KEY POINTS
The biologic response to surgery results in fluid, electrolyte, and systemic hormonal changes that must be considered in the ICU management of the surgical patient.
Nutritional support of the critically ill surgical patient must involve consideration of the effect of surgical stress on nitrogen balance and on insulin and blood glucose levels.
The hypercoagulable state that follows surgery warrants consideration of prophylaxis against thromboembolic complications, particularly in the ICU patient.
Prompt surgical control of the source of the pathology remains the most important goal in either bleeding or septic critically ill patients.
Surgery increases the demand on the cardiorespiratory system and the likelihood that temporary mechanical ventilatory assistance will be needed.
Pulmonary edema and atelectasis characterize perioperative respiratory failure; hypoventilation and aspiration also contribute.
Where possible, a reduction of pulmonary capillary hydrostatic pressure in the perioperative period improves gas exchange by decreasing lung water.
The concept of closing volume and its relationship to functional residual capacity is important in understanding perioperative atelectasis.
Risk factors for perioperative atelectasis include obesity, smoking, advanced age, anesthesia, recumbence, and incisional pain.
Diaphragmatic dysfunction is a major component of perioperative respiratory failure.
Preoperative assessment of respiratory function makes it possible to predict operative risk and to correct abnormalities before operation, particularly in the patient undergoing lung resection.
Early ambulation, physiotherapy, treatment of sepsis and shock, adequate analgesia, and early operative stabilization of fractures are key elements in the treatment and prevention of perioperative respiratory failure.
The critically ill surgical patient is at risk for developing all of the potential problems that afflict nonsurgical patients in the intensive care unit (ICU). In addition, there are factors unique to the surgical patients that warrant special consideration if management is to be appropriately directed in the ICU environment.
Surgical stress or injury stimulates an orchestrated biologic response1-4 aimed at preserving the milieu intérieur. This response includes the elaboration of adrenocortical hormones, catecholamines, and glucagon; a decrease in insulin release resulting in hyperglycemia; and the secretion of antidiuretic hormone (ADH) and aldosterone, as well as the release of cytokines and the stimulation of a hypercoagulable state.5-10 These responses affect the critically ill surgical patient in many ways. Acute fluid and electrolyte shifts may occur, the renal response to volume infusion may be altered, and the catabolic response results in a phase of negative nitrogen balance.11,12 All these responses vary in intensity, depending on the magnitude and duration of the injury, the adequacy of resuscitation, and the presence of complications such as hemorrhage and sepsis. The increase in metabolic rate increases oxygen requirement and consumption. The management implications of these responses to surgical stress are outlined in the following sections.
CONSIDERATIONS IN CRITICALLY ILL SURGICAL PATIENTS
The glucagon and insulin response to injury can lead to major changes in glucose metabolism. Hyperglycemia may occur in a patient who has previously demonstrated no evidence of abnormality in glucose levels. This situation may also unmask a latent diabetic state in some patients, as well as complicating the management of already established diabetes mellitus in the critically ill surgical patient. Close monitoring of blood glucose, ketones, electrolytes, and acid-base status is essential for proper management of the surgical patient. Although strict glycemic control has been shown to significantly increase the risk of hypoglycemia and conferred no overall mortality benefit among the critically ill population in general, this therapy may be beneficial in patients admitted to a surgical ICU after elective procedures; this has not been confirmed by studies adequately examining this subgroup.13,14
Blood loss, pain related to surgical incisions, fasting prior to surgery, nausea or vomiting, and various drug administrations are only a few of the factors that predispose the surgical patient to release of ADH and aldosterone. The resulting sodium and water retention make it very difficult to monitor the state of hydration of the patient by relying entirely on urine volumes, since these hormones tend to decrease urine output in spite of normovolemia. Other indices of adequacy of perfusion, such as level of consciousness, capillary return, skin warmth, pulse, and blood pressure need to be assessed. In addition, the syndrome of inappropriate ADH release (SIADH) is relatively common in the postoperative period, placing patients at risk of water intoxication and severe hyponatremia when even modest water loads are administered. These problems can be largely avoided if treatment is guided by frequent routine monitoring of electrolytes and fluid volume status.
Following surgical trauma, occult fluid loss may occur at several sites, including the area of injury, where extravascular fluid may accumulate in the interstitial and intracellular spaces,15,16 as well as in the retroperitoneal space during intra-abdominal manipulation. In addition, operations involving the gastrointestinal (GI) tract or abnormalities resulting from surgical diseases such as peritonitis may result in decreased motility of the gut and sequestration of large volumes of fluid within the gut lumen, the gut wall, and the entire large surface area of the peritoneal cavity. This type of fluid depletes circulating blood volume and is not easily measured by most available clinical methods. In the patient with compromised cardiorespiratory reserve, close titration of fluid balance is crucial. In such patients, central hemodynamic monitoring may be required in addition to other clinical indices of normal perfusion and volume status.
