Obesity as a Disease
David R. Saxon
Daniel H. Bessesen
CASE STUDY
A 45-year-old man with obesity (body mass index [BMI] 39 kg/m2) and prediabetes is frustrated with his weight and comes to clinic asking for help. He noticed a progressive weight gain of nearly 50 lb over the last 11 years. He had not been paying much attention to his diet or physical activity until he was diagnosed with prediabetes 6 months ago with an HbA1c of 6.0%. Since then, he reports following a strict diet of 1,200 kcal/day and running on a treadmill 4 days/week for 30 minutes each session. While he initially lost 15 lb (5% of his starting weight), his weight has stabilized over the past 3 months despite adhering to his diet and exercise routine. Both his mother and father have obesity and type 2 diabetes. His best friend from work weighs about the same that he does now and yet “can eat whatever he wants and he never exercises.” He is frustrated and comes in today to see if he has a metabolic problem. He wants to know why it is so difficult for him to lose weight. On examination, his blood pressure is 132/86, heart rate 88, and BMI 39 kg/m2. The remainder of the examination is unremarkable.
CLINICAL SIGNIFICANCE
Obesity is often viewed differently than diabetes or hypertension by patients, the general public, and even healthcare professionals (HCPs). The unstated belief that the problem is simply a result of poor lifestyle choices can lead to weight bias and suboptimal care. A greater understanding of the physiology behind weight regulation and the adaptive responses to weight loss that promote weight regain can help patients and HCPs have a more realistic view of the complexity of the problem and the challenges of treatment. While labeling obesity as a disease is useful, there are also limitations to this designation including controversies about “metabolically healthy obesity” and the “obesity paradox.” However, seeing the problem of obesity as a biological problem that can be addressed with a range of treatments from lifestyle to medications and surgery will be more productive and effective for HCPs and patients alike.
EPIDEMIOLOGY
Obesity is defined as a BMI ≥30 kg/m2 and class 3 obesity as a BMI ≥40 kg/m2. There are two data sets that provide information on the prevalence of obesity in the United States. The National Health and Nutrition Examination Survey (NHANES) directly measures height and weight annually in a nationally representative sample of 5,000 people of all ages. Because the heights and weights are directly measured, this is the most reliable data set for determining obesity prevalence; however the limited sample size cannot provide data at a state level. The most recent data from NHANES demonstrate that the prevalence of obesity among US adults aged 20 years and older increased from 26.4% in 2006 to 42.4% in 2018. The second data set is the Behavioral Risk Factor Surveillance System (BRFSS), which annually surveys a nationally representative sample of >400,000 adults by phone to get self-reported height and weight among other health data. Because people tend to overreport height and underreport weight, the data from this survey likely underestimates actual prevalence rates. However, a recent analysis used a statistical approach to correct the BRFSS data for the limitations of self-reported height and weight. The results of this analysis predict that by 2030, 48.9% of adults in the United States will have obesity and that 24.2% of adults will have severe obesity (Figure 1.1). This analysis further predicted that severe obesity will
become the most common BMI category (among four: normal weight, overweight, obesity, severe obesity) affecting women (27.6%), non-Hispanic black adults (31.7%), and low-income adults (31.7%) and thereby demonstrating the important effects of race/ethnicity and income on obesity prevalence. Obesity is common and growing in prevalence making it an important issue to address in primary care.
become the most common BMI category (among four: normal weight, overweight, obesity, severe obesity) affecting women (27.6%), non-Hispanic black adults (31.7%), and low-income adults (31.7%) and thereby demonstrating the important effects of race/ethnicity and income on obesity prevalence. Obesity is common and growing in prevalence making it an important issue to address in primary care.
 FIGURE 1.1 The estimated prevalence of obesity (BMI > 30 to 35 kg/m2) (A) or severe obesity (BMI > 35 kg/m2) (B) in each state from 1990 to 2030. BMI, body mass index. (Reprinted from Ward ZJ, Bleich SN, Cradock AL, et al. Projected U.S. state-level prevalence of adult obesity and severe obesity. N Engl J Med. 2019;381:2440-2450.) |
VALUE OF LABELING OBESITY A DISEASE
In 2013, the American Medical Association voted to recognize obesity as a disease that requires prevention and treatment. A large number of professional societies and organizations including the World Health Organization, National Institutes of Health, American College of Physicians, American Academy of Family Physicians, American Heart Association, and American Diabetes Association (ADA) among many others have agreed that obesity is a disease. The purpose of recognizing obesity as a disease was, in part, to shift the stigma that obesity is simply due to personal failings or lack of responsibility, to the modern concept that obesity results from a complex interaction of multiple factors including genetics, biology, behavior, and environment. In doing so, the medical community hoped that a greater investment would be made in understanding the causes of obesity, which would eventually lead to improved prevention and treatment options. Recognition that obesity is a disease has implications for patients, clinicians, and healthcare systems.1
For Patients
From a patient perspective, the value of recognizing obesity as a disease is that it may better allow patients to accept the disorder, seek treatment, and have treatment covered by insurance. One major barrier to obesity treatment has been that some HCPs either stigmatize patients with obesity (both implicitly and explicitly) or fail to offer obesity treatment within routine practice environments. Categorizing obesity as a disease opens up the opportunity for it to be treated in the same manner as other common metabolic diseases like diabetes, hypertension, and hyperlipidemia. Lack of consistent insurance coverage for treatment of obesity is an additional barrier from the patient perspective.
For Healthcare Professionals
From a provider perspective, the recognition of obesity as a disease opens the door to treating the condition within the context of routine clinical practice. For many HCPs, this is appealing since many of the conditions they spend time treating are a direct consequence of increased body weight. Although training in weight management and nutrition have historically been lacking for physicians, obesity competencies have been developed for undergraduate and graduate medical education,2 and there has been an exponential growth in the number of physicians seeking certification in obesity medicine (ABOM.org) by attaining additional continuing medical education (CME) credits in obesity-related educational content.3 With obesity recognized as a disease, clinicians are more readily able to have meaningful conversations with patients about their weight and about newer treatment approaches like antiobesity medications and bariatric surgery. Accordingly, recent treatment guidelines from the ADA have begun to shift toward a more obesity-focused approach.4 This includes prioritization of diabetes medications such as glucagon-like peptide 1 (GLP-1) agonists and sodium-glucose cotransporter-2 (SGLT-2) inhibitors that promote weight loss as well as recommendations for frequent discussions about lifestyle management and bariatric surgery (if indicated) during visits that are focused on diabetes management.
For the Healthcare System
Lastly, from a healthcare system perspective, the value of identifying obesity as a disease is that it promotes increased reimbursement for weight management care. Increased coverage of obesity care is beneficial in that it potentially allows systems to mobilize and centralize resources around population health and clinical obesity-focused care. Since comprehensive weight management requires a multidisciplinary team approach, reimbursement for weight management commensurate with that received for the treatment of other chronic health conditions allows systems to focus on identifying resources to build teams and clinics that can address weight management. Currently, the US healthcare system does not consistently reimburse for the treatment of obesity, creating additional hurdles for HCPs and patients.
LIMITATIONS TO LABELING OBESITY AS A DISEASE
Stigma
Some patients may find the designation of their weight as a “disease” stigmatizing. While many patients want to discuss weight management with their HCPs, they often do not appreciate a clinician “blaming everything on my weight.” For many, being labeled as “a person with obesity” is taken as a criticism.5 Therefore, clinicians should be sensitive to ask permission to discuss weight with patients and in general avoid the use of the term
obesity in favor of a more neutral term like “weight.” Also, caution should be taken in labeling 40% of the US population with a chronic disease. Not all individuals with a BMI ≥ 30 kg/m2 would consider themselves to have a chronic disease if they are in good overall health.
obesity in favor of a more neutral term like “weight.” Also, caution should be taken in labeling 40% of the US population with a chronic disease. Not all individuals with a BMI ≥ 30 kg/m2 would consider themselves to have a chronic disease if they are in good overall health.
Metabolically Healthy Obesity
Labeling something as a disease suggests that those with the condition will invariably suffer adverse consequences. While many people with obesity do develop comorbid conditions, a substantial minority of people with obesity have normal blood glucose, normal blood pressure, no evidence of cardiovascular disease, and no other problems attributable to their weight. A number of studies have shown that these so-called metabolically healthy obese (MHO) individuals have a lower risk of mortality than normal weight individuals who are metabolically unhealthy.6 However, long-term follow-up of MHO individuals shows that they do develop more metabolic illness than those of normal weight who were metabolically healthy.7 It may be that these MHO individuals have higher levels of cardiorespiratory fitness that mediates reduced risk for health problems.8 These data highlight that the health risks of obesity are variable and that BMI alone is not an adequate predictor of who will develop complications and, as a result, who is most likely to benefit from weight loss treatment. The issue of risk assessment and stratification is addressed in more detail in Chapter 3.
The Obesity Paradox
A large number of studies have been conducted to identify the relationship between BMI and adverse health consequences. These studies have consistently shown that obesity is associated with an increased risk for developing hypertension, coronary heart disease, heart failure, and atrial fibrillation among other weight-related illnesses. However, when studies examining the relationship between BMI and mortality in individuals with established heart disease, investigators were surprised to find the opposite relationship: individuals with established heart failure, coronary artery disease, acute myocardial infarction, and those with atrial fibrillation with obesity had lower mortality than those of normal weight.9,10 This protective effect of obesity, though counterintuitive, has been seen in numerous studies and confirmed in meta-analyses. In addition to these cardiovascular diseases, evidence of an obesity paradox has been found in end-stage renal disease, diabetes, renal cell carcinoma, and even ICU-related mortality. How can obesity be a disease if having the condition lowers the risk of mortality in those with associated medical illness? This area has been the subject of much ongoing controversy, but emerging data suggest that the obesity paradox may be more relevant in individuals with low levels of cardiorespiratory fitness.10
Aging and Sarcopenia
There is a curvilinear relationship between BMI and mortality. Specifically, mortality is increased in those with both low and high BMIs compared with those in the middle range of BMIs. This curvilinear relationship changes as people age with low BMIs becoming associated with greater risk and high BMI levels having a smaller relative increase in mortality in people older than 65 years.11 This may in part be a reflection of the fact that body composition plays an important role in determining health. Individuals with reduced lean mass or sarcopenia (which lowers BMI), especially in older individuals, have an increased mortality while those with increased fat mass and reduced lean mass have the highest mortality.12 Since weight loss often leads to a loss in both fat and lean mass and the risks of being overweight or obese are less in older individuals, the benefits of weight loss are less clear in older people.
COMMON MISCONCEPTIONS ABOUT OBESITY AND ITS TREATMENT
Changing the view of obesity from a problem of lifestyle choices to that of a chronic, often progressive metabolic disease leads to changes in the way we talk about treatment. Many patients initially are looking to make short-term drastic dietary changes to lose weight without understanding that changes need to be long-term and sustainable. It is important for clinicians to emphasize this point up front when discussing treatment. Many people also think that exercise alone is an effective method to lose weight. While increased levels of physical activity are undoubtedly good for health, consideration of the number of calories that can be burned with 30 to 60 minutes of moderate intensity exercise for someone who is not physically fit will make it clear how difficult it is to create an energy deficit through exercise alone. This issue is discussed in detail in Chapter 6. Importantly, most people seeking treatment are unaware that weight is regulated, that there is a biological tendency for progressive weight gain through adult life, and that if a person loses weight, the body responds with adaptive responses that promote weight regain. They assume that if caloric restriction is sustained, weight will continue to fall. The idea of a “weight plateau” as a new steady state is a concept that most patients have not considered. A fuller understanding of the nature of weight regulation can lead to an acknowledgment that treatment needs to be ongoing and a consideration for more aggressive therapies such
as a structured approach to diet, prescription of antiobesity medications, or surgery as potentially reasonable treatment options.
as a structured approach to diet, prescription of antiobesity medications, or surgery as potentially reasonable treatment options.
BODY WEIGHT AS A REGULATED PARAMETER
One might initially think that a person could choose what they weigh by making voluntary decisions about food intake and physical activity. On closer consideration, and with some simple math, it is clear that each of us eats somewhere between 15,000 and 25,000 lb of food over the course of our adult life during which our weight changes on average only 1 to 2 lb/year. When examined over long periods of time, the maintenance of energy balance is remarkably accurate despite dramatic variations in day-to-day levels of energy intake and expenditure. It is clear that maintaining long-term energy balance, weight, fat mass, or some related parameter is the focus of a highly complex regulatory system.13 However, it is not clear exactly what parameter is being regulated. The observed biology suggests the regulation is not around a “set point” but rather around a “trajectory” of gradual weight gain over the life span with a modest decline in weight in the later years of life. The rate of weight gain tends to be higher in individuals with a greater genetic propensity for obesity and lower in those with constitutional thinness. It also varies depending on qualities of the environment including access to highly palatable food and opportunities for physical activity.
Body weight is largely determined by the energy balance equation, where the currency of energy-in and energy-out is in the form of calories. If there is an imbalance between energy intake and energy expenditure, weight will change. Either the person will lose weight (energy contained in fat stores is consumed) or will gain weight (energy contained in fat stores will increase). How do the body and brain govern and respond to changes in energy balance? An understanding of this regulation requires a more detailed consideration of the components of energy expenditure and the regulation of energy balance by the brain.
Energy Expenditure (the Burning of Calories)
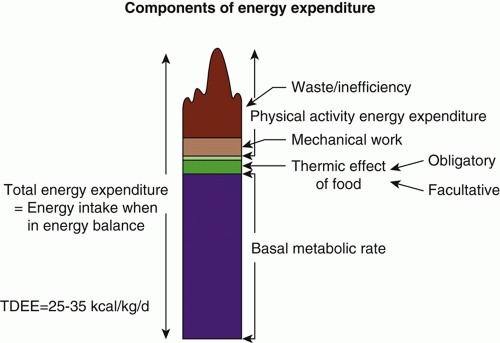
Total daily energy expenditure (TDEE) is the number of calories that the body burns per day and is an important number because it is equal to the number of calories per day that a person must consume to maintain weight. To lose weight, a person must consume fewer calories than TDEE. The components of TDEE are depicted in Figure 1.2. Basal metabolic rate (BMR) is the energy needed to support basic body functions such as maintaining heart and kidney function, body temperature, basal brain functioning, and ion gradients in cells. BMR accounts for about 65% to 75% of TDEE. BMR can be estimated with formulae such as the Mifflin-St Jeor equation, which takes into account sex, age, height, and weight (https://www.calculator.net/bmr-calculator.html). The thermic effect of feeding (TEF) is the energy requirements of food digestion and metabolism and accounts for about 8% of TDEE. The third and most variable component of TDEE is physical activity energy expenditure (PAEE). This can be energy expended in planned exercise or in so-called non-exercise activity thermogenesis—the calories burned by daily body movements. Because skeletal muscle and vital organs are the biggest energy consumers in the body, TDEE is linearly related to lean body mass (Figure 1.3), although there is some interindividual variability in this relationship. Women have lower energy requirements than men and TDEE declines for everyone as we age by roughly 5 cal/year. While this may seem like a small number, over 40 to 50 years, it amounts to a reduction in TDEE of several hundred calories. TDEE can be estimated by multiplying the calculated BMR by an activity factor of 1.2 to 1.5 based on the amount of PAEE a person does.
The linear nature of the relationship between energy expenditure and lean body mass means that people with obesity consume more energy than thin people. Many people who struggle with their weight report eating very little and not losing weight. A classic study by Lichtman measured energy expenditure and energy intake in a group of these people.14 They found no evidence of “metabolic efficiency” as a cause of obesity. Rather they found strong evidence of underreporting of food intake in all of the subjects studied, as well as overreporting of physical activity levels. What is a clinician to do with this information? It may be that when people with obesity report low levels of energy intake, they are either
not correctly perceiving how much they eat or they are relating “response bias” (they may feel pressure to give answers that are socially acceptable or desirable). It is also possible that energy density plays a role in the underreporting of calorie intake. Specifically, patients with obesity are not eating large volumes of food, but the foods they are eating are high in energy density (more fried foods and processed/snack foods, fewer vegetables). It is generally not useful to try to convince a person that they are actually eating more than they think they are or that they are being untruthful. Rather it is typically more helpful to discuss how difficult it is to know what we are eating unless we consciously monitor food intake with a diet record. Rather than challenging the person’s perception of their food intake, the HCP can focus on the value of the person’s current effort to lose weight, discuss available treatment options, and emphasize that a new course of action will be needed if the person is going to lose weight.
not correctly perceiving how much they eat or they are relating “response bias” (they may feel pressure to give answers that are socially acceptable or desirable). It is also possible that energy density plays a role in the underreporting of calorie intake. Specifically, patients with obesity are not eating large volumes of food, but the foods they are eating are high in energy density (more fried foods and processed/snack foods, fewer vegetables). It is generally not useful to try to convince a person that they are actually eating more than they think they are or that they are being untruthful. Rather it is typically more helpful to discuss how difficult it is to know what we are eating unless we consciously monitor food intake with a diet record. Rather than challenging the person’s perception of their food intake, the HCP can focus on the value of the person’s current effort to lose weight, discuss available treatment options, and emphasize that a new course of action will be needed if the person is going to lose weight.
 FIGURE 1.3 Relationship between total daily energy expenditure (TDEE) and lean body mass. Each point on this figure comes from the measurement of 24-hour energy expenditure by indirect calorimetry in one person. The figure demonstrates both the linear relationship of TDEE with lean body mass and the interindividual variability in this relationship. (Reprinted from Weyer C, Snitker S, Rising R, Bogardus C, Ravussin E. Determinants of energy expenditure and fuel utilization in man: effects of body composition, age, sex, ethnicity and glucose tolerance in 916 subjects. Int J Obes Relat Metab Disord. 1999;23(7):715-722.) |
Appetite Regulation
The other side of the energy balance equation is food intake. A great deal of progress has been made over the last 20 years on the role various brain regions play in regulating energy balance.15 The hypothalamus has been a particular focus of study since the discovery of leptin in 1994. The arcuate nucleus of the hypothalamus plays a central role in sensing energy balance through a number of hormones including insulin, leptin, and ghrelin. This brain region functions as a classical feedback system sensing energy balance and in the presence of positive energy balance, activating neural systems that decrease food intake and increase energy expenditure (catabolic pathways). The opposite occurs in the presence of negative energy balance, where activation of neural pathways leads to increased food intake and reduced energy expenditure (anabolic pathways, Figure 1.4). In the presence of sustained positive energy balance, fat mass expands and leptin, which is secreted from adipose tissue in proportion to adipose tissue mass, increases along with increased circulating levels of insulin. These two hormones act on two populations of neurons in the arcuate nucleus of the hypothalamus to regulate energy balance. A number of neurotransmitters have been identified in the hypothalamus that are involved in the catabolic and anabolic pathways. Specifically, neuropeptide Y and agouti-related peptide increase food intake and decrease energy expenditure (Figure 1.5). Conversely, proopiomelanocortin (POMC) and cocaine- and amphetamine-related transcript decrease food intake and increase energy expenditure. When weight is lost, leptin and insulin levels fall, and the activities in these pathways change in a manner that promotes food intake and reduces energy expenditure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree