preventable limb loss [37, 38]. Calf compartment syndrome is common, and forearm compartment syndrome is relatively rare in nontraumatic acute limb ischemia [37]. Calf fasciotomy, particularly in the setting of prolonged ischemia, must always be considered prior to completion of the operation. Intraoperative compartment pressure measurements may provide decision-making data [37, 38]. However, if normal pressures are initially obtained, eventual reperfusion edema and subsequent swelling may occur with delayed compartment syndrome. Serial postoperative compartment pressure measurements may be required. There are four compartments in the calf that need to be released. These include the anterior and lateral compartments on the anterolateral aspect of the calf and the deep and superficial posterior compartments (Fig. 20.5). The standard approach for release requires two incisions: one on the lateral side and other one on the medial side of the calf [41] (Fig. 20.6). Although isolated anterior compartment syndrome occurs in some settings, four-compartment release is usually required. A checklist for fasciotomy is strongly recommended and an example is included in Fig. 20.3.
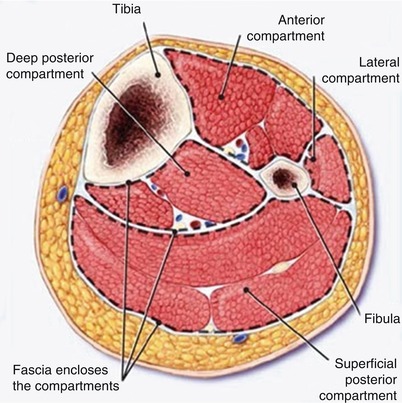
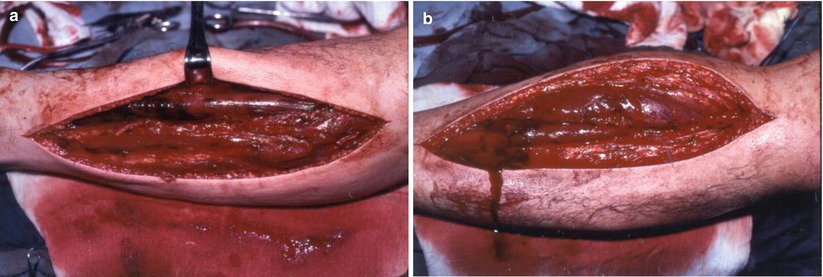

Fig. 20.5
Calf muscle compartments

Fig. 20.6
(a) Lateral incision for the release of anterior and lateral muscle compartments. (b) Medial incision for posterior and deep posterior compartments
The lateral calf incision should be generous. Start proximally no higher than three to four centimeters below the fibular head in order to avoid the superficial branch of the peroneal nerve. The incision should be taken distally to within three to four centimeters of the lateral malleolus. The fascia of both the anterior and the lateral compartments needs to be incised longitudinally along the muscular septum that separates the two compartments. One must avoid extending the incision beyond the limits of the skin incision proximally to avoid injury to the peroneal nerve. Make certain that the anterior compartment is fully released by palpating the tibia anteriorly under the fascia. Misplacing the incision lateral to the interosseous membrane can lead to not decompressing the anterior compartment with devastating consequences.
The medial incision should be made posterior to the midline of the medial side calf to avoid lacerating the greater saphenous vein. The fascia over the gastrocnemius should be fully incised proximally and distally. The gastrocnemius and soleus muscles are retracted posteriorly in the distal calf to expose the deep posterior fascia. This layer needs to be incised under direct vision to avoid lacerating the posterior tibial artery.
Once all four compartments are adequately released and hemostasis is obtained, a loose dressing is applied. Care should be taken to avoid tight dressings which can recreate the compartment syndrome when muscle swelling occurs. Subsequent wound closure is performed in 2–3 days or when edema has sufficiently resolved. Split-thickness skin graft may be required when a delayed primary closure is not possible.
Postoperative Considerations
Serial examinations after successful restoration of flow are essential to detect re-occlusion or compartment syndrome and promptly treat these limb-threatening complications. These patients usually have significant comorbidities, and postoperative care should include a period of monitoring in the intensive care unit or a specialized telemetry unit. The nursing staff needs to have training and experience in monitoring the perfusion of the distal extremity. Any evidence of recurrent ischemia, operative site hemorrhage, or compartment syndrome should prompt an immediate return to the operating room. In patients with cardiac source embolism, systemic anticoagulation should be initiated no sooner than 12 h after the operation was finished. The risk of a recurrent embolism outweighs the risk of operative site bleeding. Continuous heparin infusion without a bolus is recommended instead of weight-based subcutaneous fractionated heparin. In the early postoperative period, it is best to preserve the ability to reverse the intravenous infusion of heparin with protamine if needed because of bleeding complications. Once the patient is stable, the subcutaneous fractionated heparin every 12 h and oral warfarin can be administered. Lifelong anticoagulation is recommended in patients with cardiac source embolism.
Acute Mesenteric Ischemia
Acute mesenteric ischemia is a potentially lethal process that requires prompt recognition and treatment for successful management. The mortality rate remains more than 50 %, and there is little room for either delay or errors in management [42, 43]. Symptoms vary from the insidious onset of vague generalized abdominal pain to the sudden onset of severe and constant pain. There are four common causes: cardiac embolism to the superior mesenteric artery, acute thrombosis of a previous partial occlusion from an atherosclerotic lesion, splanchnic vasoconstriction leading to low flow and regional ischemia known also as non-occlusive mesenteric ischemia, and mesenteric venous thrombosis (Table 20.3). Each of these causes is secondary phenomenon which results from other major diseases and occurs in a high-risk setting [ 44, 45] (Table 20.4).
Table 20.3
Etiology of mesenteric ischemia
50 % | Arterial embolism |
20 % | Arterial thrombosis |
20 % | Small vessel occlusion |
10 % | Venous thrombosis |
Table 20.4
Risk factors for mesenteric ischemia
Arterial embolism or thrombosis |
Cardiac disease: |
Atrial fibrillation |
Recent myocardial infarction |
Congestive heart failure |
Digitalis therapy |
Previous arterial emboli |
Hypercoagulable state |
Hypovolemia, shock |
Venous thrombosis |
Portal hypertension |
Intra-abdominal inflammation |
Trauma or major bowel surgery |
Prothrombotic state |
Chronic renal failure |
Pathophysiology
The arterial blood supply of the gut is divided into four areas defined by the arteries which supply them. Collateral perfusion exists for each area (Table 20.5) [ 46, 47, 53]. In the absence of pre-existing occlusive disease and collateral vessels, the collaterals are not sufficient to provide adequate flow if the superior mesenteric artery is acutely occluded. In chronic mesenteric occlusive disease from atherosclerosis, patients may have total gut perfusion via a single remaining mesenteric artery or the bilateral hypogastric arteries via collateral flow to the other vessels [53]. However, many of these patients have intestinal angina when eating large meals. The venous drainage of the gut is via the portal venous system. Gastric venous drainage is via the splenic vein. The small bowel and the proximal colon through the splenic flexure drain via the superior mesenteric vein. The descending colon drains via the inferior mesenteric vein. Collateral venous vessels are also present and connect each major area.
Table 20.5
Gut regions, their blood supply, and collateral connections
Region | Blood supply | Collateral connections |
|---|---|---|
Foregut Distal esophagus through the ampulla of Vater in the duodenum | Celiac artery | Pancreaticoduodenal arteries and arc of Buhler distally |
Midgut Ampulla of Vater region of the duodenum to splenic flexure of the colon | Superior mesenteric artery | Pancreaticoduodenal arteries and arc of Buhler proximally, marginal artery of Drummond and Arc of Rioland distally |
Hindgut Splenic flexure of the colon to distal sigmoid colon | Inferior mesenteric artery | Marginal artery of Drummond and Arc of Rioland proximally Superior hemorrhoidal to middle hemorrhoidal arteries distally |
Cloacal derivatives | Branches of the bilateral hypogastric arteries | Middle hemorrhoidal to superior hemorrhoidal arteries proximally |
Cardiac source emboli have a predilection to enter the orifice of the relatively large superior mesenteric artery and then typically lodge distal to the origin of the proximal jejunal branches and the middle colic artery [53]. This gives rise to a pattern of small intestine and colon ischemia with sparing of the proximal jejunum and perfusion of the transverse colon and distal colon. Celiac artery emboli are less common as are emboli to the inferior mesenteric artery, and hypogastric artery emboli rarely cause ischemia due to a variety of pelvic collaterals [53].
The clinical manifestations of mesenteric ischemia are the result of insufficient blood flow to meet the metabolic demands of the bowel [46, 47]. The onset of acute ischemia leads to initial hyperperistalsis with gut emptying and vomiting and diarrhea. This is accompanied by intense ischemic pain from gut wall ischemia. This visceral pain is vague and projected across the area of the superficial abdominal wall depending upon the area of visceral innervation [46, 47]. Foregut structures generate pain in the epigastrium, midgut in the periumbilical regions, hindgut in the infraumbilical region, and cloacal derivatives (rectum and genitourinary organs) in the suprapubic region of the abdomen [53]. Visceral ischemia pain is intense and constant and does not increase with palpation nor is it associated with abdominal wall rigidity. This gives rise to the pathognomonic “pain out of proportion to physical findings” attributed to acute mesenteric ischemia [46, 47]. The initial vomiting and diarrhea frequently divert the examining physician’s attention to consider other diagnoses 4 [36, 47]. Ultimately, when ischemia leads to necrosis, inflammation of the gut surface leads to abdominal tenderness and associated physical findings of peritonitis. At the point of intestinal infarction, a systemic inflammatory response is initiated with an extremely high associated mortality rate [36, 37]. Elderly patients with this complication typically have cardiopulmonary and other comorbidities which further limit their ability to recover.
Clinical Presentation and Diagnosis
Acute intestinal ischemia from sudden embolic occlusion of the superior mesenteric artery causes the classical findings outlined above. Acute or chronic occlusion of a pre-existing atherosclerotic lesion may lead to a more insidious onset of pain because of pre-existing collateral flow which mitigates the severity of the resulting ischemia [7, 46–48, 51]. There may be a history of intestinal angina (postprandial pain), fear of food, and weight loss. The least common etiology, mesenteric venous thrombosis, causes an insidious onset of initially vague symptoms which worsen progressively over time [54, 55]. Non-occlusive acute mesenteric ischemia from vasoconstriction occurs in the setting of critical illness with reduced cardiac output and is associated with vague symptoms or undetectable symptoms in the intubated patient on a critical care unit.
Atrial fibrillation is the most common etiology of embolism [7, 46, 47]. Patients usually report sudden onset of pain associated with nausea, vomiting, and diarrhea. Mild abdominal distension and hypoactive bowel sounds without abdominal tenderness are the most common initial findings. In patients with acute worsening of chronic mesenteric ischemia, there is frequently a history of postprandial pain, and weight loss occurs in close to 90 % of patients (Table 20.6). Mesenteric venous thrombosis usually is associated with congenital or acquired hypercoagulability and a variety of comorbidities (Table 20.4) [54, 55, 58].
Table 20.6
Common symptoms and findings in patients with chronic mesenteric arterial occlusive disease
1. | Postprandial pain | 100 % |
2. | Weight loss | 85 % |
3. | Abdominal bruit | 70 % |
4. | Nausea, vomiting | 60 % |
The laboratory studies of patients with acute mesenteric ischemia are initially normal except for an often profound leukocytosis [7, 46]. The white blood cell count is often in excess of 20,000. This finding is an important early indicator and, when present, should prompt the inclusion of acute intestinal ischemia in the differential diagnosis. Metabolic acidosis is a late finding and usually indicates intestinal infarction. Hyperamylasemia and elevated serum lipase may also be seen early in the course of mesenteric ischemia [ 42, 43, 46, 59].
The best hope for an early diagnosis of acute mesenteric ischemia in patients at risk is a promptly performed CT scan of the abdomen with intravenous contrast. [58, 60, 61] (see Case Presentation 9).This exam definitively rules in the presence of mesenteric arterial and venous occlusion or indicates the presence of one of the other etiologies of these clinical findings. Early discovery with CT scanning allows timely diagnosis before significant bowel compromise occurs [42, 43, 60, 61]. End-stage intestinal necrosis causes severe shock and an overwhelming inflammatory response with an attendant high mortality rate [62, 63]. The outcome of acute mesenteric ischemia has not significantly improved in the last four decades because of the high incidence of both intestinal infarction at the time of diagnosis and the common associated significant comorbidities [42, 43, 46, 64, 65]. The only hope to improve outcome remains prompt recognition, early diagnosis, and successful management prior to the development of bowel necrosis [42, 43, 46].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





