CHAPTER 114
Gouty Arthritis, Acute
Presentation
Usually, a middle-aged male patient with an established diagnosis of gout or hyperuricemia rapidly develops an intensely painful monarticular arthritis, often in the middle of the night, but sometimes a few hours following a minor trauma. Any joint may be affected, usually of the lower limb, including the ankle, knee, and tarsal joints, but most common is the metatarsophalangeal joint of the great toe (podagra). The joint is red, hot, swollen, and intensely tender to touch or movement. There is usually no fever, rash, or other sign of systemic illness, although low-grade fever, leukocytosis, and an elevation of the erythrocyte sedimentation rate may occur. The patient may have predisposing factors that increase his risk for developing gout, such as obesity, moderate to heavy alcohol intake, high blood pressure, diabetes, and abnormal kidney function, or he may be taking certain drugs, including thiazide diuretics, low-dose aspirin, and tuberculosis medications (pyrazinamide and ethambutol).
What To Do:
 If the patient has not been previously diagnosed by arthrocentesis that showed crystals, tap the involved joint as described for acute monarticular arthritis (see Chapter 120). In addition to ruling out infection, look under the microscope for crystals in the joint fluid. Urate crystals look like needles and may be in white cells. The calcium pyrophosphate dihydrate crystals of pseudogout are rhomboids. Polarizing filters above and below the sample help distinguish the strongly negative birefringent crystals of sodium urate from the weakly positive birefringent calcium pyrophosphate dehydrate (Figure 114-1). The synovial fluid is typically inflammatory with a white blood cell (WBC) count ranging from 5000 to 50,000/mL, predominantly neutrophils. Higher counts should always raise the question of an infection.
If the patient has not been previously diagnosed by arthrocentesis that showed crystals, tap the involved joint as described for acute monarticular arthritis (see Chapter 120). In addition to ruling out infection, look under the microscope for crystals in the joint fluid. Urate crystals look like needles and may be in white cells. The calcium pyrophosphate dihydrate crystals of pseudogout are rhomboids. Polarizing filters above and below the sample help distinguish the strongly negative birefringent crystals of sodium urate from the weakly positive birefringent calcium pyrophosphate dehydrate (Figure 114-1). The synovial fluid is typically inflammatory with a white blood cell (WBC) count ranging from 5000 to 50,000/mL, predominantly neutrophils. Higher counts should always raise the question of an infection.
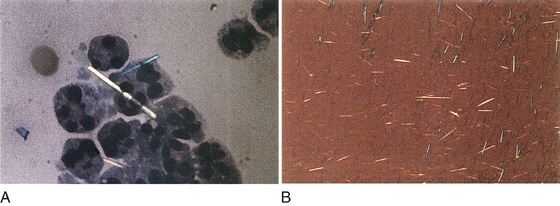
Figure 114-1 A and B, Microscopic example of monosodium urate monohydrate microcrystals under polarized light and under light microscopy. (Adapted from Knoop KJ, Stack LB, Storrow AB: Atlas of emergency medicine, ed 2. New York, 2002, McGraw-Hill.)
 Radiographs may be obtained but are only likely to be helpful in the late stages of the disease (Figure 114-2) or if other underlying disease is in question (e.g., pseudogout, tumor).
Radiographs may be obtained but are only likely to be helpful in the late stages of the disease (Figure 114-2) or if other underlying disease is in question (e.g., pseudogout, tumor).
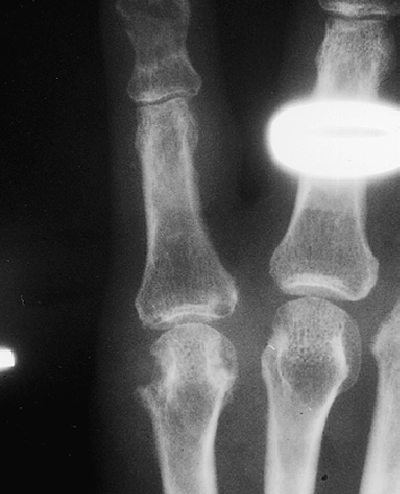
Figure 114-2 The fifth metacarpophalangeal joint in this radiograph is fairly characteristic of the late stages of gout. Marginal erosions of the metacarpal head result in prominent “overhanging” edges. (Adapted from Yu J: Musculoskeletal imaging. St Louis, 2001, Mosby.)
