CHAPTER 180
Tick Bite, Tick Removal
Presentation
The patient arrives with a tick attached to the skin (Figure 180-1), often the scalp. He is often frightened or disgusted and concerned about developing Lyme disease, Rocky Mountain spotted fever (RMSF), or “tick fever.” Alternatively, the patient may only have a history of having removed a tick within the past week or so and now has developed a spreading erythematous rash at the previous site of attachment (Figure 180-2, A to C). By this time, systemic signs and symptoms consisting of myalgia, arthralgia, fever, headache, and fatigue may be present.
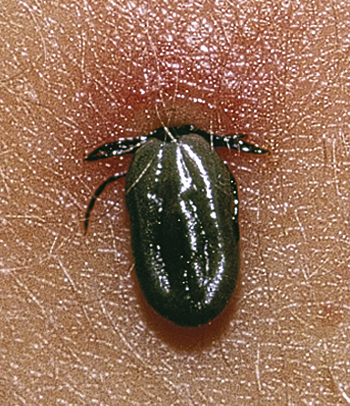
Figure 180-1 Embedded tick Ixodes pacificus (western blacklegged tick). (From White G, Cox N: Diseases of the skin, ed 2. St Louis, 2006, Mosby.)
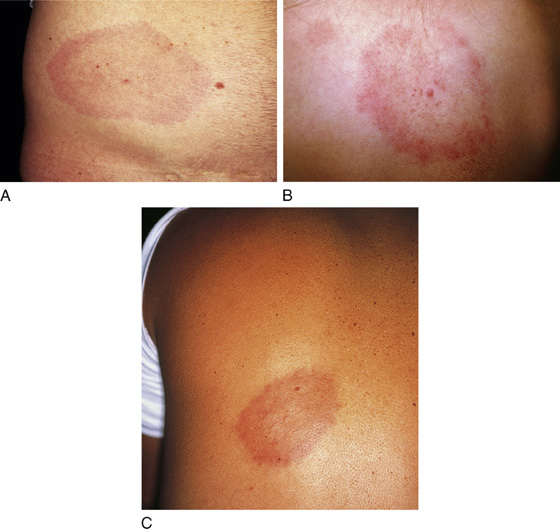
Figure 180-2 A to C, Erythema migrans. The causative organism is Borrelia burgdorferi.(From White G, Cox N: Diseases of the skin, ed 2. St Louis, 2006, Mosby.)
What To Do:
 When there is no tick present, obtaining a history of tick exposure in the recreational, occupational, and travel history can be essential for diagnosis.
When there is no tick present, obtaining a history of tick exposure in the recreational, occupational, and travel history can be essential for diagnosis.
 Carefully examine the patient for an individual tick or multiple ticks. Ticks can attach themselves to any part of the body, but certain species appear to prefer particular locations. Dog ticks may favor the head and neck, while the Lone Star tick may favor the lower extremities, buttocks, and groin. Although tick bites can be painful, the ticks are often not detected when they crawl, attach, feed, or depart from human skin.
Carefully examine the patient for an individual tick or multiple ticks. Ticks can attach themselves to any part of the body, but certain species appear to prefer particular locations. Dog ticks may favor the head and neck, while the Lone Star tick may favor the lower extremities, buttocks, and groin. Although tick bites can be painful, the ticks are often not detected when they crawl, attach, feed, or depart from human skin.
 When an embedded tick is present, apply protective gloves if available, and promptly remove the tick. Grasp the tick as close to the skin as possible with a pair of narrow-tipped forceps and slowly but firmly pull straight up until the tick mouth parts separate from the skin (Figure 180-3). Be careful not to squeeze the tick’s body or twist the tick’s head. Alternatively, the tick’s jaws may be pried away from the skin using a 20-gauge needle tip as a wedge.
When an embedded tick is present, apply protective gloves if available, and promptly remove the tick. Grasp the tick as close to the skin as possible with a pair of narrow-tipped forceps and slowly but firmly pull straight up until the tick mouth parts separate from the skin (Figure 180-3). Be careful not to squeeze the tick’s body or twist the tick’s head. Alternatively, the tick’s jaws may be pried away from the skin using a 20-gauge needle tip as a wedge.
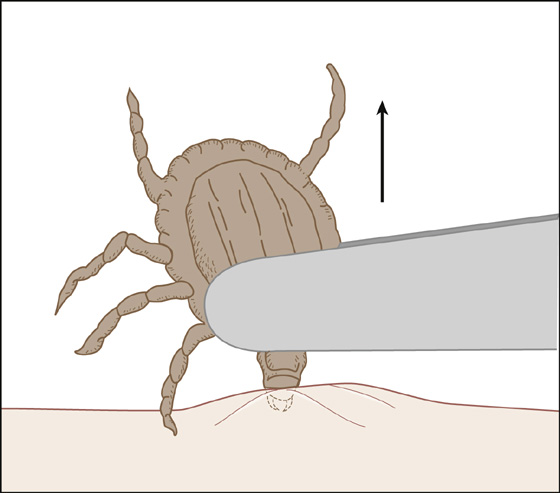
Figure 180-3 Remove tick with narrow-tipped forceps.
 Although not proven, it has been suggested that by injecting and infiltrating lidocaine with epinephrine beneath the attached tick, removal by traction is made easier. Also, covering the tick with 2% viscous lidocaine has led to spontaneous detachment within 5 minutes in a very small series.
Although not proven, it has been suggested that by injecting and infiltrating lidocaine with epinephrine beneath the attached tick, removal by traction is made easier. Also, covering the tick with 2% viscous lidocaine has led to spontaneous detachment within 5 minutes in a very small series.
 After removal of the tick, disinfect the attachment site and wash your hands with soap and water. Save the tick in a container of alcohol for future identification, or flush it down the toilet after it has been properly identified.
After removal of the tick, disinfect the attachment site and wash your hands with soap and water. Save the tick in a container of alcohol for future identification, or flush it down the toilet after it has been properly identified.
 If the mouth parts appear to remain embedded, anesthetize the area with an infiltration of 1% Xylocaine, and use the tip of a No. 15 scalpel blade to scrape (dermabrade) these fragments away.
If the mouth parts appear to remain embedded, anesthetize the area with an infiltration of 1% Xylocaine, and use the tip of a No. 15 scalpel blade to scrape (dermabrade) these fragments away.
 If it is an Ixodes scapularis or deer tick (Figure 180-4) (or Ixodes pacificus, see Figure 180-1), but it has been attached for less than 72 hours and was not engorged, there is minimal risk for disease transmission. There is no need for prophylaxis. The patient or family can be reassured, but instruct them to record the patient’s temperature daily for the next 4 weeks and to notify a physician or return at the first sign of a fever. In addition, over the same time period, instruct the family to watch for a pink patch at the site, which could be the beginning of erythema migrans. Also, if any rash, fever, cranial or peripheral neuropathies, or systemic symptoms, such as headache, myalgia, malaise, sweats, chills, or joint pains (“flu symptoms”) occur, have them return to evaluate for possible tick-borne illness, and initiate treatment as noted later.
If it is an Ixodes scapularis or deer tick (Figure 180-4) (or Ixodes pacificus, see Figure 180-1), but it has been attached for less than 72 hours and was not engorged, there is minimal risk for disease transmission. There is no need for prophylaxis. The patient or family can be reassured, but instruct them to record the patient’s temperature daily for the next 4 weeks and to notify a physician or return at the first sign of a fever. In addition, over the same time period, instruct the family to watch for a pink patch at the site, which could be the beginning of erythema migrans. Also, if any rash, fever, cranial or peripheral neuropathies, or systemic symptoms, such as headache, myalgia, malaise, sweats, chills, or joint pains (“flu symptoms”) occur, have them return to evaluate for possible tick-borne illness, and initiate treatment as noted later.

Figure 180-4 Life-sized deer tick. (From Gorman C: Deer tick turns deadly. Time, July 24, 1995, p 56.)
 If an Ixodes tick has been attached for less than 72 hours, but the tick appears to be engorged with blood, treat prophylactically for Lyme disease with doxycycline, 200 mg PO in 1 dose. Patients who are very anxious and cannot be reassured may also be given a single preventative dose.
If an Ixodes tick has been attached for less than 72 hours, but the tick appears to be engorged with blood, treat prophylactically for Lyme disease with doxycycline, 200 mg PO in 1 dose. Patients who are very anxious and cannot be reassured may also be given a single preventative dose.
 If this was an Ixodes tick that was attached for more than 72 hours, the patient presents with a focal rash at the site where the tick had been attached, or there are signs and symptoms as noted earlier, prescribe antibiotics to prevent or treat early Lyme disease (see later, this chapter). In a patient with typical erythema migrans, in an endemic area, laboratory confirmation is not required. Doxycycline, 100 mg bid × 2 to 3 weeks, is the agent of choice. For pregnant or nursing women and children younger than 8 years of age, amoxicillin, 500 mg tid or 25 to 50 mg/kg/day in divided doses for 2 to 3 weeks, can be used. Cefuroxime (Ceftin), 500 mg bid for adults and 250 mg bid for children for 2 to 3 weeks, is an alternative for patients who are allergic to doxycycline and amoxicillin.
If this was an Ixodes tick that was attached for more than 72 hours, the patient presents with a focal rash at the site where the tick had been attached, or there are signs and symptoms as noted earlier, prescribe antibiotics to prevent or treat early Lyme disease (see later, this chapter). In a patient with typical erythema migrans, in an endemic area, laboratory confirmation is not required. Doxycycline, 100 mg bid × 2 to 3 weeks, is the agent of choice. For pregnant or nursing women and children younger than 8 years of age, amoxicillin, 500 mg tid or 25 to 50 mg/kg/day in divided doses for 2 to 3 weeks, can be used. Cefuroxime (Ceftin), 500 mg bid for adults and 250 mg bid for children for 2 to 3 weeks, is an alternative for patients who are allergic to doxycycline and amoxicillin.
 If a wood tick (in the western United States) or a dog tick (in the eastern United States) has been removed (Figure 180-5), reassure the patient and family that the likelihood of developing Rocky Mountain spotted fever (RMSF) is very small (1%), and that if it should occur, prompt treatment will be quite effective on development of fever and/or headache. It is not recommended to give prophylactic antibiotics in an attempt to prevent RMSF. If transmission of infection has occurred, within 1 to 2 weeks after the tick bite, acute onset of fever, chills, severe headache, and myalgias will develop. Patients should seek immediate care at the onset of their symptoms. In most patients, fever and severe headache precede the characteristic rash that generally appears on the fourth day, starting on the wrists, ankles, and forearms as blanching red macules that progress to form papules centrally to the arms, thigh, trunk, and face. Therapy should be started when the disease is suspected and should never be delayed for confirmatory tests. Tetracyclines and chloramphenicol are the only drugs proven to be effective for the treatment of RMSF. Because of its effectiveness, broad margin of safety, and convenient dosing schedule, doxycycline is currently considered the drug of choice for nearly all patients, including young children. The current recommended regimens of treatment with doxycycline are 100 mg per dose given twice daily for adults, and 2.2 mg/kg body weight per dose given twice daily for children weighing less than 45 kg. These recommended doses may be given orally or intravenously, and treatment should be maintained for 5 to 7 days. Doxycycline therapy should be continued until the patient is afebrile for at least 2 or 3 days. Intravenous therapy is often indicated for hospital inpatients, particularly for those with vomiting, unstable vital signs, and neurologic symptoms. Chloramphenicol remains the recommended therapy for RMSF in pregnant women, despite the risk of grey baby syndrome. The indicated dose of chloramphenicol is 50 to75 mg/kg/day, divided into four doses, given for 7 days, or until 2 days after the fever has subsided. In life-threatening situations, the use of tetracyclines might be warranted during pregnancy.
If a wood tick (in the western United States) or a dog tick (in the eastern United States) has been removed (Figure 180-5), reassure the patient and family that the likelihood of developing Rocky Mountain spotted fever (RMSF) is very small (1%), and that if it should occur, prompt treatment will be quite effective on development of fever and/or headache. It is not recommended to give prophylactic antibiotics in an attempt to prevent RMSF. If transmission of infection has occurred, within 1 to 2 weeks after the tick bite, acute onset of fever, chills, severe headache, and myalgias will develop. Patients should seek immediate care at the onset of their symptoms. In most patients, fever and severe headache precede the characteristic rash that generally appears on the fourth day, starting on the wrists, ankles, and forearms as blanching red macules that progress to form papules centrally to the arms, thigh, trunk, and face. Therapy should be started when the disease is suspected and should never be delayed for confirmatory tests. Tetracyclines and chloramphenicol are the only drugs proven to be effective for the treatment of RMSF. Because of its effectiveness, broad margin of safety, and convenient dosing schedule, doxycycline is currently considered the drug of choice for nearly all patients, including young children. The current recommended regimens of treatment with doxycycline are 100 mg per dose given twice daily for adults, and 2.2 mg/kg body weight per dose given twice daily for children weighing less than 45 kg. These recommended doses may be given orally or intravenously, and treatment should be maintained for 5 to 7 days. Doxycycline therapy should be continued until the patient is afebrile for at least 2 or 3 days. Intravenous therapy is often indicated for hospital inpatients, particularly for those with vomiting, unstable vital signs, and neurologic symptoms. Chloramphenicol remains the recommended therapy for RMSF in pregnant women, despite the risk of grey baby syndrome. The indicated dose of chloramphenicol is 50 to75 mg/kg/day, divided into four doses, given for 7 days, or until 2 days after the fever has subsided. In life-threatening situations, the use of tetracyclines might be warranted during pregnancy.

Figure 180-5 Dog ticks showing normal morphology (upper photo) and after being fully engorged with blood (lower photo).(From Bolognia J, Jorizzo J, Rapini R: Dermatology. St Louis, 2003, Mosby.)
 Provide information about prevention of tick exposure. Measures to help prevent tick exposure include avoiding tick-infested areas (especially during the summer months); avoiding the grassy, overgrown areas favored by ticks; staying to the center of a trail while hiking; and avoiding sitting on logs or leaning against trees. Other measures include wearing long pants and tucking pant legs into socks, using tick repellents containing diethyltoluamide (DEET) for exposed skin and permethrin (Duranon) for clothing, and using bed nets sprayed with permethrin when sleeping on the ground or camping. Wearing light-colored clothing and checking the skin carefully at the end of the day for ticks (especially the head, scalp, and genital area—”tick checks”) will help in spotting a tick before it bites. Special graspers for removing ticks are available and can be handy in the field (e.g., Tick Nipper).
Provide information about prevention of tick exposure. Measures to help prevent tick exposure include avoiding tick-infested areas (especially during the summer months); avoiding the grassy, overgrown areas favored by ticks; staying to the center of a trail while hiking; and avoiding sitting on logs or leaning against trees. Other measures include wearing long pants and tucking pant legs into socks, using tick repellents containing diethyltoluamide (DEET) for exposed skin and permethrin (Duranon) for clothing, and using bed nets sprayed with permethrin when sleeping on the ground or camping. Wearing light-colored clothing and checking the skin carefully at the end of the day for ticks (especially the head, scalp, and genital area—”tick checks”) will help in spotting a tick before it bites. Special graspers for removing ticks are available and can be handy in the field (e.g., Tick Nipper).
What Not To Do:
 Do not use heat, occlusion, or caustics to remove a tick. Many techniques have been promoted, but they are generally ineffective and may increase the chance of infection or may potentially do harm.
Do not use heat, occlusion, or caustics to remove a tick. Many techniques have been promoted, but they are generally ineffective and may increase the chance of infection or may potentially do harm.
 Do not contaminate fingers with potentially infected tick products.
Do not contaminate fingers with potentially infected tick products.
 Do not mutilate the skin by attempting to remove the tick’s “head.” Usually what is left behind is cementum secreted by the tick, which is easily scraped off. Retained mouth parts (Figure 180-6) can produce local inflammation or minor bacterial infection, but they do not transmit Lyme disease.
Do not mutilate the skin by attempting to remove the tick’s “head.” Usually what is left behind is cementum secreted by the tick, which is easily scraped off. Retained mouth parts (Figure 180-6) can produce local inflammation or minor bacterial infection, but they do not transmit Lyme disease.
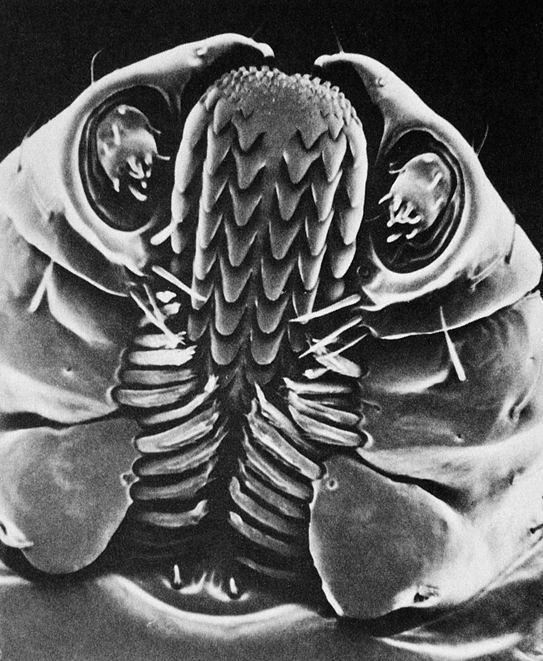
Figure 180-6 Tick mouth parts.
 Do not prescribe prophylactic antibiotics for RMSF.
Do not prescribe prophylactic antibiotics for RMSF.
 Do not prescribe prophylactic antibiotics for Lyme disease if its prevalence is very low in your geographic area (Figure 180-7).
Do not prescribe prophylactic antibiotics for Lyme disease if its prevalence is very low in your geographic area (Figure 180-7).
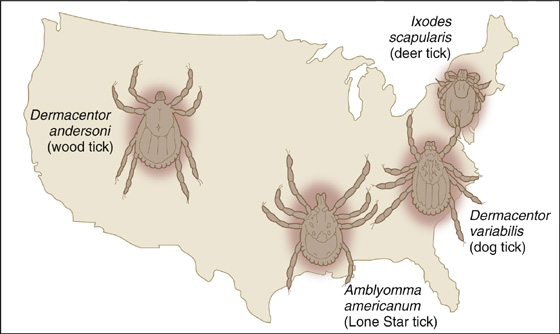
Figure 180-7 Ticks by region.
 Do not initiate serologic testing in the asymptomatic patient with suspected Lyme disease or early in the course of the disease. It is costly, inaccurate, and unnecessary. Serologic testing may be more useful in later stages of the disease, when sensitivity and specificity of the test is improved.
Do not initiate serologic testing in the asymptomatic patient with suspected Lyme disease or early in the course of the disease. It is costly, inaccurate, and unnecessary. Serologic testing may be more useful in later stages of the disease, when sensitivity and specificity of the test is improved.
Discussion
Ixodes scapularis (previously I. dammini) (Figure 180-8), the tiny deer tick of the eastern United States, and I. pacificus of the western United States (Pacific coast states) can carry babesiosis, human granulocytic ehrlichiosis, and Lyme disease. Most patients presenting in the summer with a viral syndrome have just that; however, there are other important considerations. Tick-borne diseases may present in this fashion. The clinician must maintain a high index of suspicion in the appropriate epidemiologic setting. Treatment with doxycycline or tetracycline is indicated for RMSF, Lyme disease, ehrlichiosis, and relapsing fever. In patients with clinical findings suggestive of tick-borne disease, empirical treatment should not be delayed. The same tick may harbor different infectious pathogens and may transmit several with one bite.

Figure 180-8 Three phases in the life cycle of Ixodes scapularis, or deer tick. (From Marzouk JB: Tick-borne diseases: where to expect and how to detect such bite-caused syndromes. Consultant 25:21-44, 1985.)
Ticks are divided into three families, only two of which are capable of causing infection: soft ticks (Argasidae) and hard ticks (Ixodidae), the latter being responsible for most tick-related diseases. In the United States, the most common Ixodidae genera to transmit disease to humans include Amblyomma, Ixodes, and Dermacentor. Among the most common in the United States are I. scapularis (deer tick, formerly known as I. dammini), I. pacificus, Amblyomma americanum (Lone Star tick) (Figure 180-9), Dermacentor andersoni (American wood tick), and D. variabilis (American dog tick).

Figure 180-9 Lone star ticks. (From Bolognia J, Jorizzo J, Rapini R: Dermatology. St Louis, 2003, Mosby.)
Tick-borne diseases in the United States include Lyme disease, RMSF, ehrlichiosis, tularemia, babesiosis, Colorado tick fever, and relapsing fever.
Lyme disease is the most common vector-borne disease in the United States. It has been reported in 49 of the 50 United States, but most cases occur in the Northeast to Midwest and North Central regions of the United States. Nine states account for more than 90% of the nationally reported cases, with Connecticut leading the group. The other states (in decreasing order) are Rhode Island, New York, Pennsylvania, Delaware, New Jersey, Maryland, Massachusetts, and Wisconsin. The etiologic agent is the slow-growing, motile spirochete Borrelia burgdorferi, which is transmitted by ticks. Deer tick nymphs appear to be the most important vector for transmission of Lyme disease. This nymph stage is when they are tiny and likely to be missed. A minimum of 36 to 48 hours of attachment of the tick is required for transmission. Most cases occur between May and August, which corresponds with increased outdoor human activity and nymphal activity. The risk for developing Lyme disease after a tick bite is low, even in endemic areas. It should also be noted that less than half of affected patients recall receiving a tick bite because of the small size of the tick.
The incubation period is typically 1 week, but the rash might develop as late as 16 weeks after the tick bite. The rash develops centrifugally as an erythematous, annular, round to oval, well-demarcated plaque (erythema migrans) and has a median diameter of 15 cm. Typically oval or round in shape, this is variable, with a morphology that usually consists of a flat erythema. The phenomenon of central clearing, previously emphasized, actually occurs in a minority of cases. The color may be uniform or centrally darker, and there may be induration or vesiculation.
Within days to a few weeks after the infection, hematogenous and lymphatic dissemination of the spirochete to distant sites commonly occurs. Other annular plaques develop in up to half of patients. The most common neurologic feature is unilateral or bilateral facial paralysis (unilateral paralysis mimicking Bell’s Palsy (see Chapter 5). Late-stage disease can occur months to years after the initial infection.
The diagnosis of Lyme disease is usually based on the history of a tick bite in an endemic area, with characteristic clinical findings. Serology using enzyme-linked immunosorbent assay (ELISA) is the most common laboratory test to screen for antibodies; however, the test is not standardized, and false negatives, especially with active or recent infections (or, more commonly, false positives), are common. When a positive or equivocal test is obtained using ELISA, a Western blot (immunoblot) test should be performed on the same serum sample. If the immunoblot is negative, the ELISA is likely a false positive; if the immunoblot is positive, a diagnosis of Lyme disease can be confirmed in a patient with clinical evidence of Lyme disease.
One bout of Lyme disease will not confer immunity to future infections.
RMSF, the most common acute rickettsial infection in the United States, is caused by Rickettsia rickettsii. In the United States, the disease is most commonly seen in the Southeast, West, and South Central states. The infection typically occurs in spring and summer, and the exposure is usually in rural or suburban areas. Primary offenders in the western United States are the wood tick (D. andersoni), and, in the eastern United States, the dog tick (D. variabilis). The diagnosis of RMSF is based largely on clinical presentation in a patient with history of tick exposure. Various serology tests are available to confirm the diagnosis. When administered early in the disease, tetracycline and chloramphenicol (orally and intravenously) are extremely effective. The use of tetracyclines in the treatment of tick-borne rickettsial diseases in children was controversial in the past because of the risk of permanent tooth discoloration. Today, there is a consensus that doxycycline is the drug of choice for treating presumptive or confirmed RMSF in children of any age. A prospective study reported that children treated with doxycycline for RMSF did not show substantial discoloration of permanent teeth compared with those who had never received the drug.
Ehrlichieae are a group of small, gram-negative, obligate intracellular pleomorphic bacteria that are closely related to rickettsiae. In the United States, human monocytic ehrlichiosis (HME) has been seen mainly in the Mid-Atlantic, South Central, and Southeast states as well as in California. A. americanum (Lone Star tick) is the primary vector. Most cases occur during summer and autumn, and more than 80% of patients have a history of tick exposure. The clinical manifestations of HME are similar to those of RMSF, with a lesser likelihood of rash. In general, the treatment of choice for ehrlichiosis is a tetracycline. Human granulocytic ehrlichiosis (HGE) has been reported in the Northeast, the upper Midwest, and regions of northern California. Ticks of the I. persulcatus complex serve as the principal vector. Other principal vectors are I. scapularis (northeastern America) and I. pacificus (western United States). The clinical presentation for HGE is indistinguishable from HME, and the treatment is similar.
Tularemia is caused by Francisella tularensis, a short, gram-negative, nonmotile coccobacillus. In the United States, the disease is most commonly seen in southern and western states. Inoculation of organisms into the skin most frequently occurs from bites of deer flies or ticks and from direct contact with infected animals, primarily wild rabbits, especially after skinning their hides. The tick in the Southeast and South Central United States is A. americanum (Lone Star tick), and in the West, it is D. andersoni (wood tick); the most widely distributed is D. variabilis (dog tick). Tularemia is characterized by acute onset of fever, headache, chills, myalgias, fatigue, and leukocytosis. The typical incubation period is 3 to 5 days. The most common clinical presentation is the ulceroglandular type, which begins at the site of the tick bite, usually on the lower extremities or perineum, as a papule or nodule that rapidly ulcerates. A lymphatic spread occurs with painful regional lymphadenopathy, usually inguinal or femoral, which might progress to ulceration. Streptomycin given intramuscularly is the treatment of choice.
Babesiosis is a malaria-like disease caused by an intracellular parasite that invades red blood cells. The major endemic areas in the United States are Massachusetts (Martha’s Vineyard, Nantucket), New York (eastern and southern Long Island), Connecticut, and other offshore islands of the Northeast. The causative agent is Babesia microti, which is transmitted by the larvae of I. scapularis (deer tick). The illness usually occurs in older patients or in asplenic or immunocompromised patients. The incubation period after a tick bite is about 1 week. The classic clinical presentation is high fever, drenching sweats, myalgias, and hemolytic anemia. Blood smear may be confusing: The tetrad of merozoites of babesiosis can be confused with the ring forms of Plasmodium falciparum–induced malaria. A 7- to 10-day concomitant course of quinine and intravenous clindamycin is used most commonly to treat the infection.
Colorado tick fever is caused by an RNA orbivirus transmitted by the D. andersoni wood tick. It is predominantly found in the Rocky Mountain region. Influenza-like symptoms usually begin within 1 week after inoculation. The disease usually lasts 7 to 10 days. Treatment is supportive, but at the onset of symptoms, most patients will be treated empirically with tetracycline, doxycycline, or chloramphenicol to cover other tick-borne diseases.
Tick-borne relapsing fever is caused by the spirochete within the genus Borrelia. It is transmitted by species of the soft tick genus, Ornithodoros. The tick species capable of transmitting the disease in the United States tends to exist in remote undisturbed settings in most regions. After an incubation period of 1 week, the disease is characterized by acute onset of high fever with chills, headache, myalgias, tachycardia, arthralgias, and malaise. If untreated, the primary episode lasts 3 to 6 days; rapid defervescence followed by drenching sweats marks resolution of disease. If left untreated, a second shorter course and as many as three to five relapses per year can occur. Tick-borne relapsing fever warrants treatment with either tetracycline or erythromycin for 5 to 10 days.
Tick paralysis rarely occurs in humans; when it does, it usually affects children. Most cases in the United States occur in the Northwest in the spring or summer months, and the ticks usually attach to the scalp or neck. In the United States, most cases are attributed to wood ticks, dog ticks, Lone Star ticks, and deer ticks. Paralysis usually occurs 4 to 7 days after attachment of the tick and is caused by the production of a neurotoxin secreted in the saliva of the tick. An acute ascending lower motor neuron paralysis develops, beginning in the legs and sparing sensory function. Definitive therapy is simply to remove any ticks attached to the patient’s body. However, symptoms can progress after tick removal. In general, improvement is much faster in cases in North America than in Australia. Normally, once the tick is removed in the former setting, symptoms begin to rapidly resolve within hours and are completely resolved 24 hours later. Patients who are minimally symptomatic can be observed for several hours and discharged if no progression occurs. In Australian cases, where symptoms can dramatically progress after the tick is removed, patients should be observed for a longer period, until the clinical course is clearly steadily and unequivocally improving.
Fever and a rash following a tick bite can signify a true medical emergency. Recent findings show that Rocky Mountain spotted fever continues to be the most lethal tick-borne illness in the United States and is emerging as an important disease in South America. Other important emerging diseases include human anaplasmosis, southern tick-associated rash illness, human monocytic ehrlichiosis, and a variety of rickettsial fevers, including those caused by Rickettsia parkeri and Rickettsia amblyommii.
In summary, most tick-borne illnesses respond readily to doxycycline therapy. In the case of Rocky Mountain spotted fever, therapy should be started when the disease is suspected and should never be delayed for confirmatory tests. Accurate identification of tick vectors can help establish a diagnosis and can help guide preventive measures.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


