FIGURE 85.1 Pressure points in supine position.

FIGURE 85.2 Pressure points in sitting position.
Most (66%) patients with pressure ulcers are older than 70 years old, with a prevalence rate in nursing homes of 17% to 28%; in contrast, patients admitted with an acute illness have an incidence rate of 3% to 11%. In both subsets of patients, recurrence rates as high as 90% may be seen. The operating room, where patients are immobile, is a high-risk area, and there is a report that up to 25% of all pressure ulcers are initiated there (6).
Anatomic sites affected are primarily the hip and buttocks (67% of the cases involve ischial, trochanteric, and sacral tuberosity); 25% in malleolar, heel, patellar, or pretibial area; with the remainder occurring on the nose, chin, occiput, chest, back, or elbow. In paraplegic patients, pressure ulcers are a leading cause of death, responsible for an 8% mortality rate (7). Overall, an estimated 60,000 people die each year from complications of pressure ulcers. The yearly health care cost in the United States alone is in excess of $1 billion (8).

FIGURE 85.3 Shearing effect.
Several scoring systems are used to grade the risk for ulceration. The Braden scale is a summation rating scale made up of six subscores, consisting of sensory perception, moisture, activity, mobility, nutrition, and friction/shear, each ranging from 1 to 3 or 4 points, with total scores ranging from 6 to 23. The subscores measure the functional capabilities of the patient that contribute to either higher intensity and duration of pressure or lower tissue tolerance for pressure. A lower Braden score indicates lower levels of functioning with a higher risk for pressure ulcer development.
The Daniels classification looks at muscle and subcutaneous tissue breakdown, which occurs before dermal and epidermal changes are observed. Epidermal necrosis occurs later because epidermal cells are able to better withstand prolonged absence of oxygen than cells in the deeper tissues both in vivo and in vitro (9). Once skin damage is visible, irreversible internal damage may have already occurred (10).
Shea staging describes ulcers that start superficially and progress to deeper structures:
- Grade I: Limited to superficial epidermis and dermis
- Grade II: Involving the epidermis and dermis and extending to the adipose tissue
- Grade III: Extending through the superficial structures and adipose tissue down to muscle
- Grade IV: Complete soft tissue destruction down to bone
Currently, the most widely accepted classification system for pressure ulcers is that produced by the National Pressure Ulcer Advisory Panel (NPUAP). Considered to be a modification of the Shea system, it is used only to determine the initial depth, and is not a system to follow the natural history of the ulcer. It is also limited by the presence of eschar, which will mask the underlying damage.
- Stage I represents intact skin with signs of impending ulceration. Clinically, this may consist of blanchable erythema from reactive hyperemia that should resolve within 24 hours of relief of pressure; warmth and induration may also be present. Continued pressure creates erythema that does not blanch; this may be the first outward sign of tissue destruction. As pressure necrosis increases, the skin may appear white from ischemia. With proper treatment, resolution should be expected in 5 to 10 days (11).
- Stage II represents a partial-thickness loss of skin involving epidermis and possibly dermis. This lesion may present as an abrasion, blister, or superficial ulceration, with pigmentation changes. These, too, represent a reversible condition.
- Stage III represents a full-thickness loss of skin with extension into subcutaneous tissue but not through the underlying fascia. This lesion presents as a crater with or without undermining of adjacent tissue. On examination, this will appear as a necrotic, foul-smelling crater with altered light and dark pigmentation.
- Stage IV represents full-thickness loss of skin and subcutaneous tissue and extension into muscle, bone, tendon, or joint capsule. Osteomyelitis with bone destruction, dislocations, or pathologic fractures may be present. Sinus tracts and severe undermining commonly are present.
Treatment
All modalities of care for pressure ulcers fall along four paths.
Pressure Reduction
Frequent—at least every 2 hours—turning and repositioning the patient. Historically, this was adopted because of nursing issues (it took 2 hours for the nurse to rotate all ward patients). Currently, there is debate in the literature as to this being an adequate amount of time. In addition to positioning, mattresses that reduce pressure, such as low-air-loss and air-fluidized beds should be used for patients with stage III and IV ulcers, whereas for stage I and II ulcers, the use of static mattresses such as air, foam, or water overlays are the most beneficial.
Wound Management
Once an ulcer has developed, removal of dead tissue and debris, drainage, and protecting the surrounding healthy tissue are the goals. The pressure needed to clean wounds with no necrotic material is 2 to 5 pounds per square inch (psi). If necrotic debris is present, the pressure required increases by a factor of 2 to 3. The old wound dictum—if it is dry, wet it; if it is wet, dry it—has some validity here. A draining wound needs either a hydrocolloid or alginate, whereas a wound without drainage will respond to simple moist gauze; the surrounding skin of both needs to be kept lubricated, but not wet, to reduce friction. Negative pressure therapy enhances wound healing by reducing edema, increasing the rate of granulation tissue formation, and stimulating circulation. Increased blood flow translates into a reduction in the bacterial load and delivery of infection-fighting leukocytes (12). However, there are significant contraindications for the use of vacuum-assisted or negative pressure therapy including malignancy of the wound, untreated osteomyelitis, nonenteric fistulas, and exposed vessels, organs, or nerves (13). Various dressing categories are presented in Table 85.1.
Surgical Intervention
Debridement is the process of removing devitalized tissue. Stage III and IV ulcers will require some form of debridement, whether by surgical, autolytic, mechanical, or enzymatic means. The patient’s wound and overall status will dictate the means of debridement, a more stable patient receiving a more aggressive means of removing the necrotic material. In 1938, Davis (14) was the first to suggest replacing the unstable scar of a healed pressure sore with a flap of tissue. In 1947, Kostrubala and Greeley recommended excising the bony prominence and adding padding for the exposed bone with local fascia or muscle-fascia flaps. In addition, larger wounds may respond only to the placement of flaps, either fasciocutaneous or musculocutaneous. Flap failure can be seen after insufficient excision of soft tissue and bone, and if systemic factors such as nutritional status are suboptimal.
| TABLE 85.1 Dressing Categories | |
 | |
Nutritional Support
Malnourished patients have a higher susceptibility for ulcer formation. Once formed, these patients also have a diminished ability to heal or to prevent further ulcer formation in other sites. Patients with serum albumin levels less than 3 mg/dL may be candidates for supplemental feedings via enteral or parenteral routes.
PRIMARY BACTERIOLOGIC INFECTIONS
Skin and soft tissue infections are usually easily treated, but have the lethal potential. Any break in the usual protection of the integument, such as occurs with a cut, scrape, insect bite, splinter, or traumatic injury, allows bacteria to enter underlying tissues. Although a scrape or a cut will not usually result in a cellulitis, a tender, firm, painful, and rapidly expanding area of redness on the skin surrounding violation of the skin barrier should be cause for concern. Red streaks between lymph node–bearing areas may be visible, indicative of a potentially spreading infection. Certain areas are more prone in becoming infected depending on the age group, such as facial cellulitis occurring more commonly in adults older than 50 years and in children 6 months to 3 years of age.
The most common causative organisms in skin infections are group A β-hemolytic streptococci and Staphylococcus aureus. Depending on the source of contamination and whether the patient is immunocompromised, gram-negative rods and fungus can be seen. If the insult occurs during exposure to fresh water, the causative organism may be Aeromonas, a gram-negative rod.
Predisposing states in which a minor break in the skin barrier may lead to a significant infection includes patients with diabetes; immunodeficiency; varicella; venous, arterial or lymphatic insufficiency, such as that seen after lymphatic removal during mastectomy; or vein stripping for varicosities.