 In most middle-aged patients with acute gout who are otherwise healthy, nonsteroidal anti-inflammatory drugs (NSAIDs) are the treatment of choice.
In most middle-aged patients with acute gout who are otherwise healthy, nonsteroidal anti-inflammatory drugs (NSAIDs) are the treatment of choice.
 Provide rapid pain relief with loading doses of NSAIDs, such as ketorolac (Toradol), 60 mg IM; indomethacin (Indocin), 50 mg PO tid; ibuprofen (Motrin), 800 mg PO qid; or naproxen sodium (Anaprox), 550 mg PO bid, then tapering after 24 to 48 hours, once pain has subsided, to maintenance doses for the next 5 to 7 days (e.g., indomethacin, 25 mg tid; ibuprofen, 600 mg qid; naproxen, 275 mg bid). Excruciating pain may require one dose of narcotics while the anti-inflammatory drugs take effect. All NSAIDs have been shown to be equally effective. The choice of NSAID is not as important as initiating therapy early in an attack. They should be used with caution or not at all in patients with any of the following: significant renal impairment (creatinine > 2), poorly controlled congestive heart failure, history of or active peptic ulcer disease, anticoagulation therapy, or hepatic dysfunction.
Provide rapid pain relief with loading doses of NSAIDs, such as ketorolac (Toradol), 60 mg IM; indomethacin (Indocin), 50 mg PO tid; ibuprofen (Motrin), 800 mg PO qid; or naproxen sodium (Anaprox), 550 mg PO bid, then tapering after 24 to 48 hours, once pain has subsided, to maintenance doses for the next 5 to 7 days (e.g., indomethacin, 25 mg tid; ibuprofen, 600 mg qid; naproxen, 275 mg bid). Excruciating pain may require one dose of narcotics while the anti-inflammatory drugs take effect. All NSAIDs have been shown to be equally effective. The choice of NSAID is not as important as initiating therapy early in an attack. They should be used with caution or not at all in patients with any of the following: significant renal impairment (creatinine > 2), poorly controlled congestive heart failure, history of or active peptic ulcer disease, anticoagulation therapy, or hepatic dysfunction.
 Corticosteroids are effective in the treatment of gout. When used appropriately for a short duration, they are a safe alternative for patients in whom NSAIDs are contraindicated. Care must be used in patients with diabetes, but in recent years, corticosteroids have been used more often in older patients with multiple comorbid conditions because of their low-toxicity profile.
Corticosteroids are effective in the treatment of gout. When used appropriately for a short duration, they are a safe alternative for patients in whom NSAIDs are contraindicated. Care must be used in patients with diabetes, but in recent years, corticosteroids have been used more often in older patients with multiple comorbid conditions because of their low-toxicity profile.
 For a monoarticular flare-up, an intra-articular injection of a long-acting corticosteroid is often the safest treatment. Delay injecting corticosteroids into the joint until the possibility of infection is eliminated (see Chapter 120). After draining as much fluid as possible from the joint, using aseptic techniques, inject betamethasone (Celestone Soluspan), 1 mg (small joints) to 4 mg (large joints), or methylprednisolone (Depo-Medrol), about 20 to 40 mg mixed in an appropriate volume (depending on joint size) of bupivacaine (Marcaine) 0.5% to provide immediate pain relief.
For a monoarticular flare-up, an intra-articular injection of a long-acting corticosteroid is often the safest treatment. Delay injecting corticosteroids into the joint until the possibility of infection is eliminated (see Chapter 120). After draining as much fluid as possible from the joint, using aseptic techniques, inject betamethasone (Celestone Soluspan), 1 mg (small joints) to 4 mg (large joints), or methylprednisolone (Depo-Medrol), about 20 to 40 mg mixed in an appropriate volume (depending on joint size) of bupivacaine (Marcaine) 0.5% to provide immediate pain relief.