The hypercoagulable state resulting from surgical trauma necessitates the institution of either pharmacological and/or mechanical thromboprophylaxis depending on the individual patient risk of bleeding as soon as possible, as outlined in other parts of this text.8,9 It is important to recognize that virtually every surgical patient is at risk for thromboembolic disease, and some are at extraordinarily high risk. Pharmacological prophylactic regimens pose a minor risk of bleeding, but can be employed in most surgical patients.
Although there is an early phase of negative nitrogen balance following surgical stress, it may be shortened or even aborted by appropriate nutritional support before and immediately after surgery.17 Early institution of enteric feeding has been shown to be of benefit, including a reduction in septic sequelae, in surgical patients undergoing intra-abdominal procedures.18 If daily caloric intake goal could not be achieved with enteral feeding alone before day 8 of ICU admission, institution of parenteral nutritional support should be considered to prevent further loss of muscle mass.19 Earlier initiation of parenteral nutrition appears to be associated with more infectious complications, delayed recovery, and higher health care costs.19 Adequate nutrition affects not only the maintenance of muscle mass, but also the maintenance of respiratory function, and thus both dependence on ventilatory support and weaning from mechanical ventilatory assistance.
Since the duration and magnitude of surgical procedures affect the intensity of the metabolic and endocrine response, the aim should be to decrease the magnitude, duration, and frequency of surgical insults to the critically ill patient, particularly patients with poor nutritional and cardiorespiratory reserve. This goal, however, must be considered in the context of the underlying problem. The magnitude and duration of the surgical procedure should not be minimized at the expense of incomplete eradication of a surgical lesion, such as a source of sepsis, since failure to eradicate the septic focus would lead to further complications, such as respiratory failure and dependence on mechanical ventilation in the ICU. In the setting of the multiply injured patient requiring massive blood transfusions that can lead to hypothermia, coagulopathy, and severe cardiorespiratory and renal compromise, “damage-control laparotomy” or abbreviated laparotomy should be considered. This consists of rapid control of hemorrhage (by ligation of vessels and packing) and removal of gross contamination followed by temporary closure, which should be followed as soon as possible by more definitive procedures as improvement in the patient’s condition in the ICU allows.20
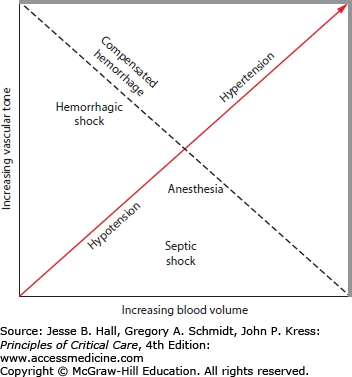
Hypotension is commonly encountered in the acutely ill surgical patient. Resuscitation of such patients should take into consideration the underlying pathophysiology, expediting surgical control and supporting organ system perfusion. Hypotension in such patients may be due to hemorrhage (eg, trauma), dehydration (eg, bowel obstruction), cardiac dysfunction, loss of vasomotor tone (eg, sepsis, spinal cord injury), or even mechanical issues (eg, tension pneumothorax, pericardial tamponade).21 Hypotension can be temporized by fluid administration but that is rarely a definitive cure. Temporary reversal of hypotension should never be mistaken for reversal of sepsis or control of hemorrhage.21 Figure 110-1 demonstrates the relationship between blood volume and the capacity of the vascular system. The ratio between the two in addition to the contractility of the heart determines the blood pressure. This relationship explains why blood pressure alone is not a good indicator of the shock state, because in the bleeding, vasoconstricted patient who may be suffering from profound occult hypoperfusion at the tissue level can have a normal blood pressure similar to that of a normal control. The goals of resuscitation include restoring the microcirculation, preventing clot disruption and thereby preventing rebleeding, and maintaining adequate perfusion pressure to the brain and other vital organs.
FIGURE 110-1
The relationship between blood volume and the capacity of the vascular system. Bleeding decreases blood volume but compensatory vasoconstriction restores blood pressure. The dotted line represents the isobaric state of preserved blood pressure in various states of shock. (Reproduced with permission from Fouche Y, Sikorski R, Dutton RP. Changing paradigms in surgical resuscitation. Crit Care Med. September 2010;38(suppl 9):S411-S420.)
Fluid administration can be beneficial to the patient who lost blood volume but is not actively bleeding. However, in the patient who is actively bleeding or who has formed early fragile clots, a fluid bolus may be deleterious despite an acute rise in blood pressure.21 This initial rise in blood pressure can potentially wash out early fragile clots and dilutes the circulating clotting factors necessary to stabilize the formed clots. Effective treatment includes definitive anatomical source control preceded by “controlled hypotensive resuscitation” until hemostasis is achieved.21 Early use of balanced mix of RBCs, plasma, and platelets may help to achieve early hemostasis and improve survival. For refractory hypotension despite aggressive fluid resuscitation, early judicial use of vasopressors, especially vasopressin that could have been depleted from the circulation with prolonged hemorrhage, should be strongly considered.21 In addition, early short-term use of the antifibrinolytic agent tranexamic acid has been shown to decrease mortality in significantly bleeding trauma patients.22
In contrast to hemorrhagic shock, early aggressive fluid administration in septic hypotensive patients have been shown to improve outcomes as a component of early goal-directed therapy during early efforts at source control. Fluid administration in the septic patient is thus a reverse image of the bleeding patient, with more value early than late.
In either bleeding or septic hypotensive patients, however, the most important principle is the surgical control of the source of the pathology, and nothing should delay the transfer to the operating room for more definitive treatment.21
The increase in metabolic rate following surgical stress is associated with an increase in oxygen requirement and utilization.23 In patients with a normal cardiorespiratory reserve, this increased oxygen demand is met without untoward sequelae. However, patients who are nutritionally depleted or whose cardiorespiratory function is already compromised may be unable to meet the increased oxygen demand. The result can be decompensation with anaerobic metabolism, muscle fatigue, and respiratory failure. In the high-risk patient, consideration should be given to providing temporary cardiorespiratory support during the phase of increased oxygen requirement. Such patients may require intubation and mechanical ventilation for short periods until the acute insult has abated.
The increased oxygen requirement of surgery and the postoperative state has caused some authors to recommend measuring and then maximizing oxygen delivery22 in critically ill patients. Use of invasive hemodynamic monitoring or gastric tonometry allows titration of catecholamine infusions, blood products, or other therapies to increase oxygen delivery to targeted supranormal levels. Many studies supporting this approach were likely flawed by inappropriate methodology, although there is some indication in high-risk surgical patients that outcome can be improved.23-25
Pain from surgical incisions may exacerbate the metabolic response to injury and mechanically restrict respiratory function, the latter potentially necessitating mechanical ventilatory assistance. Judicious parenteral analgesia as well as regional, intercostal, and epidural anesthesia may also be useful in these situations.
Neurosurgical patients and those who have suffered head trauma should be monitored closely for signs of increased intracranial pressure. If clinical signs cannot be easily elicited, an intracranial pressure monitoring device should be placed. Apart from the identification of mass lesions that may require evacuation, the mainstay of treatment for these patients is reduction of cerebral edema through the maintenance of cerebral blood flow and oxygenation and the avoidance of hypercapnia (see Chap. 86). The vascular surgery patient requires close monitoring of perfusion, particularly in the arterial territorial distribution of the repaired vessel. Skin temperature, Doppler ultrasound, and digital pressure measurements are required in the ICU setting to identify problems with vascular anastomoses as early as possible and permit early corrective measures. In the immediate postoperative period carotid artery surgery patients are at higher risk for developing neurologic complications from reocclusion at surgical sites of vascular repair, and careful monitoring of the neurologic status as well as maintenance of adequate oxygenation and perfusion are essential to avoid such complications.
Following GI surgical procedures, the gut should be used early for enteral nutrition whenever appropriate.18 When that is not possible and a prolonged period of decreased motility is anticipated, decompression of the GI tract is necessary to prevent distention and to monitor the volume and character of losses. This intervention, together with replacement of measured losses as well as determination of serum electrolytes, allows early identification and correction of abnormalities and the prevention of complications. Patients who develop sepsis following GI surgical procedures should be considered to have a source of that sepsis within the abdomen until proven otherwise. Early aggressive investigation for identifying such a source of sepsis is essential in order to avoid further deterioration in the ICU.
The gut has also been considered to play a major role in the pathogenesis of multiple organ dysfunction syndrome and decontamination of the GI tract has been suggested as a prophylactic measure in this syndrome, but there is a major concern about promoting the growth of resistant microorganisms by such intervention.26
Stress ulcers resulting in GI hemorrhage are a sequela not only of GI surgery, but of other forms of critical illness. Prophylactic measures against stress ulceration directed at decreasing acid injury to the gastroduodenal mucosa are an essential part of the management of critically ill patients.27
PERIOPERATIVE RESPIRATORY FAILURE
In the period before, during, and after surgical treatment, patients are unusually vulnerable to respiratory failure owing to special manifestations of pulmonary edema, atelectasis, hypoventilation, aspiration, sepsis, and hypotension. Awareness of the factors promoting each of these interrelated processes makes possible an effective prevention program or early diagnosis and treatment of perioperative respiratory failure.
Of the forces in the Starling equation governing transcapillary fluid flux, the ones of particular relevance to surgical patients are microvascular hydrostatic pressure and pulmonary capillary permeability.
Microvascular Pressure: As indicated, secretion of ADH and aldosterone is a major component of the metabolic response to surgery and trauma.27 Both hormones tend to conserve water and decrease urine output in the postsurgical patient. However, a focus on increasing urine output in the surgical patient by administering large volumes of fluid without regard to this metabolic response could easily result in fluid overload and pulmonary edema, leading to hypoxemia. Guidelines for fluid resuscitation in the perioperative period that focus primarily on urine output and fluid replacements based on empirical values28 can increase extravascular lung water and predispose the surgical patient to perioperative respiratory failure. Although a young, healthy patient with significant cardiopulmonary reserve might tolerate these insults, an elderly surgical patient with a brittle cardiorespiratory status is more likely to develop respiratory failure unless extreme caution is taken with fluid resuscitation, involving close, constant monitoring of central hemodynamics.
Pulmonary edema occurring in the head-injured and subarachnoid hemorrhage patients, or neurogenic edema, may be associated with a transient increase in hydrostatic pressure because of intense sympathetic discharge, although it has been suggested that there may be a component of increased capillary permeability as well in those patients.31 Therefore, monitoring of pulmonary capillary hydrostatic pressure can be helpful in determining therapeutic approaches to the head-injured patient with pulmonary edema.
High-pressure pulmonary edema does not resolve immediately after vascular pressures are normalized.32 The implication of this finding is that the timing of the measurement of pulmonary artery wedge pressure (PAWP), which is used as a reflection of pulmonary capillary hydrostatic pressure, is crucial in determining whether pulmonary edema is considered to be due to high vascular pressures or to an increase in capillary permeability. Ordinarily, the presence of normal or low PAWP in the presence of pulmonary edema would be regarded as evidence of capillary-leak pulmonary edema. However, this is not the case when the PAWP is measured during the lag phase of resolution of high-pressure pulmonary edema after PAWP has been decreased.
Diuretics such as furosemide clear edema by decreasing the central blood volume and pulmonary capillary hydrostatic pressure.30 However, these agents may produce effects on gas exchange before pulmonary edema has cleared.33 Accordingly, diuretic therapy and fluid management of the oliguric, hypoxemic perioperative patient may confuse the student of critical care at several levels. First, oliguria in the immediate postoperative period is not necessarily due to reduced blood flow to the renal cortex (prerenal oliguria), so fluid challenges aimed at increasing renal blood flow may not be appropriate in this setting. The consequent increase in pulmonary blood volume and pressure predictably increases pulmonary edema. On the other hand, diuretic therapy in such a patient will increase urine output, even when the oliguria is due to reduced renal blood flow, thereby aggravating the prerenal failure. The best approach to this common perioperative conundrum is to recognize that urine output may be an unreliable index of adequate perfusion in the immediate postoperative period, and to seek other indices of perfusion through careful history-taking, physical examination, and first-hand knowledge of the patient’s perioperative course. For example, a prior history of congestive heart failure predicts susceptibility to fluid overload, and thus should slow the physician’s hand in administering fluid. Similarly, familiarity with the patient’s preoperative blood pressure, heart rate, heart sounds, pulse volume, and digital perfusion allow the discerning physician to detect early signs of hypoperfusion requiring volume replacement. Often, the critical distinction between fluid overload and hypovolemia is not clear even to the astute clinician, and in such cases central hemodynamic measurements can be helpful.30 Alternatively, numerous noninvasive methods for assessment of volume responsiveness can provide valuable information. For example, trends over time in the respiratory variations in arterial pressure and stroke volume can provide important qualitative information on fluid responsiveness in patients receiving mechanical ventilation with no spontaneous breathing effort.35 In spontaneously breathing patients, examining the stroke volume (or its surrogates) response to a simple test like passive leg-raising has been shown to be a valuable approach.36 However, it needs to be emphasized that an increase in stroke volume in response to fluids does not mean that the patient needs this increase in his/her stroke volume.35
Pulmonary Capillary Permeability: A frequent cause of increased capillary permeability and respiratory failure in the surgical patient is unrecognized sepsis, which is commonly seen in the abdomen; this source often requires a surgical or percutaneous radiologic approach. Therefore, a major component of the prevention and treatment of respiratory failure in the surgical patient is the early identification of occult sources of sepsis, aggressive investigation for abdominal causes of sepsis, and the provision of adequate drainage and treatment of septic foci, particularly within the abdomen.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree