Fig. 8.1
Operating room setup. Organization of operating table, robot cart, scrub nurse and table, surgeon console, anesthesia, and surgical assistant
Incision, Port Placement, and Instrumentation
The patient’s abdomen is prepped and draped in standard sterile fashion. The infra-umbilical crease is incised in a transverse, curvilinear fashion approximately 2–2.5 cm in length (Fig. 8.2a). The subcutaneous fat is dissected off the fascia to allow a 2.5-cm transverse fascial incision. The peritoneum is entered sharply and carefully to avoid intra-abdominal organ injury. The peritoneal cavity is swept with a finger to assess for intra-abdominal adhesions and ensure safe insertion of the multichannel umbilical port. Any local adhesions are lysed sharply to facilitate port insertion.
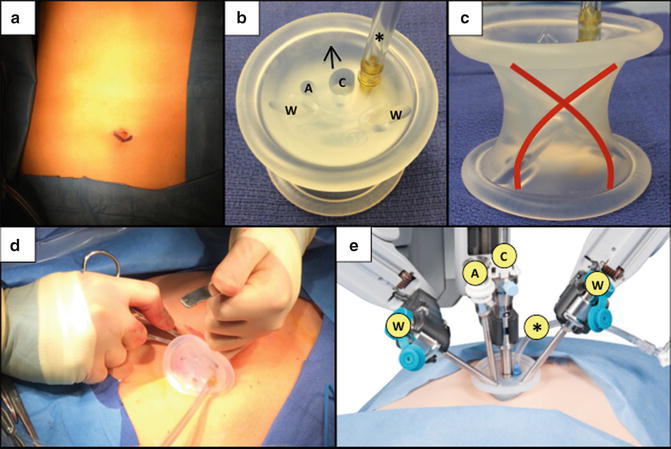

Fig. 8.2
Umbilical port. An infraumbilical curvilinear incision is made (a) for insertion of the da Vinci ® Single-Site ® port platform (b), whose working ports cross at the abdominal wall (red lines) (c). Using an S-retractor, the umbilical port is gently inserted with the arrow pointing toward the right upper quadrant (d). The robotic ports are then docked with their appropriate instrument arms (e). ©2015 Intuitive Surgical, Inc. W working instrument port, A assistant’s accessory instrument port, C camera Port, Asterisk insufflation adaptor
In order to place several instruments through a single infraumbilical incision, the multichannel da Vinci ® Single–Site ® port platform (Intuitive Surgical Inc., Sunnyvale, CA, USA) is utilized. The port is cylindrical with five lumens that provide access for two working instruments, a 8.5 mm 3D-HD endoscope camera, an assistant’s accessory port, and insufflation adaptor (Fig. 8.2). The working ports cross at the abdominal wall (Fig. 8.2c), so that the right instrument is positioned on the left side of the operative field, and the left instrument is positioned on the right—this arrangement minimizes extracorporeal clashing of the robotic arms. The robotic console corrects for this switch in laterality so that the operating surgeon’s right hand controls the “left extracorporeal” robotic arm, and the left hand controls the “right extracorporeal” arm. This configuration has been shown to improve single-site simulator task performance by eliminating instrument collisions, decreasing camera manipulations, and improving clutching efficiency [72].
After umbilical port placement, the abdomen is insufflated with CO2 to 15 mmHg, and the camera and instruments are inserted under direct vision into the abdominal cavity. Specifically, a laparoscopic grasper is inserted through the accessory port by the assistant at the bedside for cephalad retraction of the gallbladder fundus. The “right extracorporeal” robotic arm (i.e., left intracorporeal instrument) is dedicated for the infundibulum grasper. The “left extracorporeal” robotic arm (i.e., right intracorporeal instrument) is designated for electrocautery (Covidien ForceTriad monopolar electrocautery platform, ValleyLab, Boulder, CO, USA), suction, and the clip applier.
Triangulation and Flexible Instrumentation
Both the SILC and SSRC techniques require the surgeon and assistant to control four instruments in a limited space through a 2.5-cm fascial incision. Thus, it is of utmost importance to employ the spatial principle of triangulation of instrumentation to maximize the operative field. The single-site platform allows the surgeon to direct instruments from a radial position extracorporeally towards an opposite point intracorporeally, thereby crossing the plane of visualization. This crossing vector is not conventional in multi-port laparoscopy; however in SILC and SSRC, it maximizes retraction in the opposite direction of port insertion (Fig. 8.3a). Specifically, a grasper is placed through the assistant’s accessory port to grasp the fundus and retract superiorly, while the endoscope is inserted through the camera port and aimed inferiorly. This triangulation maximizes the angle of cephalad gallbladder retraction through the narrow umbilical incision while maintaining an unobscured view of the operative field.
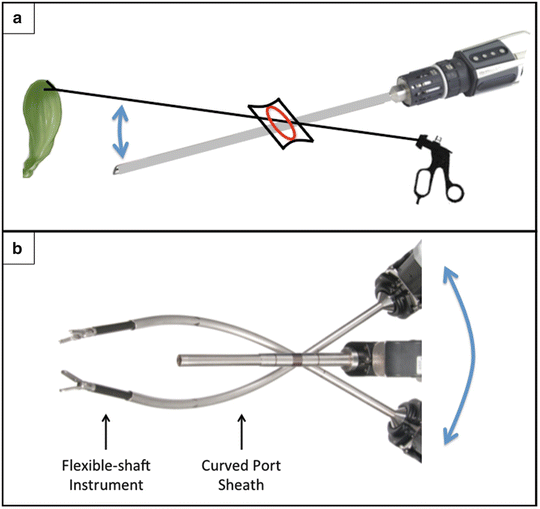

Fig. 8.3
Triangulation of crossing vectors. (a) Crossing vectors of the camera and fundal grasper maximize cephalad retraction and visualization of the gallbladder (arrow). (b) Crossing vectors of working port instruments maximize extracorporeal robotic arm range of motion (arrow); the curved port sheaths redirect the flexible instruments towards the operative field and minimize intracorporeal instrument collisions. ©2015 Intuitive Surgical, Inc.
To minimize instrument collisions, the two working port vectors are crossed at the abdominal wall. Rigid curved cannulas are inserted through the two working ports, through which flexible instruments will be placed (Fig. 8.3b). These crossing vectors maximize the extracorporeal robotic arms’ range of motion, thereby reducing collisions. Additionally, due to the flexibility of the instruments, the rigid curved cannula allows for redirection of the radially oriented instruments back to the center of the operative field [73]. This minimizes intracorporeal instrument collision, provides adequate infundibulum retraction (“right extracorporeal” robotic arm), and facilitates electrocautery dissection (“left extracorporeal” robotic arm).
Approaches to Dissection
There are two main approaches to gallbladder dissection: anterograde (fundus-last) and retrograde (fundus-first). The retrograde approach is commonly used in open cholecystectomy as a safe method of dissection with the visual plane in the anterior–posterior direction. This allows a safe dissection plane from the fundus toward the cystic duct by retracting the gallbladder away from the liver bed, particularly if there is marked cholecystitis obscuring the anatomy. The anterograde approach is the method of choice during minimally invasive procedures. This approach allows safe identification of Calot’s triangle from the laparoscopic field of view, which is directed from the umbilicus in the caudal-cephalad plane.
The anterograde approach for SILC and SSRC is similar to MPLC. It begins with identification of the gallbladder and careful lysing of any adhesions preventing adequate visualization and exposure. The fundus is then grasped through the assistant’s accessory port and retracted superiorly, using the triangulation concept as described above. Adhesions to surrounding structures, such as the duodenum and omentum, are then stripped away until the inferolateral aspect of the gallbladder is completely visualized.
Identification of Calot’s triangle is the next critical step. This is achieved by careful retraction of the infundibulum using the “right extracorporeal” robotic arm and dissection using the “left extracorporeal” arm. The peritoneal reflection is taken down medially and laterally to allow for visualization of Calot’s triangle. The surgeon can dissect with a Maryland forceps, electrocautery, or suction device when appropriate, until the cystic duct, common hepatic duct, and inferior border of the liver are identified. It is imperative to avoid bleeding, as this can obscure the visual field. Ultimately, a window around the cystic duct is created to allow the surgeon to visualize the isolated cystic duct and cystic artery entering the gallbladder. Of note, it is important to minimize the use of electrocautery near Calot’s triangle to avoid thermal injury to the cystic and common hepatic ducts. The cystic duct and artery are each clipped and divided with two clips remaining on each of the in situ structures. The gallbladder is then dissected off the liver bed with electrocautery, being mindful to avoid an aberrant right hepatic artery.
In order to aid in anatomical identification during dissection, the robotic platform allows for near-infrared fluorescent cholangiography using ICG, a low-toxicity fluorescent dye containing sodium iodide that binds plasma proteins and is excreted exclusively in bile. When exposed to near-infrared light by the robotic endoscope, ICG emits light detectable at a peak wavelength of 830 mm, which illuminates the biliary tree and surrounding vasculature [74]. Specifically, three milliliters (mL) of ICG is injected intravenously 30 min prior to incision. Its peak absorption after excretion into the biliary tree occurs 45–60 min postinjection, which aids in identification of the cystic duct, CBD, common hepatic duct, and potential aberrant ducts. Additional 3 mL aliquots can be injected intraoperatively to properly identify surrounding vasculature (e.g., the cystic artery) within 45 s postinjection (Fig. 8.4).
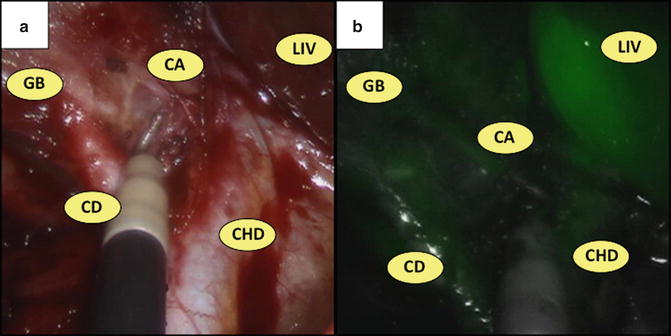

Fig. 8.4
Near-infrared fluorescent cholangiography. Identification of the gallbladder (GB), cystic duct (CD), cystic artery (CA), common hepatic duct (CHD), and liver (LIV) during cholecystectomy (a) using near-infrared fluorescent cholangiography (b)
Unlike traditional cholangiography, near-infrared fluorescent cholangiography allows the surgeon to evaluate biliary anatomy in real time without inserting catheters into the cystic duct, and can quickly assist in delineating biliary anatomy during dissection of Calot’s triangle. Although contraindicated in pregnancy and patients with iodide allergies, this technique has been demonstrated as safe and effective for identifying biliary anatomy in both laparoscopic and single-site robotic cholecystectomy—specifically, it identifies the cystic duct, CBD, and common hepatic duct in 97–100 %, 83–100 %, 67–100 % of cases, respectively [75–78]. Moreover, it requires only a simple injection without implementation of a C-arm or exposure to radiation. Therefore, we recommend routine usage of real-time near-infrared cholangiography during SSRC only to aid during anatomical dissection; it is currently not indicated to evaluate for choledocholithiasis. If ICG imaging is negative during SSRC, proceed with careful dissection and employ traditional intraoperative cholangiography as indicated (see below).
A retrograde approach has been described as a safe alternative during laparoscopic cholecystectomy, usually performed in cases with severe inflammation and an inability to safely identify Calot’s triangle [79–86]. This method may reduce the expected conversion-to-open rate without increasing the risk of injury to the biliary tree [79, 83]. It has also been described as a safe option during SILC to obtain a 360° view around the cystic duct before transection [87, 88]; only two case series have reported this approach in SSRC [89, 90]. This method begins with caudal retraction of the gallbladder away from the liver. A fundal dissection plane is started while leaving a small rind of the edematous gallbladder wall on the liver bed. This rind can be grasped and provide retraction of the liver bed away from the gallbladder. The dissection is continued until the cystic duct and artery are identified, while confirming a 360° view around the cystic duct prior to transection. This method is reserved as an attempt to avoid conversion to open; however, if safe dissection is difficult, we recommend converting directly to the open approach. We typically do not convert to a multi-port laparoscopic approach as an intermediate step because it has not been shown to reduce conversion rates after a failed anterograde and retrograde laparoscopic approach, and it will only prolong the operative time. However, during the learning phase, conversion to MPLC may be employed to aid in difficult dissections, or when there is bleeding that cannot be controlled via the single-site platform.
Intraoperative Cholangiography and Choledocholithiasis
Intraoperative cholangiography (IOC) is generally employed to investigate suspected choledocholithiasis or to identify aberrant or obscured anatomy. Specific indications include a clinical history of jaundice, transaminitis, direct hyperbilirubinemia, pancreatitis, increased amylase or lipase levels, ultrasound findings of dilated common duct or intra-ductal stones, or failed preoperative endoscopic retrograde cholangiopancreatography (ERCP) [91–93]. Some groups have concluded hyperbilirubinemia and dilated common duct on preoperative ultrasound should be the sole indications for IOC [94].
If preoperative choledocholithiasis is suspected, there are several management options including preoperative ERCP followed by cholecystectomy (two-stage) or cholecystectomy with common bile duct exploration (single-stage). One study performed a decision tree analysis summarizing the literature and found that single-stage laparoscopic common bile duct exploration during cholecystectomy has lower morbidity and mortality rates compared to preoperative ERCP followed by cholecystectomy [95]. While common bile duct exploration has been well described in MPLC, there has been only one small retrospective review describing 13 patients who underwent successful SILC with common duct exploration [96]. The single-incision CBD exploration technique has not been reproduced by another group yet, and only multi-port robotic CBD explorations have been reported [97, 98].
There are substantial data advocating for routine IOC to be performed during cholecystectomy, suggesting that it decreases the risk of common bile duct injury [99–102]. However, there is conflicting evidence reporting no improvement in bile duct injury, overall morbidity, or mortality with routine IOC [103–105]. One systematic review of eight randomized trials evaluating the ability of IOC to detect choledocholithiasis and biliary injury concluded there is no evidence to support or abandon use of IOC, and the decision to perform IOC should be left to the discretion of the surgeon [106]. Routine IOC during SILC without making a separate incision for the cholangiogram catheter has been demonstrated as safe and feasible [107]. However, while IOC during SSRC has also been reported in the literature [20, 108], it extends operative times, involves a multidisciplinary team, and requires C-arm placement which typically necessitates re-docking at least one robotic arm. Thus, most robotic surgeons do not routinely perform IOC during SSRC, and will selectively pursue it only if there is a clinical suspicion for choledocholithiasis or if the biliary anatomy needs to be clarified before further dissection.
When traditional IOC is indicated in SSRC, a 14-gauge angiocath is placed through 2 mm stab incision in the right upper quadrant under direct visualization. The needle is removed, leaving the angiocath sheath in place to provide access for the cholangiogram catheter. As in MPLC, a small transverse incision is made in the cystic duct with robotic shears. The cholangiogram catheter is then inserted into the cystic duct and secured with a laparoscopic clip applier. Saline is flushed to ensure adequate flow and the IOC is performed using intraoperative fluoroscopy. Typically, the robot will have to be undocked to accommodate the C-arm. Upon completion of the cholangiogram, the clip, cholangiogram catheter, and angiocath are removed and discarded.
If stones are identified in the common hepatic or common bile duct, we recommend completing the cholecystectomy and performing intraoperative or postoperative ERCP for stone retrieval. If this is not possible, a common duct exploration needs to be performed. As mentioned above, CBD exploration has been described via single-incision laparoscopic, multi-port robotic, and open approaches depending on the expertise of the surgeon. However, at this point, further studies analyzing the feasibility, safety, and outcomes of single-incision CBD exploration are warranted before we recommend it for routine usage.
Organ Extraction and Closure
Upon successful completion of gallbladder dissection, and IOC if necessary, the gallbladder is placed in an Endo Catch specimen retrieval bag (Covidien, Mansfield, MA, USA). The liver bed is inspected for hemostasis and the operative field is irrigated with saline. The instruments are then removed from the ports and the abdomen is desufflated. The robotic arms are detached from the instrument ports, the multichannel da Vinci ® Single–Site ® port is removed, and the specimen is extracted through the umbilical incision. Finally, the transverse fascia is closed with at least four 0-vicryl sutures on a GU needle and the skin is closed with a continuous subcuticular 4-0 monofilament absorbable suture.
Aberrant Anatomy
The most common cause of iatrogenic liver and common duct injury is secondary to misperception of anatomy, not a failure in technical skill, knowledge, or judgment [109]. There are several common anatomic variants that the surgeon must be aware of to avoid a catastrophic complication, especially when learning the single-incision and robotic techniques.
Alterations in normal gallbladder geometry can obscure the anatomy. Enlargement of the cystic duct and infundibulum secondary to an obstructing cystic duct stone, also known as the Hartman’s pouch, can obscure the cystic and common bile duct anatomy. The most severe form of this scenario is Mirizzi’s syndrome, where a large stone in Hartman’s pouch causes obstruction, adhesion, or erosion into the common duct [110]. Although the incidence of Mirizzi’s syndrome is only estimated at 0.5–1.0 % of all laparoscopic cholecystectomies, it has a conversion-to-open rate of 40–75 % [22–24].
There are several distinct features of the cystic duct that promote accurate identification. A normal cystic duct is 2–4 cm in length and approximately 5 mm in diameter. The absence of a true cystic duct is extremely rare, thus an inability to identify the duct is likely secondary to obscured anatomy, not an anatomical variant. Additionally, any tubular structure larger than 5 mm should be fully delineated before identifying it as a dilated cystic duct. The incidence of multiple cystic ducts is also quite rare, thus identification of multiple tubular structures near the gallbladder should raise suspicion of a tortuous CBD or accessory ducts. Furthermore, the cystic duct can take a variable course originating from the CBD: it may course perpendicular, parallel, or spiral around the CBD before entering the gallbladder [110]. Regardless of configuration, the best way to identify the cystic duct is by clearly dissecting Calot’s triangle and visualizing the duct fully entering the gallbladder.
Accessory extrahepatic bile ducts draining directly from the liver bed exist in up to 20 % of patients and can be misidentified as the cystic duct during cholecystectomy. An accessory right anterior or right posterior hepatic duct can originate from the right lobe of the liver, cross Calot’s triangle, and insert into the cystic, common hepatic, or common bile ducts [110, 111]. These ducts may be of significant diameter and biliary drainage, thus injury to them may require Roux-en-Y hepaticojejunostomy reconstruction [112]. Real-time near-infrared cholangiography during SSRC could potentially help the surgeon differentiate between these variants.
The cystic artery must be fully isolated before ligation. It originates from the right hepatic artery (RHA) and is commonly found in the center of Calot’s triangle in approximately 85 % of cases. It divides into an anterior and posterior branch near the gallbladder wall, also supplying the cystic duct with a small accessory branch. There are several variants of which the laparoscopic surgeon must be aware. A double cystic artery occurs in 15–25 % of patients, where two vessels originate from the RHA and travel through Calot’s triangle into the gallbladder. Approximately 13 % of patients may have a cystic artery that does not travel through Calot’s triangle secondary to variations in arterial origin. These variant vessels can arise from the gastroduodenal artery, superior mesenteric artery, a replaced right hepatic artery, the left hepatic artery, or directly from the liver parenchyma [113]. Lastly, the cystic artery can be misidentified as the right hepatic artery, which normally courses posterior to the common duct then enters the liver at the superior edge of Calot’s triangle. In up to 5 % of cases, the right hepatic artery may tortuously travel through Calot’s triangle, known as Moynihan’s hump, resulting in a short cystic artery and high risk of vascular injury [114]. These anatomic variants must be properly identified to avoid inadvertent ligation or injury. Since ICG remains intravascular before being excreted by the liver, intravenous injection 45 s prior to clipping could help identify the cystic artery.
Indications for Conversion
Although one of the major benefits of performing SILC or SSRC is to attain a nearly “scarless” cosmetic result, the surgeon must be cognizant to abandon the goal of improved cosmesis and convert to an open procedure for patient safety. The conversion-to-open rate for acute cholecystitis in MPLC is estimated to be 9.0–9.5 %, and is associated with a 1.3-fold increase in morbidity [115, 116]. Similarly, the rate of conversion in SSRC to multi-port or open cholecystectomy (in a patient population including acute cholecystitis) is approximately 6 % [17]. Thus, it is important to understand that this patient population can be very ill, and certain scenarios should be recognized to convince the surgeon to convert to open and minimize operative length and risk for biliary injury.
We typically do not recommend conversion to four-port conventional cholecystectomy since it likely does not confer a higher success rate and may just prolong the operation unnecessarily. Several studies have supported this as a method of avoiding laparotomy during SSRC [17]; however, if dissection is too difficult for a single-incision approach, it is generally safer to convert directly to an open approach. The only scenarios warranting conversion to MPLC is bleeding or assistance during the learning curve.
Severe cholecystitis may be an indication for conversion to avoid biliary or vascular injury, depending on the surgeon’s laparoscopic skill level. However, some centers advocate laparoscopic subtotal cholecystectomy with IOC as a safe, viable method to avoid laparotomy or biliary injury [117–120]. This procedure has been associated with a longer operative time and higher operative blood loss; however, these risks do not necessarily outweigh those of conversion to open [121]. These patients usually require intraoperative drain placement to monitor for biliary leak and potential ERCP with stent placement if one is detected. Nevertheless, there are risks of recurrent cholecystitis in patients with a gallbladder remnant, thus it may be preferable to completely remove the gallbladder even if via laparotomy.
A less common relative indication for conversion to open includes Mirizzi syndrome, which has a 75 % conversion rate, with some patients ultimately requiring a common duct repair intraoperatively or even a Roux-en-Y hepaticojejunostomy reconstruction depending on the degree of CBD involvement [23]. Additionally, there is up to 28 % incidence of gallbladder carcinoma associated with Mirizzi syndrome, thus an open operation with an experienced surgical oncologist should be considered [122].
Morbidity
Two studies have summarized the growing body of literature regarding the safety and morbidity of single-incision cholecystectomy, each evaluating nearly 1200 SILC patients with comparable results [69, 123]. The overall technical success rate without conversion to multi-port or open was over 90 %, but only 60 % for acute cholecystitis. While there were no mortalities reported, major complications requiring intervention or readmission occurred in 2.7 % of patients including retained stones (0.9 %), biliary leak (0.6 %), CBD stricture (0.1 %), and bile duct injury (<0.1 %). The minor complication rate was approximately 3 %, including wound infection (2 %), seroma (1.5 %), and ileus (0.2 %). Notably, the presence of acute cholecystitis did not appear to significantly affect complication rates. Thus, these two large studies have elucidated an acceptably low rate of overall complications, bile duct injury, readmission, and minor complications similar to MPLC provided adequate laparoscopic skills of the operating surgeon.
Nevertheless, there have been reports in the literature documenting higher rates of postoperative incisional hernia after single-incision cholecystectomy. Marks et al. performed a large, prospective, randomized, multicenter study analyzing SILC vs. MPLC [124]. While there was no difference in adverse events between groups, they found a significantly higher incisional hernia rate in the SILC group at 12-month follow-up (8.4 % vs. 1.2 %, p = 0.03). One-half of these hernias required operative repair by the time of publication. However, fascial closure during cholecystectomy was left to surgeon preference and there were not enough patients enrolled to determine if different techniques predisposed to hernia formation. Furthermore, the study noted it included surgeons with prior experience of at least ten SILC operations—since the learning curve may take approximately 20 cases, the study’s outcomes may have varied if it required surgeons with more SILC experience. Indeed, one single-surgeon, prospective study analyzed the incisional hernia rate after SILC with mean follow-up of 17 months and observed a 2 % incisional hernia rate [125], which is comparable to the known MPLC hernia rate of 1.7 % [126]. This lower rate may be a result of using the same closure technique on each patient, which were several interrupted Vicryl-1 (or PDS-1) sutures. Other studies including meta-analyses have also observed no difference in rates of postoperative hernia after single-incision cholecystectomy [12, 13, 70, 127, 128]. However, further prospective long-term studies are warranted to evaluate the optimal orientation of fascial incision and method of closure in order to ensure a low rate postoperative incisional hernia.
The rate of bile duct injury for MPLC has been estimated at 0.5 % and is associated with a more than twofold increased risk for mortality [129]. These patients usually require at minimum an ERCP and biliary stent placement if there is a minor injury, but more commonly these patients require a hepaticojejunostomy reconstruction by an experienced hepatobiliary surgeon. Although there have been over 350 SSRC cases reported in the literature without biliary injury [15–20] and the two SILC reviews discussed above reported <0.1 % rate of bile duct injury, one recent study suggested otherwise [130]. These authors performed a comprehensive literature search of SILC, including 45 studies and 2626 patients. The calculated overall complication rate was 4.2 % with a bile duct injury rate of 0.72 %. This is higher than the expected 0.4–0.5 % bile duct injury rate known for MPLC. There were no comparison groups to assess for statistical significance in this study; however, it is important to conclude that there may be a higher rate of biliary injury with the introduction of a single-incision procedure, and the technique should be reserved to surgeons with capable skills on minimally invasive platforms. It is also important to determine if these complications occur during the learning curve phase or afterwards with experienced surgeons.
Convalescence Data
As the main benefit of the single-incision technique is to be minimally invasive and “scarless,” there are convalescence data now reporting recovery events such as patient satisfaction, postoperative pain, mean hospital stay, and time to resume normal functional capabilities. These findings have been well studied in the SILC population, and will be reviewed here.
Convalescence data has been well documented for MPLC [131, 132]. Pain is most intense for the first 72 h postoperatively, which can be minimized with local anesthesia, oral opioids, or nonsteroidal anti-inflammatory drugs (NSAIDs). Most patients require 1 week until return to work and 2 days until return to recreational activity. Additionally, some studies advocate satisfactory cosmetic outcome based on postoperative patient questionnaire [133].
Now that single-incision cholecystectomy has been a surgical option for over a decade, there are reports in the literature documenting improved patient satisfaction with SILC when compared to MPLC [12]. One prospective trial demonstrated that there was no difference in scar assessment at early postoperative follow-up; however, patients do perceive a statistically significant superior scar assessment at longer-term postoperative follow-up [134]. Another similar randomized prospective trial supported this finding by observing increased patient satisfaction with wound appearance as early as two weeks postoperatively [135]. Other meta-analyses had consistent findings of improved cosmetic outcomes with SILC [11, 13, 70, 136].
The consensus of postoperative pain control has been controversial. Some randomized trials as well as meta-analyses have not found any statistical difference in postoperative pain control between SILC and MPLC [12–14, 70, 135, 137]. On the other hand, several studies and meta-analyses have documented improved pain control postoperatively, especially within 24-h [11, 127, 136, 138, 139]. This discrepancy is likely due to the widely different methods of pain control available, including oral opioids, NSAIDs, subcutaneous injection of local anesthetic, and even epidural anesthesia. We recommend local anesthetic for all patients combined with an oral modality best fit for the individual patient.
As the SILC procedure is a laparoscopic approach, one would expect the length of hospital stay and mean days until return to work to be similar to MPLC. Indeed, several studies have confirmed this hypothesis, finding the mean length-of-stay to be between 0 and 3 days without significant difference when compared to MPLC [11, 12, 14, 70, 128, 140]. Initial SSRC cohorts have reported an average number of days until return to normal activity and work of 4.5 and 7.5, respectively [16].
These comprehensive results are reiterated in one of the largest meta-analyses to date, which analyzed 25 randomized controlled trials including 944 SILC and 897 MPLC patients [136]. They observed that SILC was statistically superior to MPLC in cosmetic score, shorter length of incision, and postoperative pain within 12 h. In conclusion, these data support the use for patients interested in improved cosmesis and possibly improved pain control postoperatively; however, further convalescence data regarding SSRC remains to be elucidated.
Comparison to Standard Laparoscopy
Since no studies have compared SSRC to either SILC or MPLC, this chapter has compared the SILC technique vs. MPLC in several important aspects. With regard to operative times, SILC typically takes about 40–80 min depending on degree of dissection difficulty. This has been shown to be significantly longer compared to MPLC by approximately 15–20 min [13, 136]. Initial SSRC cohorts have reported a mean overall operative time of approximately 70–100, with console time ranging from 30 to 65 min [15–20]. These times will likely decrease as surgeons and dedicated operative room staffs become familiar with the robotic platform.
The overall main benefit of SILC compared to MPLC is improved patient satisfaction, cosmesis, and potentially pain control. Many studies confirm that SILC is as safe and efficacious as MPLC, showing comparable complication rates including minor (e.g., wound infection, seromas) and major complications (e.g., bile duct injury). While some studies report increased rates of bile duct injury and hernia formation, more studies are warranted to confirm these findings. Additionally, it is difficult to control for individual surgeon skill in these analyses. Thus, the literature should only be used as a guideline based on one’s own expertise and comfort level with single-incision surgery.
Integration into Practice
Perhaps the most important aspect of implementing a novel technique is incorporating it into surgical practice safely. Several studies have addressed the observed learning curve of experienced laparoscopic surgeons in developing an efficient operative time for SILC while maintaining patient safety and operative success. The data suggest there is an initial 20-patient SILC training phase, which have the longest operative times. The operative time improves gradually over the subsequent 20 patients. After the 40th case, surgeons’ operative times reach a nadir, stabilize, and become reproducible [141]. These results are analogous to another single surgeon’s initial experience [142]. This study observed that the initial mean operative time for the first 20 patients was 91 min. However, this improved to 81 min for the second 20 patients, followed by 64 min in the final series of patients. Furthermore, other studies describe learning curves that showed an improvement and plateau in operative times after only ten patients [143, 144]. Most importantly, regardless of the learning curve, these studies did not observe any increase in complication rate compared to MPLC.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








