7 The role of clinical reasoning in the differential diagnosis and management of chronic pelvic pain
Evidence-based practice – Where did it come from? Where is it going?
Understanding pain: What do we need to know?
It’s about more than pain – Integrated systems for optimal health
The Integrated Systems Model for disability and pain: A framework for understanding the whole person and their problem
Introduction
1. Propositional, theoretical or scientific knowledge (Higgs & Titchen 1995), also known as declarative knowledge (Jensen et al. 2007);
2. Non-propositional or professional craft knowledge (knowing how to do something) (Higgs & Titchen 1995) or procedural (Jensen et al. 2007). Non-propositional knowledge also includes personal knowledge or knowing oneself as a person and in relationship with others.
Propositional, or declarative, knowledge refers to the content knowledge that one’s profession is based on and includes factual information derived from formal research trials. In addition, this category includes theoretical knowledge developed from existing empirical protocols and principles, derived from dialogue with professionals in the same discipline, and logic (Higgs 2004).
Most practitioners continue to take post-graduate courses or attend professional conferences to improve their knowledge pertaining to clinical theory and research (propositional) as well as their technical skills (non-propositional or craft); however, Rivett & Jones (2004) note that there is a tendency in both courses and conferences to neglect an essential component of daily clinical practice – clinical reasoning. How should the practitioner integrate into clinical practice the newly learned scientific and theoretical knowledge? Who is it appropriate for and when is the new skill appropriate to use? Clinical practice is, and always will be, a blend of science and ‘art’ with a healthy dose of logic and reasoning. Clinical expertise comes from reasoning, reflection, skill acquisition and the continual life-long pursuit of knowledge (propositional (declarative) and non-propositional (procedural and personal)) (Figure 7.1) (Jensen et al. 2007). This takes time, discipline and often mentorship and professional affiliation with both individuals and groups.
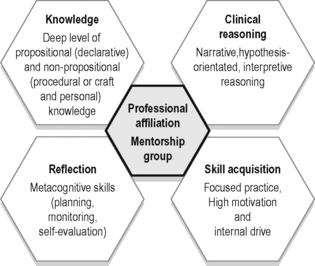
Figure 7.1 • Five components for the development of clinical expertise.
Adapted from Jensen et al. (2007) Expertise in Physical Therapy Practice, second ed. Saunders.
Evidence-based practice: Where did it come from? Where is it going?
The term ‘evidence-based’ was first used in 1990 by David Eddy and ‘evidence-based medicine’ by Guyatt et al. in 1992. The methodologies used to determine ‘best evidence’ were largely established by the Canadian McMaster University research group led by David Sackett and Gordon Guyatt. Professor Archie Cochrane, a Scottish epidemiologist, has been credited with increasing the acceptance of the principles behind evidence-based practice (Cochrane 1972). Cochrane’s work was honoured through the naming of centres of evidence-based medical research, Cochrane Centers, and an international organization, the Cochrane Collaboration. Since the early 1990s there has been an explosion of research evidence, and accessibility to this evidence has been facilitated for those involved in research or formal study through easy internet access to full-text articles in indexed journals. Unfortunately, access to full-text articles is still limited, or expensive, for clinicians not affiliated with research centres or universities.
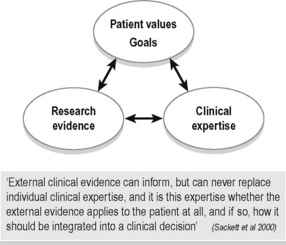
Evidence-based practice (EBP) embraces all disciplines of health care (not just medicine) and has become synonymous with best practice, but what does the term really mean? To some, it appears that EBP means that a clinician can only use assessment tests and treatment techniques/protocols that have been validated through the scientific process with high-ranking studies as valued by the ‘levels of evidence’. This is difficult to adhere to for many reasons, one being that there is not enough evidence at this time. Indeed, could there ever be enough scientific evidence for every situation met in clinical practice? Sackett and colleagues define EBP as ‘the integration of best research evidence, with clinical expertise and patient values’ (Sackett et al. 2000) (Figure 7.2). They note that:
Understanding pain: What do we need to know?
Understanding the neurophysiology of pain mechanisms is essential knowledge for treating patients with pelvic pain. Since the proposal of the gate control theory of pain by Melzack and Wall in 1965, significant advances in pain research and therapy have occurred. It is not our intent to provide an in-depth coverage of this topic here, but instead to highlight key features and establish a common language to be used throughout this chapter. See Chapter 3 for a full discussion of pain mechanisms in general, and as these relate to chronic pelvic pain.
What causes pain? Searching for the pain driver
It is now well recognized that the pathoanatomical model is limited in several ways. Pelvic pain commonly exists in the absence of any findings on diagnostic tests (X-ray, CT scan, blood tests, nerve conduction tests, etc.), and damaged tissues can be identified in people who experience no pain (Nachemson 1999, Waddell 2004). Tissues heal and yet the pain experience persists. Furthermore, a focus on only treating ‘the painful tissue’ neglects to consider that other systems or structures, which may be dysfunctional but painfree, could be the underlying cause of excessive mechanical stresses on the painful structures, or the cause of decreased blood flow or nutritional supply. In order to resolve the pain, the painfree but impaired structures or systems need to be treated for long-term resolution. Identification of what tissue hurts does not provide insight as to why it hurts. Finally, significant developments in neuroscience have changed our understanding of what pain is, and have required us to reframe and change our thinking.
We now understand that at any time in one patient there are many ‘pain drivers’ that do not exist solely in the peripheral tissues. Rather than looking for one source of pain, we need to consider that multiple mechanisms are at play in the experience of pain in all our patients. These mechanisms can be broadly separated into peripherally mediated (nociception and peripheral neurogenic pain) or centrally mediated (related to processing in the central nervous system (CNS)) (Butler 2000), and will be discussed in more detail later in this section.
Classifying pain
Timelines and mechanism of injury
Patients are commonly classified according to the timeline or duration of their pain experience, and the cause or mechanism of their injury. In general, problems are considered to be acute if they are within the first 6 weeks to 3 months (depending on the type of tissue injured) after an initiating incident (Brukner & Khan 2002, Magee et al. 2007). Tissue injury results in a known sequence of events aimed at protecting and repairing the damaged structures. These stages of tissue healing occur in three overlapping stages that have been given multiple names but refer to the same processes:
The term chronic is often used to indicate the persistence of pain beyond the normal timeline for tissue healing (Bonica 1953, Merskey & Bogduk 1994), as opposed to a stage of the tissue-healing process. In the Classification of Chronic Pain (Merskey & Bogduk 1994) published by the International Association for the Study of Pain, it is noted that the normal time of healing ‘may be less than one month, or more often, more than six months. With nonmalignant pain, three months is the most convenient point of division between acute and chronic pain, but for research purposes six months will often be preferred.’ Chronic pain is also further outlined as ‘a persistent pain that is not amenable, as a rule to treatments based upon specific remedies, or to the routine methods of pain control such as non-narcotic analgesics’ (Merskey & Bogduk 1994).
More recently, the term persistent low back pain has emerged in the literature, to indicate pain that continues past the expected timeframe for tissue healing. Others are suggesting that acute episodes of low back pain would be better termed recurrent episodes in a chronic problem as the underlying mechanisms contributing to recurrent low back pain are likely to be different from a first-time traumatic episode of low back pain, and recurrence of pain after an acute episode is a common problem (Pengel et al. 2003).
Thus, although the mechanism of onset and timeframes related to the pain experience are important to know, we must take care that this information does not lead us to assume that certain timelines necessitate certain pain mechanisms. Acute pain can be largely driven by central mechanisms. Persistent pain can also be largely driven by peripheral mechanisms. That is, persistent or chronic pain states may have central components, but these are not necessarily the dominant mechanism for every patient simply because the pain experience has persisted for a long period of time. While evidence supports that ‘the relationship between pain and the state of the tissues becomes less predictable as pain persists (Moseley 2007), we need to remember that the pain experience is uniquely individual. Regardless of whether the pain is a newly occurring event or a persistent experience, it is a multidimensional experience, and thus any person presenting with pain should be evaluated with a framework in mind that allows for the consideration of all these factors. As Butler (2000, p. 53) notes:
Classification by pain mechanisms
So what are the different biological mechanisms that drive the pain experience? Pain mechanisms can be further categorized (Gifford 1998, Butler 2000) as they relate to:
• Contextual factors of the immediate circumstance (i.e. how dangerous is this sensation in the light of environmental and internal factors?); as well as
• Past experiences and personal knowledge that collectively contribute to the individual’s beliefs, attitudes, emotions and physical responses.
Input mechanisms as they pertain to pain include all the sensory information reaching the CNS from the body internally and externally. This includes nociceptive pain from tissues including bones, ligaments, tendons, muscles, connective tissue, viscera, etc. (Gifford 1998, Butler 2000, Wright 2002) and peripheral neurogenic pain from neural tissue outside of the CNS. Processing occurs in the dorsal root ganglion and in the CNS. In the brain, an individual’s thoughts and feelings (cognitions + emotions = perception) are integrated and can influence the output mechanisms, which include:
1. Somatic or motor (altered posture, altered motor control);
2. Autonomic (increased sympathetic response for ‘fight or flight’);
3. Neuroendocrine (increased stress, heightened emotions, hormonal changes);
Thinking within the context of stress biology creates a broader framework for understanding pain. Gifford (1998), in proposing the Mature Organism Model (Figure 7.3), notes that
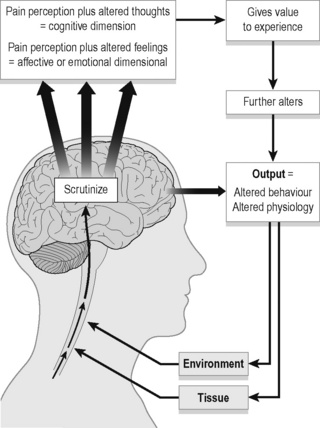
Figure 7.3 • The Mature Organism Model of Gifford (1998).
Adapted from Rivett & Jones (2004). Improving clinical reasoning in manual therapy. In: Jones, M.A., Rivett, D. (Eds.), Clinical reasoning for manual therapists, Churchill Livingstone; and Gifford (1998) Physiotherapy 84:27.
It has been proposed that continued activation of the stress-regulation systems and excessive or prolonged cortisol output has a destructive effect on peripheral tissues such as muscle, bone and nerve tissue, thereby perpetuating a vicious cycle of stress, pain and tissue injury (Melzack 2005).
Melzack’s model has four components (2001, 2005):
1. The body-self neuromatrix – an anatomical substrate in the brain of the body-self;
2. Cyclical processing and synthesis of nerve impulses which produces a neurosignature;
3. The flow of neurosignatures is projected back to areas of the brain, the sentient neural hub, which converts them into the flow of awareness;
4. Activation of an action neuromatrix occurs to provide the pattern of movements to bring about the desired goal.
Figure 7.4 is a modification of Melzack’s representation of the body-self neuromatrix to illustrate the sensorial, cognitive and emotional dimensions of pain. Perhaps the best summary for this section highlighting the broad view we need to take when considering pain comes from this leader in the study of pain himself, Ronald Melzack (2001):
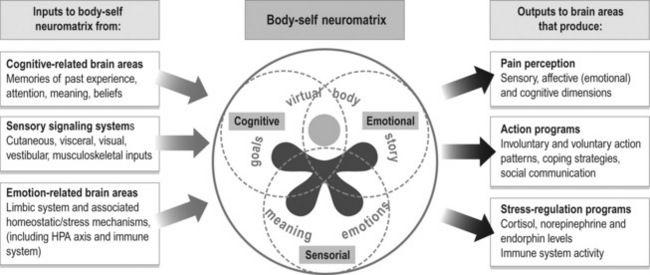
Figure 7.4 • An adaptation of Melzack’s Body-Self Neuromatrix (Melzack 2001, 2005). HPA, hypothalamic–pituitary–adrenal
Classification and clinical prediction rules: Are we searching for the holy grail?
Given the multidimensional nature of pain, it is not surprising that using pain presentation (location, duration, onset) as the sole means to classify patients and determine best treatment has been ineffective. Fritz and colleagues report that despite over 1000 randomized clinical trials investigating the effectiveness of interventions for the management of low back pain, ‘the evidence remains contradictory and inconclusive’ (Fritz et al. 2007). One key reason believed to contribute to this state of the evidence is the lack of classification of low back pain patients into subgroups, not only for studying treatment efficacy, but also for determining aetiological and prognostic factors (Leboeuf-Yde et al. 1997, Riddle 1998, Gombatto et al. 2007).
Sahrmann in the late 1980s noted:
The classification for lumbopelvic pain has evolved since the pathoanatomically based classification of MacNab (1977) with a variety of patient characteristics proposed for use in creating homogeneous subgroups (McKenzie 1981, Kirkaldy-Willis 1983, Bernard & Kirkaldy-Willis 1987, Coste et al. 1992, Delitto et al. 1995, Sahrmann 2001, O’sullivan 2005, Reeves et al. 2005, Fritz et al. 2007, O’sullivan & Beales 2007) (Table 7.1).
Table 7.1 Multiple proposals for the classification of patients
| Model/system | Description | Diagnostic/classification determinants |
|---|---|---|
| Pathoanatomical (McNab 1977, Kirkaldy-Willis & Hill 1979, Kirkaldy-Willis 1983, Nachemson 1999) | Focuses on structural changes which occur as a consequence of inflammation, infection, metabolic disorders, trauma and/or disease (pathology-based) | Radiological diagnosis, blood work |
| Mechanical diagnosis and therapy (The McKenzie Method) (McKenzie 1981) | Directional preference and centralization or peripheralization of pain with repeated movements. Four subgroups: | Rapidly reversible symptoms with repeated movements in a specific direction |
| Peripheral pain generator model (Laslett & Williams 1994, Laslett et al. 2005) | Attempts to identify the painful peripheral pain-generating structure with the main therapeutic intervention being to block or denervate the nociceptive source | Diagnostic blocks of various peripheral structures seeking to relieve pain |
| Neurophysiological pain model (Butler 2000) | Generation and maintenance of pain both peripherally and/or centrally mediated (central and/or peripheral sensitization of neural networks) | Subjective examination (confirmed/negated by features of the objective examination) |
| Psychosocial model (Waddell 2004) | Cognitive and emotional factors such as negative thinking, fear-avoidance behaviours and hypervigilance | Subjective examination |
| Treatment-based Classification System (Delitto et al. 1995) updated criteria (Fritz et al. 2007) | Intended for patients with acute/acute exacerbation of low back pain (LBP). Patients placed into treatment categories based on patterns of signs and symptoms: | Subjective examination, objective examination features based on clinical experience and propositional knowledge. Specific exercise grouping based primarily on centralization/peripheralization principles (McKenzie 1981). Updated criteria include disability questionnaire data and is based on CPRs and scientific research. Traction group removed in updated classification |
| Movement System Impairment System (Sahrmann 2001) | Based on the kinesiopathic model of movement; musculoskeletal pain develops as a result of repeated movements and postural alignments in the same direction across daily activities, causing repeated loading and microtrauma. LBP subgroups: | Subjective and objective examination aimed to identify the direction of movement and alignment that is related to LBP. Symptoms are monitored in response to standardized movement and alignment tests, along with observation of timing of relative motion of body segments, and the response to modification of alignment/movement |
O’sullivan (2005) noted that a limitation of many classification systems is that often only a single dimension (pathoanatomical, psychosocial, neurophysiological, motor control, signs and symptoms, etc.) is used to create subgroups. Classification systems will be most useful in clinical practice if variables across multiple domains are used to create subgroups.
• Presence or absence of identifiable underlying pathology (pathoanatomical, peripheral pain generator models);
• Pain presentation (central, unilateral, with or without radiation of symptoms to the lower extremity) (signs and symptoms models);
• Underlying pain mechanisms/neurophysiology;
• Response of pain to movement (centralization or peripheralization) (signs and symptoms models) (movement impairment models);
• Physical impairments such as loss or increase of mobility, altered motor control, altered posture/spinal alignment, and the relationship of symptom provocation to these impairments (motor control models, signs and symptoms models, movement impairment models);
• Response to specific treatments (manipulation, stabilization exercises, specific exercises, traction); and
• Psychosocial and cognitive features such as fear avoidance, coping strategies and beliefs (biopsychosocial models).
In recent years, the development of clinical prediction rules (CPRs) has emerged as another way to classify patients. CPRs are derived statistically with the aim of identifying the combinations of clinical examination findings that can predict a condition or outcome. Thus, they are proposed to be a useful tool to assist in clinical decision-making by improving the accuracy of diagnosis, prognosis or prediction of response to specific treatment protocols (Beattie & Nelson 2006, Cook 2008, Fritz 2009). Development of CPRs in physiotherapy has mainly focused on the response to treatment protocols (Fritz 2009) in order to identify subgroups of patients most likely to respond to a specific treatment approach. It is important to note that, at this time, CPRs are still in their infancy of development and validation, and are not yet at the appropriate stage to be widely applied in clinical practice (Cook 2008).
It has been suggested that CPRs will best impact physiotherapy practice where there is complexity in the clinical decision-making process, and that ‘an appeal of CPRs is their potential to make [the] subgrouping process more evidence based and less reliant on unfounded theories and tradition’ (Fritz 2009). However, the use of CPRs should be balanced with the knowledge that:
Consider the one domain of underlying pain mechanism as a way to create subgroups.
Butler (2000) notes that:
In their classification of pelvic pain disorders, O’sullivan & Beales (2007) categorize non-specific pelvic pain disorders into two groups: one that has centrally mediated pain, and one that has peripherally mediated pain. Although the group of centrally mediated pain is further classified into those with non-dominant psychosocial factors and those with dominant psychosocial factors, the treatment protocol for the subgroup of centrally mediated pelvic girdle pain is medical management (central nervous system modulation), psychological (cognitive-behavioural therapy), and functional capacity rehabilitation. Specific interventions directed at identified physical impairments in the periphery are not recommended, and yet it is highly unlikely that many patients will have 100% centrally mediated pain. In the authors’ experience, even in patients with a strong contributor of central sensitization to their pain experience, careful assessment often reveals specific meaningful tasks that relate to a consistent reproduction of symptoms. It is reasonable to suggest that even if peripheral mechanisms only contribute 20% to the complete picture, addressing that 20% in addition to the other approaches will provide the greatest chance for the best outcome. Furthermore, it is likely that by addressing the physical impairments, psychosocial variables will also be impacted, further advancing the goals of treating drivers of central sensitization. It is also crucial to recognize that our patients change as a result of their changing life circumstances and our interactions with them (both physical and personal). Thus, during the course of treatment continual re-evaluation is necessary to adapt the treatment programme accordingly. Sticking to a rigid plan based on an initial placement into a subgroup may result in the provision of sub-optimal care.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree