Electron microscopy of endothelial cells in the glomerular capillaries using ThO2 staining. The glycocalyx covers the endothelial cell surface in regularly distributed focal structures. The glycocalyx “bridges” the endothelial cell fenestrations. GBM, glomerular basement membrane.
The glycocalyx is a negatively charged, organized mesh of membranous glycoproteins, with core proteoglycans of the syndecan and glypican family carrying highly sulfated, linear glycosaminoglycan attachments (mostly of the heparan, chondroitin, and dermatan sulfate families). Hyaluronic acid and the negatively charged heparan sulfate proteoglycans are its major constituents.[1,2,3,5] This structure of core proteins “decorated” with long glycosaminoglycans provides a sea-grass-like surface where components of functional systems such as the coagulation cascade or the complement system can be located and plasma constituents may interact intensely and dynamically. This “habitat” on the surface of the endothelium has also been named an endothelial surface layer (ESL) and may attain a thickness up to 1 µm in certain blood vessels (Figure 11.2).
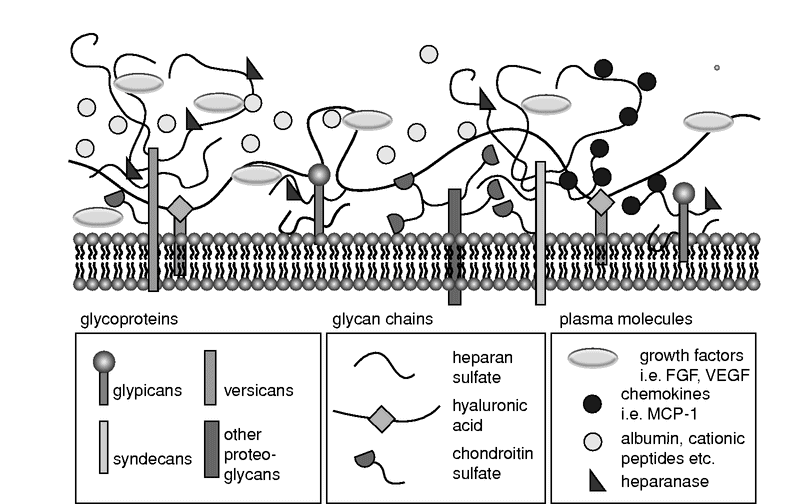
Schematic diagram showing the components and spatial organization of the endothelial glycocalyx. FGF, fibroblast growth factor; VEGF, vascular endothelial growth factor; MCP-1, monocyte chemoattractant protein-1.
It is obvious that this glycocalyx or ESL, whatever nomenclature we use, plays an important role both under physiological and under pathophysiological conditions. In this chapter we will briefly review the contribution of the glycocalyx for vascular permeability, as a mechanotransducer, and its role in inflammation. We will also discuss the methods to assess the glycocalyx in humans and describe under which pathophysiological conditions the glycocalyx is damaged and disturbed.
