Fig. 63.1
Axial contrast-enhanced CT showing a mildly enlarged pancreas (A), with normal contrast enhancement and peripancreatic fat stranding and fluid (B), consistent with acute interstitial edematous pancreatitis
These recommendations acknowledge that initial serum amylase is less sensitive for acute pancreatitis than serum lipase (especially in alcoholic and gallstone pancreatitis) and remains elevated for a shorter period of time than serum lipase, and is therefore not necessary to make the diagnosis [3, 4]. Sensitivity of lipase alone in diagnosing acute pancreatitis within the first day of onset has been estimated at 100 %, with specificity during this same time period around 84 % (lipase above the upper limit of normal) [4]. Lipase can be elevated in non-pancreatic pathology including kidney disease, appendicitis, cholecystitis, and other diseases of the gastrointestinal system, so diagnosis cannot be based on this laboratory value alone. Radiographic imaging is not necessary but can be helpful if the history and/or laboratory parameters do not confirm the diagnosis but suspicion for acute pancreatitis remains.
Principles of Management
Disease Burden, Diagnosis, and Assessment of Severity
Acute pancreatitis is the most common gastrointestinal diagnosis necessitating hospital admission, with US estimations in 2012 totaling 274,119 discharges, almost 1.5 million hospital days, and a total annual cost of $2.6 billion [5]. While estimations of risk of mortality due to acute pancreatitis are generally low (around 1 %) [5], significant morbidity can result from local complications including pancreatic pseudocyst and necrosis as well as systemic complications including sepsis, renal failure, and the acute respiratory distress syndrome. There are a number of different scoring systems to assess pancreatitis severity, and two commonly used systems are the Atlanta Criteria and more recently, the BISAP criteria [6]. The Atlanta Criteria have long been used to assess severity in acute pancreatitis, and most recently were updated in 2012 (Table 63.1) [2, 7]. While they are easy to use and can assist in triage and decisions regarding specialist consultation and/or referral, they do not predict outcomes. The BISAP criteria were more recently presented, and have the advantage of being simple to calculate, with only one subjective measure and are able to predict mortality (Table 63.2) [6].
Table 63.1
Pancreatitis severity, revised atlanta criteria
Mild acute pancreatitis | Moderately severe acute pancreatitis | Severe acute pancreatitis |
|---|---|---|
No organ failure and no local complications | Local complications and/or transient organ failure (less than 48 h) | Persistent organ failure (greater than 48 h), may involve one or more organs |
Table 63.2
BISAP criteria
BISAP score | In-hospital mortality (percent) |
|---|---|
0 | 0.1 |
1 | 0.2 |
2 | 1.6 |
3 | 3.6 |
4 | 7.4 |
5 | 9.5 |
Intravenous Fluids
There are no approved pharmacologic treatments for acute pancreatitis, and the only intervention that has strong data to suggest reduced morbidity and mortality is aggressive fluid resuscitation [8]. The optimal timing, dose, and monitoring strategy are still not agreed upon, and there is little prospective data to inform recommendations [9]. One of the few prospective studies to investigate volume resuscitation in acute pancreatitis was a four arm trial of forty patients that compared goal-directed fluid therapy with normal saline, goal-directed fluid therapy with Lactated Ringer’s (targeting BUN decrease or normalization), standard fluid therapy with normal saline, and standard fluid therapy with Lactated Ringer’s. Mean volume administered in both goal-directed and standard therapy groups was similar, and there was a significant decrease in the systemic inflammatory response syndrome and C-reactive protein at 24 h in the Lactated Ringer’s groups compared to the normal saline groups, but no difference in length of stay, infection, organ failure, or death [10]. Reviews and professional societies recommend variations in dosing, however a common practice is to provide 250–500 ml/h isotonic crystalloid solution for the first 12–24 h in the absence of limiting cardiac and renal comorbidities [1].
Antibiotics
Routine antibiotics in acute pancreatitis are not recommended [1]. A recent meta-analysis of six clinical trials of patients with necrotizing pancreatitis found that prophylactic antibiotic use was not associated with reduced risk of infected necrosis or mortality, but did find a statistically significant reduction in hospital stay [11]. ACG recommends empiric antibiotics only in extrapancreatic infection (including cholangitis and bacteremia), no antibiotics for sterile necrosis, and consideration of antibiotics in patients with necrosis who fail to improve after 7–10 days of supportive management, with consideration of CT-guided fine-needle aspiration to guide therapy, using antibiotics only if there is a positive Gram stain or culture [1].
Pain Control
Analgesia in acute pancreatitis is typically achieved with intravenous opioids. Data surrounding optimal drug, route, dose, and monitoring has not been established. A recent Cochrane review on the topic concluded the existing evidence does not support any one practice for pain control over another and additional evidence should be pursued [12].
Etiology
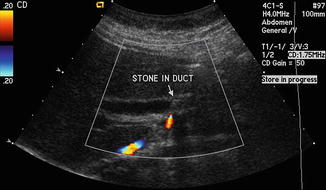
The most common etiologies of acute pancreatitis are alcoholic and gallstone, while hypertriglyceridemia, post-endoscopic retrograde cholangiopancreatography, acute human immunodeficiency virus infection, and medications (commonly furosemide, vinca alkaloids, azothiaprine, and didanosine) are also frequent causes [13]. In the United States, recent estimations suggest that the etiology of acute pancreatitis is most commonly idiopathic (36 %), followed by gallstones (28 %), and alcoholic (19 %) [14]. In addition to taking a thorough history focusing on previous episodes of acute pancreatitis, alcohol use, history of gallstone disease, and medications, ACG recommends performing a transabdominal ultrasound in all patients with acute pancreatitis to assess for gallstone disease [1]. The Dutch Pancreatitis Study Group notes that abdominal ultrasound is widely available, noninvasive, and inexpensive, although it is not sensitive for common bile duct stones [15, 16]. In patients without another clear explanation for their pancreatitis and in whom abdominal ultrasound was negative for cholelithiasis, contrast enhanced computed tomography and/or magnetic resonance cholangiopancreatography should be considered, with the latter demonstrating higher sensitivity for smaller stones than the former (40 % vs. 80 %, respectively) [16]. Imaging should only be pursued if there is no clear etiology of the acute pancreatitis or the patient fails to improve clinically within 48–72 h of presentation [11]. In the absence of suggestive alcohol use or obstructing gallstone on imaging, serum triglycerides should be checked with >1000 mg/dL consistent with triglyceride-induced pancreatitis [1]. Risks and benefits of ERCP should be discussed in light of clinical suspicion of a radiographically occult obstructive stone or other foreign body in the biliary tree [1] Fig. 63.2, Table 63.3.