 Oral corticosteroids may also be used. Start with 20 to 40 mg of prednisone (Deltasone) and then taper throughout 8 days. If tapered too rapidly, a rebound flare-up of gout may occur.
Oral corticosteroids may also be used. Start with 20 to 40 mg of prednisone (Deltasone) and then taper throughout 8 days. If tapered too rapidly, a rebound flare-up of gout may occur.
 An alternative to oral steroids is a single intramuscular injection of methylprednisolone (Depo-Medrol), 40 mg. This has no therapeutic advantage over oral dosing but is useful if the patient cannot take oral medications.
An alternative to oral steroids is a single intramuscular injection of methylprednisolone (Depo-Medrol), 40 mg. This has no therapeutic advantage over oral dosing but is useful if the patient cannot take oral medications.
 Adrenocorticotropic hormone (ACTH), 80 units IM, is also effective and can be used in patients with multiple medical problems, including congestive heart failure, chronic renal insufficiency, and peptic ulcer disease. Its use is limited by patient comfort (IM administration), cost, and availability.
Adrenocorticotropic hormone (ACTH), 80 units IM, is also effective and can be used in patients with multiple medical problems, including congestive heart failure, chronic renal insufficiency, and peptic ulcer disease. Its use is limited by patient comfort (IM administration), cost, and availability.
 An alternative treatment for acute gouty arthritis within the first 12 to 24 hours of an attack is colchicine, 0.6 mg PO qh, until pain is relieved, the patient develops nausea, vomiting, or diarrhea, or a maximum dose of 6 mg is reached. Often patients experience adverse gastrointestinal (GI) effects before relief of gout symptoms. Colchicine can also be given IV, 2 mg q6h to a maximum of 4 mg. After these maximum doses, no more colchicine should be prescribed for 1 week to avoid toxicity. IV administration can cause anaphylaxis, and extravasation can cause tissue necrosis. At high doses, colchicine is bone marrow–suppressive and with renal insufficiency or patients taking cyclosporine or statins, colchicine can cause neuromyopathy. Because of its small benefit-to-toxicity ratio, colchicine should only be considered if there is no alternative therapy.
An alternative treatment for acute gouty arthritis within the first 12 to 24 hours of an attack is colchicine, 0.6 mg PO qh, until pain is relieved, the patient develops nausea, vomiting, or diarrhea, or a maximum dose of 6 mg is reached. Often patients experience adverse gastrointestinal (GI) effects before relief of gout symptoms. Colchicine can also be given IV, 2 mg q6h to a maximum of 4 mg. After these maximum doses, no more colchicine should be prescribed for 1 week to avoid toxicity. IV administration can cause anaphylaxis, and extravasation can cause tissue necrosis. At high doses, colchicine is bone marrow–suppressive and with renal insufficiency or patients taking cyclosporine or statins, colchicine can cause neuromyopathy. Because of its small benefit-to-toxicity ratio, colchicine should only be considered if there is no alternative therapy.
 Colchicine is usually used at low doses, 0.6 mg once or twice daily, to prevent attacks or rebound flare-ups in patients in whom steroids are being tapered or urate-lowering therapy is being started. It should still be used with caution in older patients with reduced renal function.
Colchicine is usually used at low doses, 0.6 mg once or twice daily, to prevent attacks or rebound flare-ups in patients in whom steroids are being tapered or urate-lowering therapy is being started. It should still be used with caution in older patients with reduced renal function.
 It should be remembered that gout is a self-limited disease. At times, the risks of certain treatments may outweigh the benefits, especially in elderly patients.
It should be remembered that gout is a self-limited disease. At times, the risks of certain treatments may outweigh the benefits, especially in elderly patients.
 Instruct the patient to elevate and rest the painful extremity, apply ice packs, and arrange for follow-up. In gouty arthritis, cold applications, in addition to being a useful adjuvant treatment, are helpful for discriminating gout from other forms of inflammatory arthritis. Topical ice has been shown to help relieve joint pain in patients with gouty arthritis but not in patients with other inflammatory arthritides.
Instruct the patient to elevate and rest the painful extremity, apply ice packs, and arrange for follow-up. In gouty arthritis, cold applications, in addition to being a useful adjuvant treatment, are helpful for discriminating gout from other forms of inflammatory arthritis. Topical ice has been shown to help relieve joint pain in patients with gouty arthritis but not in patients with other inflammatory arthritides.
 Patients should be informed about the factors contributing to their hyperuricemia, such as obesity, a high-purine diet, regular alcohol consumption, and diuretic therapy, which may all be correctable.
Patients should be informed about the factors contributing to their hyperuricemia, such as obesity, a high-purine diet, regular alcohol consumption, and diuretic therapy, which may all be correctable.
 Urate-lowering therapy with probenecid or allopurinol is considered cost effective for patients who have two or more attacks of gout per year. Some physicians advocate using this treatment in patients who experience more than four attacks per year.
Urate-lowering therapy with probenecid or allopurinol is considered cost effective for patients who have two or more attacks of gout per year. Some physicians advocate using this treatment in patients who experience more than four attacks per year.
 Most patients will note improvement within the first 12 to 24 hours, with resolution of symptoms in the next 7 to 10 days.
Most patients will note improvement within the first 12 to 24 hours, with resolution of symptoms in the next 7 to 10 days.
What Not To Do:
 Do not depend on serum uric acid to diagnose acute gouty arthritis—it may or may not be elevated (>8 mg/dL) at the time of an acute attack. Hyperuricemia will be found in 70% of patients with their first attack of gout.
Do not depend on serum uric acid to diagnose acute gouty arthritis—it may or may not be elevated (>8 mg/dL) at the time of an acute attack. Hyperuricemia will be found in 70% of patients with their first attack of gout.
 Do not use NSAIDs when a patient has a history of active peptic ulcer disease with bleeding. Relative contraindications include renal insufficiency, volume depletion, gastritis, inflammatory bowel disease, asthma, and congestive heart disease.
Do not use NSAIDs when a patient has a history of active peptic ulcer disease with bleeding. Relative contraindications include renal insufficiency, volume depletion, gastritis, inflammatory bowel disease, asthma, and congestive heart disease.
 Do not insist on reconfirming an established diagnosis of gout by ordering serum uric acid levels (which are often normal during the acute attack) or tapping an exquisitely painful joint at every attack in a patient with known gout and a typical presentation.
Do not insist on reconfirming an established diagnosis of gout by ordering serum uric acid levels (which are often normal during the acute attack) or tapping an exquisitely painful joint at every attack in a patient with known gout and a typical presentation.
 Do not, on the other hand, miss a septic arthritis in a patient with gout who is toxic and has a high fever, an elevated WBC count, an identified source of infection, or comorbidities, such as diabetes, alcohol abuse, and advanced age. Septic arthritis carries the potential for a high morbidity and mortality.
Do not, on the other hand, miss a septic arthritis in a patient with gout who is toxic and has a high fever, an elevated WBC count, an identified source of infection, or comorbidities, such as diabetes, alcohol abuse, and advanced age. Septic arthritis carries the potential for a high morbidity and mortality.
 Do not attempt to reduce the serum uric acid level with probenecid or allopurinol during an acute attack of gouty arthritis. This will not help the arthritis and may even be counterproductive. Leave it for follow-up. Because of the high frequency of comorbid conditions and decreased life expectancy in elderly patients, it may be less important to institute urate-lowering therapy in these patients than in younger patients with many years of cumulative attacks and joint damage in their future.
Do not attempt to reduce the serum uric acid level with probenecid or allopurinol during an acute attack of gouty arthritis. This will not help the arthritis and may even be counterproductive. Leave it for follow-up. Because of the high frequency of comorbid conditions and decreased life expectancy in elderly patients, it may be less important to institute urate-lowering therapy in these patients than in younger patients with many years of cumulative attacks and joint damage in their future.
 Do not stop urate-lowering maintenance therapy during an acute attack. In these cases, therapy should be continued, and the acute gouty flare treated in the usual manner.
Do not stop urate-lowering maintenance therapy during an acute attack. In these cases, therapy should be continued, and the acute gouty flare treated in the usual manner.
Discussion
Among mammals, only humans and other primate species excrete uric acid as the end product of purine metabolism. This is because humans and primates lack the enzyme uricase, which converts uric acid to allantoin, a more soluble excretory product.
When overproduction or underexcretion of uric acid occurs, the serum urate concentration may exceed the solubility of urate (a concentration of approximately >6.8 mg/dL), and supersaturation of urate in the serum and other extracellular spaces results. This state (called hyperuricemia), increases the risk for crystal deposition of urate, from the supersaturated fluids, in tissues. Hyperuricemia is defined as a serum uric acid level of more than 7.0 mg/dL in men or more than 6.0 mg/dL in women.
Hyperuricemia is clearly associated with an increased risk for the development of gout, although most patients with hyperuricemia are asymptomatic and never develop gout. In the general population, 80% to 90% of gout patients are underexcreters, although renal function is otherwise normal. The risk for the development of gout increases with increasing serum uric acid level.
In younger patients, hyperuricemia and gout are overwhelmingly observed in men. The initial attack of gout is monarticular in 85% to 90% of patients. Lower-extremity joints are usually affected, with approximately 60% of first attacks involving the first metatarsophalangeal joints. Attacks may last from a few days to 2 to 3 weeks without treatment, with a gradual resolution of all inflammatory signs and a return to apparent normalcy. An “intercritical” period, lasting weeks to months, may elapse before a new attack occurs in the same or another joint. Without specific therapy, a second attack will occur in 78% of patients within 2 years, and in 93% within 10 years. Over subsequent years, attacks occur more frequently and may be polyarticular and associated with fever and constitutional symptoms. Tophaceous deposits become apparent over the elbows, fingers, or other areas over the years (tophaceous gout), and chronic polyarticular arthritis may develop, which is often less severe, sometimes resembling rheumatoid arthritis or degenerative joint disease. Distal interphalangeal (DIP) joint involvement is a little more common than proximal interphalangeal (PIP) involvement, and tophaceous deposits on Heberden nodes can often be confused with osteoarthritis.
In patients older than 60 years with newly diagnosed gout, approximately 50% are women. Elderly women may have more finger involvement than men; 25% of women present with hand involvement and polyarticular disease.
Obesity, genetic predisposition, high intake of meat and seafood, hyperlipidemia, hypertension, and heavy alcohol use are associated with younger gout patients, whereas renal insufficiency, low-dose salicylates, and thiazide diuretic use are more often associated with elderly onset gout.
Transplant patients and patients on cyclosporine therapy are also at increased risk for developing gout, as are patients with myeloproliferative disorders, polycythemia vera, myeloid metaplasia, and chronic myelogenous leukemia.
Trauma, surgery, infection, and starvation as well as alcoholic or dietary indiscretions may provoke acute attacks. Acute attacks have been known to follow a game of golf, a long walk, or a hunting trip, leading to the name “pheasant hunter’s toe.” The solubility of uric acid decreases with a lower body temperature and in a lowering pH. These properties may provide an explanation for the increase in gouty attacks in the peripheral joints in cold weather.
Radiographs in gout characteristically demonstrate normal bone mineral density until the late stages of the disease. Well-marginated para-articular erosion with overhanging edges or margins is the characteristic lesion of chronic gouty arthritis.
The gold standard for establishing a definite diagnosis of gout is the presence of monosodium urate crystals in aspirated joint fluid or tophus.
Physicians often opt to reach a diagnosis based on clinical features and demonstration of hyperuricemia. No studies have been published on the usefulness or validity of any diagnostic clinical criteria. Serum uric acid levels are commonly elevated in patients without gout and can be normal or even low in patients with gout.
When it is impractical or not possible to obtain joint fluid, supportive data that can be used to make a diagnosis of gout include a history of gout; a typical clinical history of sudden onset of an exquisitely painful joint, classically the first metatarsophalangeal joint; a history of underlying renal disease or use of medications that cause hyperuricemia; an elevated serum urate level; radiologic evidence suggestive of gouty arthritis; and a favorable response to topical cold applications.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