Glycocalyx and vascular permeability
The glycocalyx influences vascular permeability. The importance of the loss of glycocalyx barrier functions leading to increased vascular permeability was suggested from experiments showing that glycocalyx degradation is associated with a reduction in the exclusion of anionic dextrans, with an increased protein permeability, with an increased glomerular clearance of albumin, and with the formation of perivascular edema. The phenomenon has been mostly investigated in glomerular capillaries.[8] Under physiological perfusion conditions, albumin is confined to the glomerular capillary lumen and endothelial fenestrae, implying resistance at the level of glomerular endothelial surface.[9] Chappell et al. have demonstrated that shedding of the glycocalyx induced by the inflammatory cytokine tumor necrosis factor-alpha (TNF-α), or by ischemia-reperfusion, causes a substantial increase in vascular permeability to both plasma and colloids.[10] Selective removal of endothelial glycocalyx from coronary vessels increases permeability, which provides evidence that it constitutes a barrier to macromolecular permeability.[11]
This association between loss of glycocalyx and an increase in vascular permeability has been shown in animal models other than that of Chappell. Once removed, recovery of the hydrodynamically relevant glycocalyx required 5–7 days. It seems that the composition of the glycocalyx can be modulated by different factors. Salmon et al. have used angiopoietin-1 (Ang-1), a ligand for the tyrosine kinase receptor Tie-2, and were able to demonstrate that the increased permeability after digestion of glycocalyx was mitigated by pre-perfusion with Ang-1.[12] In addition, perfusion with Ang-1 nearly doubled glycocalyx thickness. Mice treated with glycocalyx-degrading enzymes show reduction in thickness of glomerular endothelial glycocalyx coinciding with increased albumin excretion. Therefore, the presence of a significant endothelial glycocalyx contributes significantly to the barrier to macromolecules. Loss or alterations of the glycocalyx may contribute to states of edema associated with endothelial dysfunction such as in infections and/or sepsis. Whether this is a property of the physicochemical characteristics of the glycocalyx, or whether an intact glycocalyx improves the barrier function of the endothelium, is as yet unclear.[5]
Glycocalyx as a mechanotransducer
To maintain vascular homeostasis, endothelial cells are endowed with a complex set of mechanisms to sense mechanical forces imparted by blood flow.[13] The concept that the glycocalyx contributes to the regulation of microvascular perfusion was originally hypothesized by the group of Duling in 1990, when they showed that the adenosine-induced increase in capillary tube hematocrit in hamster cremaster muscle vessels was diminished after enzymatic glycocalyx degradation.[14]
Based on the structure of the glycocalyx, with its regular grass-like distribution of the core proteoglycans, it has been assumed that these core proteoglycans bend under the influence of the bloodstream velocity and transmit signals to the intracellular cytoskeleton and/or other signaling complexes. The evidence that supports a central role for the glycocalyx in mechanotransduction comes from experiments involving degrading specific components of the glycocalyx, followed by a reassessment of function, mostly nitric oxide synthase (NOS) activation and/or NO production. Several glycosamine-degrading enzymes have been used.[15] Heparinase (which selectively degrades heparan sulfates), neuramidase (degrading sialic acid), and hyaluronidase (affecting hyaluronic acid) have been shown to reduce NO production and flow-dependent vasodilation significantly. Because flow-dependent vasodilation is mediated by NO release in many arteries, these studies suggest that glycosaminoglycans contributes to shear-induced production of NO. The core proteoglycans may also contribute to the mechano-sensing properties of the glycocalyx.
The different proteoglycans and their location on the cell mebrane seem to influence erent intracellular pathways. Both syndecan-1, a transmembrane proteoglycan that is linked to the cytoskeleton and mediates endothelial cell remodeling in response to shear stress, and the membrane-bound but not transmembranous proteoglycan glypican-1 that is enriched in caveolae near the intracellular endothelial nitric oxide synthase (eNOS) mediate vasodilation with shear-induced eNOS activation and cytoskeletal remodeling.[16] However, gene knockdown of glypican-1 blocked only eNOS activation, not remodeling, and knockdown of syndecan-1 blocked only remodeling, not eNOS activation, demonstrating that the glycocalyx can convert the mechanical signal shear stress into diverse cellular responses through distinct proteoglycans. Another possible mechanism for how the glycocalyx regulates vasodilation is the binding of substrates such as arginine to heparan sulfates. Heparan sulfates may play a role in the availability of arginine to its transporters close to the endothelial cell surface, thereby providing eNOS with its necessary substrate.[17]
Glycocalyx as a storage system
Heparan sulfates display high affinities for polycationic molecules. In addition, they provide a significant storage volume which is easily accessible from the fluid phase. Oberleithner et al. suggested that plasma sodium is stored in the glycocalyx, partially neutralizing the negative surface charges.[18] A “good” glycocalyx has a high sodium store capacity, but still maintains sufficient surface negativity at normal plasma sodium. A “bad” glycocalyx shows the opposite. Thus, proteoglycans and glycosaminoglycans (i.e. mainly extracellular polyanions) may provide potential sinks for sodium and may serve as a means of concentrating cations close to the plasma membrane.
Heparan sulfates adsorbed to a surface undergo a conformational change when exposed to flow: their core proteins unfold from a random coil to an extended filament, and their heparan sulfate chains elongate significantly. This finding was used to illustrate how sodium ions bound to heparan sulfates could not only be stored, but be delivered by the stretched glycosaminoglycan to their transporter channels. Such a concept of “storage space” in an intact glycocalyx has important implications for the binding of other cations to the heparan sulfates of the glycocalyx. Nucleic acids bind to the glycocalyx, and incorporation of DNA increases markedly in cells that lack the major anionic components of the glycocalyx, sialic acid and glycosaminoglycans, and anionic oligosaccharides may provide a barrier to the uptake of nucleic acids by mammalian cells. This phenomenon could also play an important role in the development of crystalloid solutions.
Glycocalyx and inflammation
The vascular endothelium is one of the earliest sites of injury during inflammation.[19] The glycocalyx and its glycosaminoglycans play an important role in various aspects of inflammation and in the physiological functioning of a range of inflammatory mediators, including chemokines, growth factors, endothelial adhesion molecules, and inflammatory cell emigration. A multitude of studies suggest that at least two mechanisms of the glycocalyx, a change in glycosamine composition as well as enzymatic disruption of the proteoglycans, may have a strong effect on inflammatory responses.[20]
A change in glycosamine composition of the glycocalyx affects the early inflammatory mechanisms in several ways: van der Vlag and his group have demonstrated that an early response of the glycocalyx to inflammatory stimuli is a change in the N-deacetylase-N-sulfotransferase-mediated composition of the heparan sulfate. They have argued that modulation of heparan sulfates in the endothelial glycocalyx significantly reduces or enhances the inflammatory response in inflammatory kidney disease.[20]
The binding properties of the glycosaminoglycans play a second important role in inflammation. A hallmark of immune cell trafficking is directional guidance via gradients of soluble or surface-bound chemokines. Vascular endothelial cells produce, transport, and deposit either their own chemokines or chemokines produced by the underlying stroma. Endothelial heparan sulfate has been suggested to be a critical scaffold for these chemokine pools. Stoler-Barak et al. have shown that the blood vasculature creates steep gradients of heparan sulfate scaffolds between the luminal and basolateral endothelium, and that inflammatory processes can further enrich the heparan sulfate content near inflamed vessels.[21] They proposed that chemokine gradients between the luminal and abluminal sides of vessels could be generated by these sharp heparan sulfate scaffold gradients in the glycocalyx. In addition, certain chemokines require interactions with glycosaminoglycans for their in vivo function. Proudfoot has shown that chemokines oligomerize on immobilized glycosaminoglycans, and this ability to form higher-order oligomers has also been shown to be essential for the activity of certain chemokines in vivo.[22]
The binding properties of the glycocalyx also play a role in the regulation of the complement system, which is a major player in innate immunity. One of the most important regulators of the complement cascade is complement factor H (CFH). The surface-bound glycocalyx glycosaminoglycan constituent heparan sulfate is crucial for CFH binding and function, both in recognition of host tissue and prevention of spontaneous complement activation via the alternative pathway.[23] Most of the clinically relevant genetic mutations in CFH result in incorrect binding to heparan sulfate. Loss of the heparan sulfate binding site will results in impaired binding of CFH, thereby reducing inhibitory signals to the complement system, which increases its local activation.[24]
A second mechanism whereby glycocalyx affects and influences the complement system is via thrombomodulin (TM).[24] TM is a cell-surface-expressed transmembrane glycoprotein which was originally identified on vascular endothelium, and is by definition a part of the glycocalyx. It is a co-factor for thrombin binding that mediates protein C activation and inhibits thrombin activity. In addition to its anticoagulant activity, recent evidence has revealed that TM, especially its lectin-like domain, has potent anti-inflammatory function through a variety of molecular mechanisms. The lectin-like domain of TM plays an important role in suppressing inflammation independent of the TM anticoagulant activity. Loss of the glycocalyx proteoglycan TM is therefore associated with increased complement activity and enhanced inflammatory response.