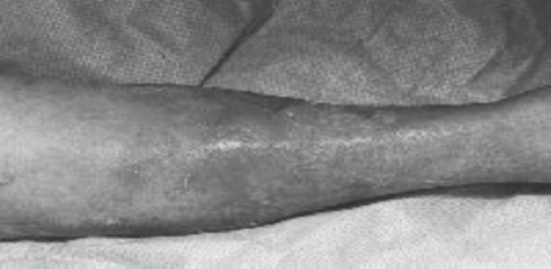
FIGURE 85.4 Erysipelas.
Treatment of uncomplicated cellulitis begins with removing the nidus of infection, cleansing the wound with an antiseptic agent, dressing the wound with an antiseptic ointment if indicated, and considering a course of oral antibiotics, such as dicloxacillin, 500 mg PO (orally) four times a day for 7 days, or cephalexin, 500 mg PO four times a day for 7 days. For patients with a suspected or known penicillin allergy, clindamycin, 400 mg, is given PO four times a day (15).
Erysipelas
Erysipelas (Fig. 85.4) is a form of cellulitis that affects the epidermis primarily, extending into the cutaneous lymphatics. During the Middle Ages, it was referred to as St. Anthony’s fire, named after the Egyptian healer who was successful in treating this condition. It shares the same underlying cause as cellulitis with bacterial inoculation into an area of skin violation. It is more commonly seen in children and the elderly. Erysipelas differs from cellulitis in that the inflamed area is distinct from the surrounding skin, being raised and demarcated. Erysipelas is often found on the face; however, it can also develop on the arms and legs. Sometimes the skin will have what is called a peau d’orange, or orange peel, look to it. As with cellulitis, Streptococcus is the primary organism identified with its toxin responsible for the brisk inflammation associated with this condition.
Treatment consists of elevation of the affected extremity, penicillin, 250 to 500 mg PO or 0.6 to 1.2 million units intramuscularly, given every 4 to 6 hours for a 10- to 20-day course. In cases of penicillin allergy, a macrolide or cephalosporin usually suffices. If the area affected becomes ulcerated, saline dressings changed every 12 hours will assist with wound closure.
Impetigo
Also known as pyoderma, impetigo (Fig. 85.5) is the most common bacterial infection of the skin seen. It is contagious and can happen at any age, but is more common in young children. Patients report skin lesions, often with associated adenopathy, with minimal systemic signs and symptoms. Impetigo may present in two forms: small vesicles with a honey-colored crust known as impetigo contagiosa, or purulent-appearing bullae, known as bullous impetigo (occurring principally in newborns and young children). While most commonly caused by S. aureus, group A β-hemolytic Streptococcus is also commonly seen in the over-2-year-old population. Warm temperatures, humidity, poor hygiene, and crowded living conditions can exacerbate the spread of impetigo. When associated with lymphadenitis in deeper infections, the term ecthyma is given. While topical mupirocin or retapamulin ointments can be used for treating limited impetigo, systemic therapy is preferred when treating widespread impetigo. Lesions usually resolve completely within 7 to 10 days.

FIGURE 85.5 Impetigo due to Staphylococcus aureus in a 68-year-old diabetic who fell onto concrete while walking and developed this lesion after 6 days. This resolved with conservative care and antibiotics.
CUTANEOUS FUNGAL INFECTIONS
The most common and important fungal infections that occur in the ICU setting are, for the most part, due to Candida, especially albicans, glabrata, and tropicalis (16). In immunocompromised or morbidly obese patients, this usually manifests itself as cutaneous moniliasis and can be treated with topical powders or ointments. Vaginitis, of course, should be treated with suppositories, and funguria is addressed by removing or replacing the urinary catheter, which will be successful in about one-third of patients.
SUBCUTANEOUS TISSUE INFECTIONS AND MYONECROSIS
The clinical distinction between superficial pyodermas and deep subcutaneous infections such as necrotizing fasciitis (NF) is often readily apparent. However, clinically distinguishing between NF and other forms of deep infections is often difficult due to commonalities in clinical presentation, anatomic features, and infecting organisms. Common among all of these types of infections is the variable tendency to form gas as well as the need for surgical exploration to differentiate and treat these processes. For example, clostridial and nonclostridial anerobic cellulitis both result in extensive gas formation, typically present with mild to moderate systemic symptoms, and lack muscle involvement; on the other hand, clostridial myonecrosis presents as a life-threatening infection with significant muscle involvement. The differentiation between these infections is made in the operating room.
The most common gas forming soft tissue infections include clostridial cellulitis, nonclostridial anerobic cellulitis, clostridial myonecrosis (gas gangrene), NF (Types I and II), Fournier gangrene, and synergistic necrotizing cellulitis (Meleney gangrene). Immediate surgical debridement is mandated once the diagnosis of any of these infections is made.
Necrotizing Fasciitis
Synergistic necrotizing cellulitis was first clearly documented in 1926. In their initial report, Brewer and Meleny (17) presented clinical and bacteriologic evidence of symbiotic growth and enhanced invasiveness between anerobic Streptococcus and Staphylococcus aureus. Their early description of what is now considered a variant of NF defined many of the prominent clinical and anatomic features of this type of infection. An early retrospective case series from the time period between 1958 and 1970 reported 76% mortality among patients with this infection; a mortality rate over twice that reported by contemporary reports, emphasizing that early recognition, surgical debridement, and targeted antimicrobial therapy have dramatically improved survival (18).
The term NF was first coined in 1952 by Wilson (19); the pathology involves the underlying fascia and subcutaneous tissue, but spares muscle. Myositis results in muscle involvement, which becomes exquisitely tender and indurated. The muscle involvement leads to elevation of the creatine phosphokinase (CPK) and can spread over several hours to contiguous muscle groups, thus heightening the need for early diagnosis and treatment. Fournier gangrene is listed here as a separate entity due to its predilection for the perineum.
All of these subgroups have in common pathogenicity with the organisms spreading from subcutaneous tissues to both superficial and deep fascial planes (Fig. 85.6). The local effect is vascular occlusion, ischemia, and necrosis. Prompt recognition is critical due to the rapidity with which this infection progresses. The reported mortality due to NF ranges from 24% to 34%, but decreases to about 12% with prompt recognition and treatment; Fournier gangrene results in a slightly lower mortality rate (15%) (20,21).
NF may be classified as types I and II, Fournier, and gas gangrene; these are discussed in the paragraphs that follows.
Type I (polymicrobial)
Usually occurring after injury or surgery, type I NF can be misdiagnosed as a simple cellulitis; however, as tissue necrosis and hypoxia continue, pain and systemic symptoms of fever, chills, and malaise increase as the underlying tissue liquefies; the overlying skin may show minimal changes. In the later stages, extension into the muscle itself occurs and, over 2 to 3 days, the erythema increases, with occasional bullae formation. Cultures may reveal a combination of aerobic and anaerobic organisms. Deep soft tissue infection of the perineum is termed Fournier gangrene (19). Many of these patients have predisposing comorbidities, such as diabetes or the presence of immunosuppressed states. Histologically, thrombosis of blood vessels and abundant bacteria with many polymorphonuclear cells are typically seen.
Type II (group A streptococcal)
Also known, colloquially, as “flesh-eating bacteria” or, by clinicians, as synergistic gangrene of Meleney (Figs. 85.7 and 85.8). As with type I NF, a nearly normal-appearing overlying skin may result in a delay in diagnosing the underlying, and ongoing, necrosis. A simple incision into the region affected will demonstrate drainage or even, in advanced cases, gas. Predisposing factors include varicella infection and the use of nonsteroidal anti-inflammatory drugs (NSAIDs) (22). NSAID use is seen as an immunomodulator, which may predispose to this condition. With type II NF, there is an association with the Streptococcal toxic shock syndrome, similar to its Staphylococcal counterpart except for the presence of necrosis as the precipitant event.

FIGURE 85.6 Pathophysiology of necrotizing fasciitis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








