
FIGURE 125.1 Peptic ulcer disease. Endoscopic view of a gastric ulcer with a visible vessel (arrow) before (A) and after (B) endoscopic intervention with gold probe cautery.

FIGURE 125.2 Severe esophagitis. Endoscopic view of severe esophagitis.

FIGURE 125.3 Portal gastropathy and esophageal varices. Endoscopic view of (A) portal gastropathy, (B) a large esophageal varix at the gastroesophageal junction, (C) two banded esophageal varices. (Courtesy of Dr. Martin Cohen.)
Most SRMD lesions are asymptomatic and clinically insignificant, although some patients may develop clinically evident bleeding, presenting with hematemesis, coffee-ground emesis, melena, and hematochezia. Although clinically evident bleeding due to SRMD may occur up to 25% of critically ill patients who do not receive prophylactic therapy, more recent studies suggest that only a small proportion (1% to 6%) of patients have clinically significant bleeding, which is associated with an increased length of ICU stay and morbidity and mortality (18,24).
Severity of GI Bleeding
Determination of estimated blood loss is the single, most important aspect of care in patients with upper GI bleeding. This estimation helps with the aggressiveness of volume resuscitation and triage to an appropriate level of care (i.e., transfer to ICU). Most complications associated with blood loss result from the adverse effects of hypovolemia and hemorrhagic shock on other organs, and are compounded by the presence of pre-existing atherosclerosis or previous organ damage. Estimation of blood loss is often incorrect and requires an accurate assessment of vital signs, central venous pressure, hemoglobin, and a degree of clinical experience.
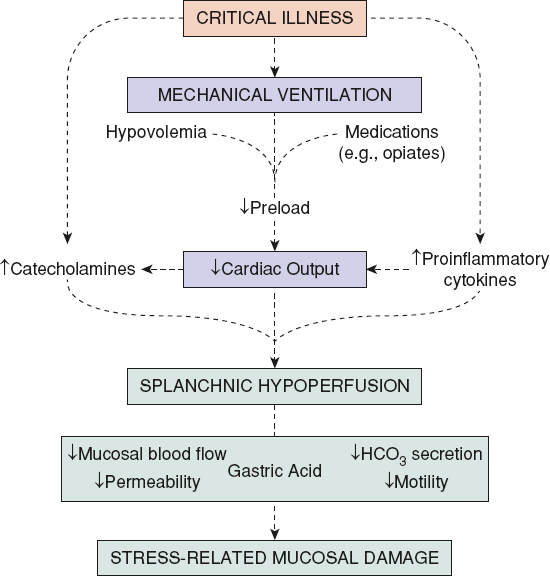
FIGURE 125.4 Mechanisms important in the pathogenesis of stress-related mucosa damage. Splanchnic hypoperfusion leading to diminished mucosal blood flow and other changes in gastrointestinal epithelial function plays a pivotal role in development of stress ulceration. (Adapted from Mutlu GM, Mutlu EA, Factor P. GI complications in patients receiving mechanical ventilation. Chest. 2001;119:1222–1241.)
Severe hemorrhage is usually defined as greater than 1,000 mL of blood loss. Initial hematocrit may be misleading due to loss of whole blood, which results in equal loss of plasma and erythrocytes. Redistribution of plasma from the extracellular to intravascular space, within 24 to 48 hours of the initial hemorrhage, results in dilution of red cell mass and a fall in hematocrit. The hematocrit fall may occur even more rapidly with volume replacement with crystalloid fluids.
Physiologic changes in the cardiovascular system in response to blood loss are helpful in determining the severity of GI bleeding. Acute responses to blood loss represent a spectrum of changes, including resting tachycardia, orthostasis, peripheral vasoconstriction (cold, clammy skin), and acute end-organ dysfunction (mental status changes, oliguria). Chronic blood loss is usually associated with stable hemodynamic responses, retention of hypotonic fluid, and an absence of impaired organ function due to compensatory changes in the cardiovascular system. Many factors may impair or unmask normal responses to blood loss, including drugs, pre-existing dehydration, oxygen desaturation from pulmonary disease, the state of the cardiovascular system (particularly atherosclerotic cerebrovascular disease), abnormal concentration of plasma proteins, and miscellaneous conditions such as spinal cord disease, neuropathy, renal dysfunction, shock, and congestive heart failure.
Gastric Lavage
Nasogastric lavage is important for confirmation of the diagnosis and may be predictive of a high-risk lesion if bright red blood is present in the lavage (25). A nasogastric tube may also decrease the risk of aspiration in patients with active hematemesis. A non-bloody nasogastric aspirate may be seen in up to 16% of patients with upper GI bleeding, usually if bleeding has ceased or if from a duodenal source, particularly if the pylorus is closed (26). Even the presence of bile in the aspirate is often (50%) misleading and does not necessarily rule out a postpyloric source of bleeding (26). Testing stool or emesis for blood during an acute episode of bleeding is not usually helpful (27). Testing of gastric contents can frequently be misleading because of nasogastric tube–related trauma. In addition, the low pH of gastric contents may interfere with the guaiac test’s design for occult blood testing in stool, giving false results.
Diagnostic Testing
Upper GI endoscopy is the diagnostic modality of choice for further evaluation of upper GI bleeding (10,28). Endoscopy helps in localization and identification of the bleeding lesion in the upper GI tract, and can be therapeutic in establishing hemostasis and preventing recurrent bleeding. Upper GI studies with radiocontrast materials such as barium are contraindicated in the setting of acute upper GI bleeding due to interference with subsequent endoscopic intervention, angiography, and surgery (10).
Other diagnostic tests for workup of upper GI bleeding include angiography and tagged red blood cell scan (29). Angiographic diagnosis of the source of upper GI bleeding is made by extravasation of contrast material. The bleeding must be brisk, with a rate of about 0.5 to 1 mL/min. Angiography can be helpful in establishing the diagnosis in 75% of patients; about 85% of bleeding originates from a branch of the left gastric artery (30). Nuclear medicine studies using 99mTc-pertechnetate-labeled red blood cell scan may also aid in localization of the bleeding site and has the ability to detect bleeding at lower rates (less than 0.5 mL/min) than contrast angiography.
Endoscopy is also the diagnostic method of choice for esophagogastric varices. In cases where endoscopy is nondiagnostic and gastric variceal bleeding is suspected, studies such as endoscopic ultrasound, portal venography, or computed tomography (CT) angiography can be used.
Risk Stratification for Rebleeding and Mortality
Prognostic scales are recommended for early stratification of patients into low- and high-risk categories for rebleeding (3,31). The risk stratification is based on clinical, laboratory, and endoscopic criteria. Clinical parameters that are independent predictors of rebleeding and mortality include older age (age greater than 65), hemodynamic instability, presence of comorbidities, hematochezia, fresh red blood in emesis, or gastric lavage (Table 125.2) (3,32). Laboratory tests that are associated with increased risk as low hemoglobin level at presentation and elevated BUN, creatinine and transaminases.
In addition to clinical and laboratory parameters, findings during endoscopic assessment provide further prognostic information and may guide subsequent management decisions (Table 125.3). Endoscopic predictors of increased risk for rebleeding and mortality include active bleeding, presence of nonbleeding visible vessel, adherent clot, ulcer size greater than 2 cm, location of ulcer (posterior lesser gastric curvature or posterior duodenal wall), and type of the lesion (ulcer vs. cancer) (3,33,34).
There are several scoring systems that have been developed to predict rebleeding and mortality. The Blatchford and Clinical Rockall scores use clinical and laboratory data to identify patients who need intervention while complete Rockall score uses endoscopic findings to predict rebleeding and mortality (35,36). A Blatchford scoring system has an excellent sensitivity (99% to 100%) for identifying severe bleeding and has been suggested to perform better than the Clinical Rockall score; its specificity is very low (4% to 44%) (37,38).
| TABLE 125.2 Clinical and Laboratory Predictors of High-Risk Upper GI Bleeding |
 |
TREATMENT
Initial evaluation of a patient with upper GI bleeding includes the assessment of hemodynamic stability and need for aggressive resuscitation, if necessary (3). Early stratification of patients into low- and high-risk categories for rebleeding and mortality is very important. All patients with hemodynamic instability or a hematocrit drop of more than 6%, a transfusion requirement greater than two units of PRBCs, or significant active bleeding as evidenced by continued hematemesis with bright red blood from gastric lavage or hematochezia should be admitted to ICU for close observation and resuscitation. Table 125.4 summarizes the general principles of management.
General Management
The initial management of upper GI bleeding should be directed at restoring blood and volume loss to maintain hemodynamic stability. Hemodynamic stabilization with adequate volume and blood resuscitation prior to endoscopic evaluation also helps to minimize treatment-associated complications (39). Intravenous (IV) access with two large-bore (14- to 16-gauge) catheters is preferred. In cases where peripheral IV catheters cannot be placed, a large-bore central line (9 to 12 French) should be inserted; 15-cm-long triple-lumen catheters have one 16- and two 18-gauge ports and should, therefore, be avoided due to unacceptably high resistance to flow. Close monitoring of end-organ perfusion (coronary, central nervous system, and renal) and preventing ischemic organ injury improve survival. In addition to a baseline electrocardiogram (ECG) and telemetry monitoring, urine output should be monitored continuously. In patients with massive hemorrhage or variceal bleeding, monitoring of preload via a central venous catheter may also be useful.
| TABLE 125.3 Endoscopic Findings that Predict Rebleeding in Peptic Ulcer Disease | ||
 | ||
| TABLE 125.4 Principles of Management of Upper GI Bleeding |
 |
Hypovolemia and hypotension should be treated promptly with a fluid resuscitation protocol using crystalloid solutions. Blood transfusion should be administered to patients with a hemoglobin less than 7 g/dL with a target level between 7 and 9 g/dL unless there is evidence for tissue hypoperfusion or acute coronary ischemia (40–43). In older patients with severe comorbidities such as ischemic heart disease or congestive heart failure, the transfusion target for hemoglobin may need to be individualized.
Current data on the correction of coagulopathy are conflicting. While coagulopathy should be corrected as needed, endoscopy should not be delayed unless international normalized ratio (INR) is supratherapeutic (39,44). There is no consensus on the target INR value, which may range between 1.5 and 2.5. Routine use of recombinant factor VIIa for variceal bleeding is not recommended as it does not provide survival benefit over standard treatment (45,46). Elective intubation should be considered in patients with ongoing hematemesis or altered mental status to facilitate endoscopy and decrease the risk of aspiration. Gastroenterology and, in cases of severe bleeding, surgery consultations should be requested.
Pharmacologic Therapy
Gastric Acid Suppression
Acid-suppressive therapy with proton pump inhibitors (PPIs) is an essential adjunct to therapeutic endoscopy for management of patients with peptic ulcer disease–related upper GI bleeding (47,48). High-dose PPI infusion for 72 hours following a bolus injection significantly reduces the rate of rebleeding and mortality in patients with high stigmata who underwent endoscopic intervention (47,49). Other acid-suppressing agents, including H2-receptor antagonists (H2RAs) should not be used as they have not been shown to reduce the rate of rebleeding or transfusion requirement in peptic ulcer disease (47,50). It is unknown whether oral PPI will provide benefits similar to IV PPI. The superiority of PPI over H2RAs has been attributed to better maintenance of gastric pH above 6.0, which may lead to clot stabilization by prevention of fibrinolysis, and thus rebleeding (51).
Other Pharmacologic Therapies
Splanchnic vasoconstrictors are important adjunct therapies to variceal hemorrhage. The current agent of choice in the United States is the somatostatin analogue, octreotide. Somatostatin and its analogues inhibit the release of vasodilator hormones such as glucagon, thereby indirectly causing splanchnic vasoconstriction and decreased portal inflow. For octreotide, the recommended dose is a 50-μg IV bolus followed by an infusion of 50 μg/hr for 5 days. Additionally, a longer-acting analogue of vasopressin, terlipressin, may also be used. Its efficacy is similar to octreotide and endoscopic sclerotherapy (52). Octreotide may also be considered as an adjunct therapy in the management of nonvariceal bleeding (53).
Cirrhotic patients with upper GI bleeding should be administered a prophylactic antibiotic (oral or IV quinolone or IV ceftriaxone) preferably prior to endoscopy and continued for 7 days to decrease the risk of bacterial infection (54,55). If a vasopressor is needed temporarily for the maintenance of blood pressure, medications that have β2-adrenergic activity, such as dopamine or albuterol, should be avoided due to potential risk of splanchnic vasodilation.
Endoscopic Treatment
Identification and hemostasis of the source of upper GI bleeding is critical in the patient’s outcome. Early upper endoscopy within the first 24 hours of presentation is recommended and is the standard of care for patients with upper GI bleeding (3,34). Early endoscopy leads to significant reduction in the length of hospital stay in both low- and high-risk patients compared to delayed endoscopy (34).
Endoscopy should be performed as soon as possible in patients who present with hemodynamic instability because endoscopic intervention in patients with major bleeding may reveal high-risk findings, such as varices, ulcers with active bleeding, or visible vessel that may be amenable to endoscopic hemostatic therapy. On the contrary, early endoscopy increases costs without a change in outcomes in patients with low-risk clinical and endoscopic features. Thus, patients with clean-base ulcers, nonbleeding Mallory–Weiss tears, and erosive or hemorrhagic gastropathy who have no hemodynamic or hemoglobin instability and no other medical problems can be considered for discharge to home.
Several endoscopic therapeutic techniques have been developed to achieve hemostasis in nonvariceal upper GI bleeding, including injection therapy (e.g., epinephrine or sclerosants such as ethanol, polidocanol), cautery, and mechanical therapy (e.g., clips) (Fig. 125.5) (56). The two most commonly used techniques in the United States are injection therapy with epinephrine (1:10,000) and thermal contact devices, which stop bleeding by producing vasoconstriction of bleeding vessels and coagulation, with subsequent destruction of bleeding vessels, respectively. Combination therapy with injection and thermal coagulation are superior to monotherapy in patients with high-risk endoscopic stigmata (34,56). Visible vessels should be treated with thermal coagulation, whereas endoscopic intervention is not required for low-risk lesions such as a clean-base ulcer or a flat spot.

FIGURE 125.5 Endoscopic management of upper gastrointestinal bleeding. Bleeding Dieulafoy lesion before (A) and after (B) endoscopic intervention (clipping). (Courtesy of Dr. Sri Komanduri.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