The loss of constituents of the endothelial glycocalyx has been termed “shedding.” Shedding can range from selective cleavage of heparan sulfates to major alterations with removal of entire syndecan and glypican core proteins together with the attached glycosaminoglycan side chains. The shedding of the glycocalyx in response to inflammatory mediators such as cytokines and chemoattractants was found to occur in arterioles, capillaries, and venules under various experimental models of inflammation. The major enzyme responsible for shedding of proteoglycans is heparanase, which is the only known mammalian endoglycosidase capable of degrading heparan sulfate glycosaminoglycan. Enzymatic degradation of glycocalyx by heparanase profoundly affects numerous physiological and pathological processes.[25]
Besides heparinase, there are other molecules that can affect glycocalyx composition and shedding.[26] Based on mechanism of action, inflammatory mediators can directly affect endothelial cells that, in response, alter glycocalyx structures. Additionally, under inflammatory conditions, activated subsets of leukocytes, such as polymorphonuclear leukocytes, macrophages, and mast cells, release enzymes which then can contribute to degradation of the glycocalyx. Upon activation, inflammatory cells release a wide range of enzymes and reactive species that can contribute to glycocalyx damage. In particular, activated neutrophil granulocytes, the most abundant blood leukocytes in humans, can induce glycocalyx damage by producing reactive oxygen and nitrogen species and releasing proteases from their storage granules. Moreover, mast cells, a less abundant leukocyte subset, can release heparanase directly with a significant potential to disturb glycocalyx structure through the degradation of heparan sulfates.
The cleavage of heparan sulfates due to reactive oxygen, and the subsequent increase in macromolecular permeability, follows a similar pattern to that seen after treatment with glycocalyx-degrading enzymes. This suggests common pathways in the degradation of the glycocalyx. Moreover, the protein core of proteoglycans can also be subject to oxidation/nitrosation and these oxidative and nitrosative modifications at the level of proteoglycans could negatively affect glycocalyx integrity.
The loss of the glycocalyx uncovers membrane surface adhesion molecules providing a direct contact area between leukocytes and the endothelium, and thereby potentiating leukocyte adhesion to the vessel wall. It should also be appreciated that degradation of the glycocalyx by inflammatory mediators, and the release of its fragments into the circulation, can significantly contribute to the potentiation of inflammatory processes, starting and maintaining a potentially destructive feedback mechanism. The glycocalyx fragments that have been shed, which may be released after glycocalyx damage, are suggested to act as pro-inflammatory molecules with significant chemotactic properties.[27]
How to assess glycocalyx in patients
Visualization of the glycocalyx is no easy task. Recent advances in staining techiques and advanced electron microscopy techniques (serial block face scanning electron microscopy [SBF], focused ion beam milling scanning electron microscopy [FIB-SEM], transmission electron tomography [Tom-TEM]) have made a better analysis of the glycocalyx under experimental conditions possible.[6,7] However, to analyze the glycocalyx in patients is still a challenge. Basically two approaches are feasible. One can either try to visualize the glycocalyx by determining the area between the capillary wall and circulating blood cells in capillaries,[6,28,29] or one can measure the constituents of the glycocalyx in the bloodstream, assuming that an increase in glycocalyx components such as hyaluronan or syndecan reflect damge to the glycocalyx in patients.[30,31] Both methods have been used by several groups.
The first method to measure glycocalyx health in vivo is intravital microscopy utilizing the property of the endothelial glycocalyx to function as a barrier to maintain a certain distance between red blood cells and the endothelial cell membrane.[6,28,29] The method measures these features in sublingual capillaries and depends on the hypothesis that impaired glycocalyx barrier properties in the sublingual microcirculation are associated with changes in microvascular perfusion capacity in other vascular beds (Figure 11.3). The camera uses green light-emitting diodes (LED; 540 nm) to detect the hemoglobin of passing red blood cells. To determine the spatio-temporal variation in radial displacement of individual red blood cells into the endothelial glycocalyx in microvessels, sidestream dark field (SDF) imaging is used.

Sidestream dark field (SDF) imaging and data assessment in sublingual microvessels. A: Typical example of a single SDF image of the sublingual capillary bed. B, C: During each period a large number of capillaries are assessed. D: Cartoon depicting the median RBC column width, perfused diameter, and the PBR in a blood vessel. PBR, perfused boundary region; RBC, red blood cell.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




