11.1 Soft tissue manipulation approaches to chronic pelvic pain (external)
Introduction
There is increasing evidence demonstrating the importance of treating muscle and connective tissue in patients with chronic pelvic pain (CPP). Eighty-five percent of patients with CPP present with dysfunction or impairments in the musculoskeletal system, including poor posture and pelvic floor muscle (PFM) imbalances (Baker 1993, Hetrick et al. 2003, Prendergast & Weiss 2003, Tu et al. 2006). Shoskes et al. (2008) found that 51% of men with chronic CPP reported tenderness to palpation of the PFM, and Tu et al. (2008) demonstrated that women with CPP had a greater prevalence of musculoskeletal disorders compared with women without CPP. Furthermore, tenderness to palpation of the PFM was related to a decreased ability to relax these muscles (Tu et al. 2008).
It is apparent that proper functioning of the pelvic region is directly related to appropriate integration of the connective tissue and muscles of the lower quadrant. The presence of musculoskeletal dysfunctions may contribute to improper functioning of the pelvic region from both a biomechanical and neurophysiological perspective; for example, increasing tension and/or shortening of the PFM (Haugstad et al. 2006), and initiation or maintenance of a neurogenic inflammation (Wesselmann 2001). In the animal model, Miranda et al. (2004) found that irritation of pelvic musculoskeletal structures promoted antidromic transmission of nociceptive inputs to bladder sensory neurons, promoting a state of neurogenic inflammation.
The diversity of clinical symptoms and physical findings found in patients with CPP emphasizes the necessity of multimodal approaches for the management of this patient population (FitzGerald & Kotarinos 2003a, Fox 2009), as outlined Chapters 8.1 and 8.2. Additionally, there is clinical and scientific evidence for suggesting that CPP can become a chronic syndrome (Bajaj et al. 2003), and hence treatment should be directed at both biomechanical and neurophysiological issues (Samraj et al. 2005). This multimodal approach is also based on the clinical relationship between CPP and pelvic girdle pain (PGP), as many patients diagnosed with PGP also suffer from CPP (Vleeming et al. 2008).
Local muscle dysfunction: Muscle trigger points
Trigger points and chronic pelvic pain
The association between CPP and myofascial pain syndrome was identified several years ago (Slocumb 1984, 1990, Schmidt 1991). Myofascial pain is also related to urogenital pain (Doggweiler-Wiygul 2004). Jarrell (2004) found that abdominal trigger points (TrPs) predicted evidence of visceral disease in 90% of a sample of 55 patients with CPP. Montenegro et al. (2009) recently proposed that abdominal myofascial syndrome should be considered in the differential diagnosis of CPP. In 2009 the European Association of Urology published guidelines suggesting that TrPs should be considered in the diagnosis of CPP (Fall et al. 2010). In fact, Anderson et al. (2009) found that TrPs in the abdominal muscles were the most prevalent in male with CPP.
Myofascial pain syndrome can be associated with both TrPs and restrictions of the fascial tissue. The most commonly accepted definition for TrP is: ‘a hyperirritable spot in a taut band of a skeletal muscle that is painful on compression, stretch, overload or contraction which responds with a referred pain that is perceived distant from the spot’ (Simons et al. 1999). From a clinical viewpoint, we distinguish active and latent TrPs. Active TrPs are those in which local and referred pain reproduce symptoms reported by the patient, with the pain being recognized by the patient as a ‘familiar’ pain (Simons et al. 1999). For instance, in patients with CPP, active TrPs will reproduce perineal or pelvic pain. Latent TrPs are those where local and referred pain do not reproduce pain symptoms, or where elicited pain is not familiar to the patient (Simons et al. 1999). For instance, in a patient with neuropathic pelvic pain, referred pain can be elicited but does not reproduce the patient’s symptoms. Furthermore, a relevant feature of both active and latent TrPs is that each can induce muscle imbalances or altered motor recruitment (Lucas et al. 2004).
There are several studies demonstrating a relationship between CPP and muscle TrPs. Weiss (2001) reported the successful amelioration of symptoms in patients with interstitial cystitis using myofascial TrP release. Doggweiler-Wiygul & Wiygul (2002) found that inactivation of TrPs in PFM, gluteus and piriformis muscles improved or resolved the pain in four patients with severe CPP, interstitial cystitis and irritative voiding symptoms. Anderson et al. (2005) showed that incorporation of TrP inactivation into a multimodal approach for CPP in men resulted in an effective therapeutic approach, by providing a reduction in pain and urinary symptoms superior to that of traditional therapy. This study included voluntary isometric contractions and relaxation, to induce post-isometric relaxation and reciprocal inhibition, together with deep soft tissue mobilization (stripping, strumming, skin rolling and effleurage) as interventions directed at TrPs (Anderson et al. 2005). Anderson et al. (2006, 2009) also found that TrP inactivation was associated with significant improvement in urinary symptoms, libido, ejaculatory and erectile pain, and ejaculatory dysfunction in men with CPP. See Chapter 12 for more detail of Anderson’s studies. Langford et al. (2007) demonstrated the effectiveness of TrPs inactivation of the levator ani muscle for the management of some patients with CPP. In this study 13 of 18 women improved with the first TrP injection resulting in a success rate of 72%, whereas 6 of 18 (33%) were completely pain-free. FitzGerald et al. (2009) demonstrated a better response rate (57%) in CPP patients treated with TrP therapy as compared to the response rate (21%) in those patients receiving global therapeutic massage. In a review of prostatitis and CPP, Anderson (2002) described palpation and treatment protocols for locating muscle TrPs associated with prostatitis symptoms. In a subsequent later study, Anderson et al. (2009) confirmed a relationship between muscle TrPs and CPP in men, identifying the most common location of TrPs: pubococcygeus or puborectalis (90%), external oblique (80%), rectus abdominis (75%), adductors (19%) and gluteus medius (18%) muscles. Other relevant muscles in which TrPs also contribute to CPP are levator ani, iliopsoas, quadratus lumborum, gluteus maximus and the thoracolumbar extensor muscles (Simons et al. 1999, Carter 2000, Liebenson 2000, FitzGerald & Kotarinos 2003a, Chaitow 2007a, Montenegro et al. 2008, Anderson et al. 2009).
Why is inactivation of trigger points in chronic pelvic pain important?
The role of neurogenic inflammation has been emphasized as contributing to the pathophysiology of CPP (Wesselmann 2001). It is well accepted that noxious (nociceptive) stimuli can increase the production of pain-promoting substances at the nerve-free endings of the primary afferent nociceptors. When a sensitive nerve fibre is stimulated the impulse runs towards the spinal cord (orthodromic flow) and towards the periphery (antidromic). When the antidromic stimulus reaches the periphery, there is a release of several neuropeptides (e.g. nitric oxide, substance P, calcitonin gene-related protein) promoting neurogenic inflammation, characterized by vasodilatation, oedema and hyperalgesia (Wesselmann 2001). Clinicians should be aware of the neurophysiological theories for inactivating muscle TrPs in CPP.
1. Trigger points are a focus of peripheral nociception. Muscle pain is associated with the activation of nociceptors by a variety of endogenous substances, e.g. bradykinin or serotonin (Babenko et al. 1999a), substance P (Babenko et al. 1999b) and glutamate (Svensson et al. 2003). Microdialysis studies have found that concentrations of bradykinin, calcitonin gene-related peptide, substance P, tumour necrosis factor-α, interleukin-1β, serotonin or norepinephrine were significantly higher in active TrPs as compared to latent TrP or non-TrP tissues (Shah et al. 2005, 2008). Another study has demonstrated the existence of nociceptive hypersensitivity (hyperalgesia) and non-nociceptive hypersensitivity (allodynia) at the sites of muscle TrPs (Li et al. 2009). These studies support the proposal that TrPs constitute a focus of sensitization of both nociceptive and non-nociceptive nerve endings.
2. Trigger point nociception induces central sensitization.When muscle tissue is sensitized, nociceptors are more readily activated and respond inappropriately to normal innocuous or weak stimuli, e.g. light pressure or movement. The presence of multiple TrPs in different muscles (spatial summation), or the presence of TrPs for prolonged periods of time (temporal summation), can sensitize the spinal cord and supraspinal structures by means of a continued nociceptive afferent barrage into the central nervous system (Mense 1994). Kuan et al. (2007) demonstrated that spinal cord connections of muscle TrPs were effective in inducing neuroplastic changes in the dorsal horn neurons. Niddam et al. (2007) demonstrated that pain associated with TrPs is at least partially processed at supraspinal levels, particularly the peri-aqueductal grey matter. Readers are referred to Chapter 3 for a review of neurophysiology.
3. Trigger points and the sympathetic nervous system. There is evidence of an association between TrPs and the sympathetic nervous system (McNulty et al. 1994, Chen et al. 1998, Chung et al. 2004). Ge et al. (2006) found increased referred pain intensity and tenderness with sympathetic hyperactivity at muscle TrPs, suggesting a sympathetic contribution to the mechanisms responsible for the generation of referred pain. A study by Zhang et al. (2009) demonstrated an attenuated skin blood flow response after painful stimulation of latent TrPs, as compared with control non-TrPs, suggesting increased sympathetic vasoconstriction activity at latent TrPs.
Best evidence of soft tissue interventions for muscle trigger points
In this section we review the evidence for soft tissue interventions targeted at inactivating muscle TrPs. However, clinicians should consider that current evidence is based on the application of single treatments applied to TrPs, when multimodal approaches are usually practised by clinicians. Further, it is clear that management of myofascial dysfunction in patients with CPP requires a multidisciplinary approach (Srinivasan et al. 2007; see also Chapters 8.1 and 8.2). The inclusion of these techniques into a multimodal approach for patients with CPP has been found to be effective (Weiss 2001, Doggweiler-Wiygul & Wiygul 2002, Anderson et al. 2005, 2006).
Among the different interventions directed at inactivating TrPs, manual therapy is the first treatment option (Dommerholt et al. 2006). Different soft tissue interventions have been suggested, including: static compression (Hong et al. 1993, Simons et al. 1999, Fryer & Hodgson 2005, Fernández-de-las-Peñas et al. 2006, Gemmell et al. 2008, Dommerholt & McEvoy 2010), massage (Simons et al. 1999), stretching (Hong et al. 1993, Simons et al. 1999, Hanten et al. 2000), muscle energy techniques (Lewit 1999, Chaitow 2006, Rodríguez-Blanco et al. 2006), strain–counterstrain (Ibáñez-Garcia et al. 2009, Lewit 1999), neuromuscular techniques (Chaitow & Delany 2008, Ibáñez-García et al. 2009, Palomeque-del-Cerro & Fernández-de-las- Peñas 2009), positional release techniques (Chaitow 2007b) and manipulative interventions (Ruiz-Sáez et al. 2007, Fernández-de-las-Peñas 2009).
Systematic reviews have investigated the effectiveness of soft tissue manual intervention for inactivating TrPs (Fernández-de-las-Peñas et al. 2005, Rickards 2006, Vernon & Schneider 2009). These reviews found moderate to strong evidence supporting the use of static compression for immediate pain relief of muscle TrPs but limited evidence for long-term pain relief. Additionally, there is preliminary evidence demonstrating changes in muscle sensitivity after spinal manipulations (Ruíz-Sáez et al. 2006, Fernández-de-las-Peñas 2009), although further studies are required.
Application of soft tissue interventions for trigger points
In this section soft tissue interventions applied to TrPs in those muscles in which referred pain can contribute to CPP are described. Clinicians are encouraged to develop their own techniques based on a clinical reasoning process (see Chapter 7). Selection of any technique should include consideration of TrP irritability and the degree of sensitization of the central nervous system of the patient with CPP.
Compression interventions
Different compression techniques, depending on the amount of pressure applied, presence/absence of pain (Lewit 1999, Simons et al. 1999), duration of application (Hou et al. 2002), or position of the tissue (shortened or lengthened), have been described. In our clinical practice, the pressure level, duration of application, and position of the muscle, depend on sensitization mechanisms of the patient, and degree of irritability of the TrP. Table 11.1.1 summarizes clinical application of four different forms of compression: ischaemic compression (Travell & Simons 1983), TrP pressure release (Lewit 1999, Simons et al. 1999), strain/counter-strain (Jones 1981) or positional release therapies (Chaitow 2007b), and intermittent compression (Chaitow 1994).
Simons (2002) proposed that compressing the sarcomeres by direct pressure in a vertical and perpendicular manner may equalize the length of the muscle sarcomeres in the involved TrP and decrease pain. Hou et al. (2002) suggested that pain relief may result from reactive hyperaemia within the TrP or a spinal reflex mechanism for the relief of muscle tension.
Under the next heading, we describe different forms of compression interventions applied to pelvic muscle TrPs. Clinicians can apply ischaemic compression (Travell & Simons 1983), pressure release (Lewit 1999, Simons et al. 1999) or positional release therapy (Chaitow 2007b) principles depending on the patient’s characteristics.
Static compression of piriformis/external obturator muscle trigger points 
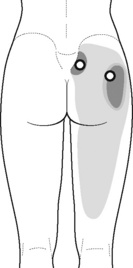
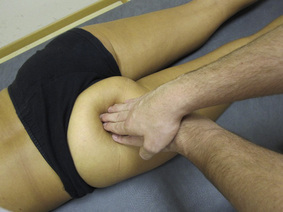
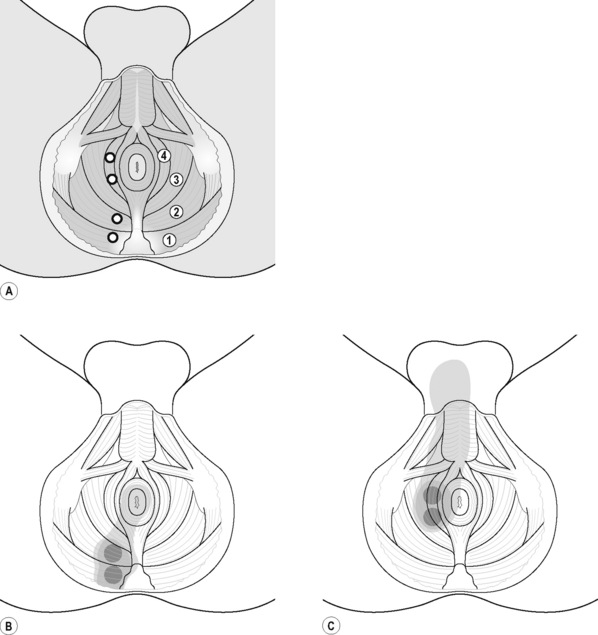
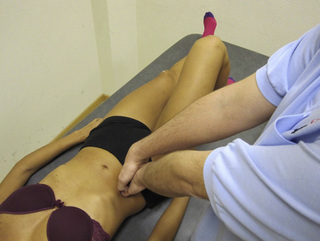
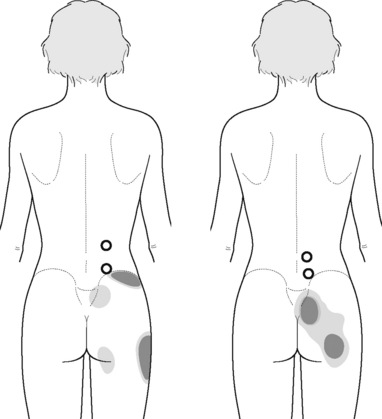
Typical piriformis TrP referred pain is shown in Figure 11.1.1 (Simons et al. 1999). TrPs in this muscle may contribute to pain in the lower back, buttock, hip, posterior thigh and leg, but also to pain into the groin, perineum and sometimes in the rectum during defecation. For this technique, the patient is prone with the therapist standing to the side. The therapist localizes the TrP (it can be located just lateral to the sacrum or the muscle belly), and applies a static compression directly over it. Clinicians are encouraged to use both hands during the technique (Figure 11.1.2). A similar technique may be applied over the external obturator muscle belly which has a similar pattern of referral to that of the piriformis (Cox & Bakkum 2005).
Static compression of pectineus muscle trigger points
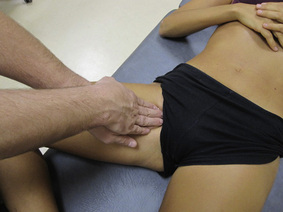
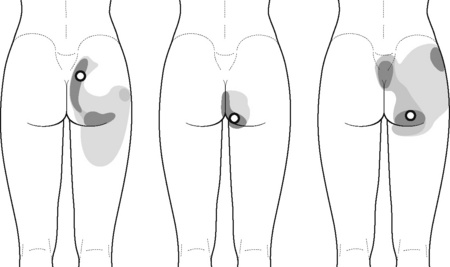
The pectineus muscle is an important adductor muscle in relation to CPP since TrP referred pain is commonly perceived as a deep dull pain in the lateral groin area (Figure 11.1.3A). For the technique, the patient lies supine with the therapist standing to the side. The therapist localizes the TrP which is usually located in the muscle belly (just lateral to the tendon of the adductor longus muscle at the pubic bone), and applies a static compression directly over it. Clinicians are encouraged to use both hands during the compression, although the thumb is also frequently employed (Figure 11.1.4).
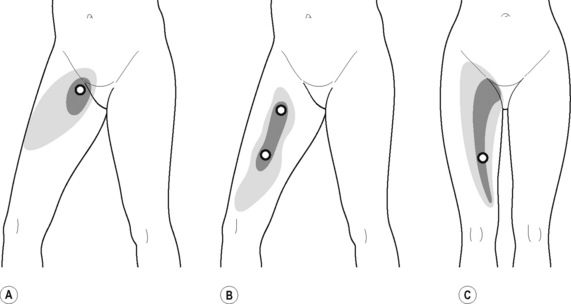
Figure 11.1.3 • Referred pain elicited by pectineus (A), adductor longus (B) and adductor magnus (C) muscle TrPs
Intermittent compression of pelvic floor muscle trigger points
PFM TrPs can mimic symptoms of painful coccydynia or levator ani syndrome (Simons et al. 1999, Chaitow 2007a). In general, pelvic floor muscle TrPs refer pain toward the perineum, vagina, penile base, and give a sensation of fullness into the rectum and an urgency to urinate (Figure 11.1.5A–C). Nevertheless, it seems that TrPs in some PFM are more prevalent in CPP (Anderson et al. 2009). For instance, referred pain from pubococcygeus and puborectalis muscles spreads to the perineum and adjacent urogenital structures. Lewit & Horacek (2004) demonstrated that inactivation of pubococcygeus muscle TrPs induced secondary inactivation of erector spine TrPs. Levator ani and coccygeus TrPs refer pain to the sacrococcygeal region and also to the vagina or penis. TrPs in the internal obturator refer pain to the anococcygeal region and to the vagina. It has also been observed clinically that in some patients with CPP, pelvic floor muscle TrPs can refer pain to the sacrum.
In our clinical experience, PFM respond very well to TrP pressure release. For this technique the patient lies supine or side-lying. The therapist localizes the TrP which is usually located in a specific PFM (particularly located in the ischiorectal fossa) and applies an intermittent digital (finger or thumb) compression to it (Figure 11.1.6). Further details regarding PFM trigger point techniques can be found in Chapter 13.
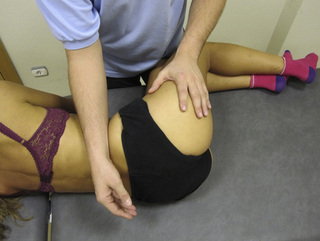
Compression and contraction of gluteus maximus muscle trigger points
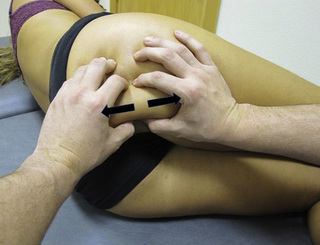
TrP-referred pain from gluteus maximus muscle is perceived as deep and burning pain located in the sacroiliac, coccyx and buttock areas (Figure 11.1.7). This technique consists of applying a TrP compression combined with an isometric contraction of the compressed muscle (Gröbli & Dejung 2003). For that purpose, the patient is side-lying with the therapist behind. TrPs within the gluteus maximus muscle are best palpated and compressed by pincer palpation. Once the therapist locates a TrP (it can placed in any part of the muscle belly) a pincer compression is applied using one or both hands (as illustrated in Figure 11.1.8). When the therapist perceives a slight relaxation of the TrP, the patient is asked to contract the muscle by squeezing both buttocks for 5 seconds. The therapist should maintain the compression during the contraction. If two hands are employed, stretching of the tissues housing the trigger point should follow the contraction, as illustrated. In the authors’ clinical experience, a total of ten repetitions is usually sufficient to achieve inactivation of gluteal muscle TrPs.
Stretching compression of iliopsoas muscle trigger points 
TrPs in the iliopsoas muscle refer pain to the groin area, superior part of the thigh and to the back (Figure 11.1.9). This is an important muscle since it is anatomically related to several urogenital structures and the lumbar plexus (Stepnik et al. 2006).
A stretching compression technique combines a compression intervention with passive or active stretching of the TrP taut band. For this purpose the patient is supine with the knee and hip flexed, and the foot on the table. The therapist compresses the TrP (usually located within the muscle belly reached through overlying abdominal muscles) with the tips of the fingers of one or both hands. At the time that the therapist perceives a slightly relaxation of the TrP taut band, the patient is asked to straighten the knee and the hip, either passively or actively, to increase the tension in the taut band (Figure 11.1.10). The aim of this technique is for the patient to achieve pain-free extension of the hip and knee, at the same time that the therapist maintains the compression.
Massage
Massage has multiple clinical applications with positive effects, but often lacks scientific evidence. This may be related to the fact that there are so many different forms of massage that still remain under-researched. The application of massage for inactivating muscle TrPs was discussed by Simons (2002) and Hong et al. (1993), who proposed that massage may exert a lengthening effect, similar to compression interventions. Massage can be performed along the TrP taut band (stretching longitudinal massage) or across the taut band (transverse massage). Hence, transverse massage offers transverse mobilization to the TrP taut band, whereas a longitudinal massage offers longitudinal mobilization to the taut band. In those muscles where clinicians can use pincer palpation, fingers can grasp the taut band from both sides of the TrP. Strokes centrifugally away from the TrP can lengthen the tissues (Simons 2002).
Transverse massage of quadratus lumborum muscle trigger points 
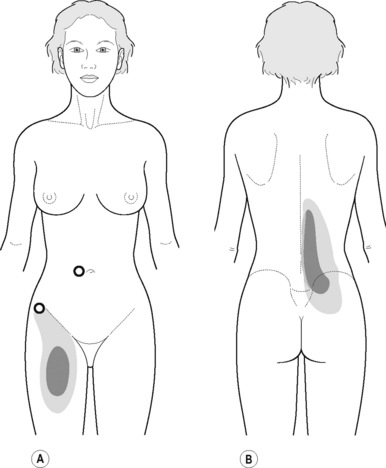
Referred pain from quadratus lumborum TrPs spreads along the crest of the iliac bone, to the outer upper aspect of the groin, the greater trochanter, the sacroiliac joint and the lower buttock (Figure 11.1.11). In some patients, additional referred pain to the anterior thigh, testicle and scrotum has been described (Simons et al. 1999).
For this technique the patient is side-lying with the therapist standing in front of the patient. The ulnar aspect of the therapist’s forearm should be placed over the muscle belly of the quadratus lumborum (where TrPs are usually located). The technique consists of applying a smooth and slow transverse massage over the TrP taut band (Figure 11.1.12). It is important to note that there is no established guideline (number of repetitions, time of application, or amount of pressure) for this technique. In the authors’ experience, the transverse massage should be painless, and applied until the therapist feels the tissues relax.
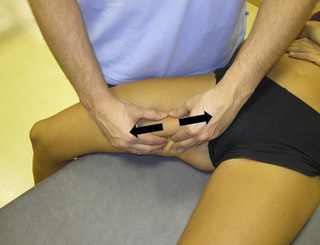
 Stretching longitudinal massage of adductor muscle trigger points
Stretching longitudinal massage of adductor muscle trigger points
TrPs within the adductor (brevis, longus and magnus) muscles refer pain to the medial aspect of the thigh and to the groin area (Figure 11.1.3B,C). In some patients, the adductor magnus TrPs also elicit an intrapelvic referred pain (Simons et al, 1999). For this technique the patient is supine or side-lying. Once the therapist locates a TrP (it can located in any of the adductor muscles), a pincer palpation of the TrP taut band is applied. The fingers of the therapist grasp the taut band from both sides, and stroke centrifugally away from the TrP (Figure 11.1.13).
Muscle energy interventions
There are several stretching applications targeted at inactivating TrPs: passive stretching (where the therapist passively stretches the muscle without participation of the patient), active stretching (where the patient actively stretches the muscle without participation of the therapist), spray and stretch involving a vapo-coolant spray applied during stretch (Hong et al. 1993, Simons et al. 1999), or muscle energy techniques (Fryer & Fossum 2009).
Muscle energy techniques comprise a system of manual procedures that utilize isometric and isotonic muscle contraction efforts from the patient, usually against a controlled matching counterforce from the therapist. Although there are different approaches for application of muscle energy techniques, the ‘contract–relax–release’, is the most commonly utilized technique. This involves the accurate localization of an isometric contraction (3–7 seconds) at a barrier defined as ‘the first sign of resistance’. An unyielding counterforce is supplied by the therapist. After the patient releases the contraction effort a new barrier is engaged, or stretching is introduced, past the previous barrier, actively or passively. The force and duration of isometric contraction can be varied, depending on the objective of the technique and the tissues involved. In fact, different durations of contraction have been proposed: 2–3 seconds (Mitchell & Mitchell 1995), 3–5 seconds (Greenman 2003), or 5–7 seconds (Chaitow 2006).
Several studies have demonstrated that muscle energy techniques increase muscle extensibility (Feland et al. 2001, Ferber et al. 2002, Ballantyne et al. 2003) and range of motion (Fryer & Ruszkowski 2004, Burns & Wells 2006) in healthy subjects. For a review of scientific evidence of muscle energy techniques, readers are referred to another text (Fryer 2006). The physiological therapeutic mechanisms by which muscle energy techniques exert their effect are speculative and controversial. Of the three most studied mechanisms that have been proposed, i.e. reflex relaxation, viscoelastic or muscle property changes (Fryer 2000), and increased tolerance to stretch, it is the latter that is most supported by the scientific literature (Fryer 2006).
Increased stretch tolerance may result from a decrease in pain perception (hypoalgesia) through the activation of muscle and joint mechanoreceptors, peripheral and central (activation of descending inhibitory pain systems) mechanisms (Fryer & Fossum 2009), and/or reduced concentrations of pro-inflammatory cytokines, and reduced sensitivity of peripheral nociceptors. Enhanced release of endocannabinoids may be one of the mechanisms of osteopathic manipulative treatment (McPartland et al. 2005), parallel to the effects of manipulative treatment upon serum endorphin levels (Vernon et al. 1986).