Antiserotonin Drugs in Migraine Prophylaxis
Peer Tfelt-Hansen
Pramod R. Saxena
Antiserotonin drugs are the first group of effective agents available for migraine prophylaxis. Originally, these drugs were thought to act via antagonism at serotonin (5-hydroxytryptamine; 5-HT) D receptors (38), now classified as 5-HT2 receptors (45). This view is, however, no longer tenable for several reasons. First, many selective and potent 5-HT2 receptor antagonists, including ketanserin, ICI 169,369, sergolexole, and mianserin, are either ineffective or only weakly effective in migraine (83). Second, the antimigraine potency of these drugs does not correlate with their affinity at the 5-HT2A, 5-HT2B, or 5-HT2C receptors (89). Furthermore, the antimigraine drugs ergotamine and dihydroergotamine have an agonist (not antagonist) action at the 5-HT2C receptor (11). Thus, although conceding that the title of this chapter may be a misnomer, from the outset we wish to emphasize that the prophylactic effect of antiserotonin drugs does not depend on their antiserotonin property. For further discussion of mechanisms, see the sections on individual drugs.
METHYSERGIDE
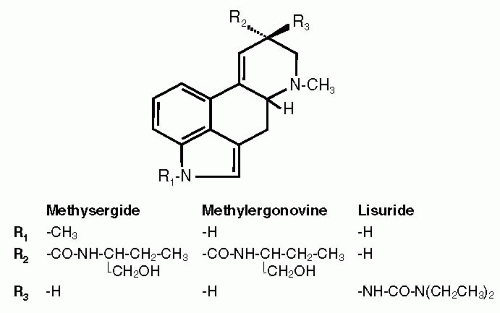
Methysergide is a semisynthetic compound derived from the ergot alkaloid methylergometrine by adding a methyl group at the indole nitrogen (Fig. 55-1). It was introduced in pharmacotherapy as a specific 5-HT receptor antagonist (29,87).
Pharmacokinetics
Pharmacokinetic studies in humans indicate that methysergide is probably a prodrug; its main metabolite is methylergometrine (10). After oral administration, the bioavailability of methysergide is about 13%, owing to a high degree of first-pass metabolic conversion to methylergometrine (see Fig. 55-1). Whereas the area under the plasma concentration curve (AUC) for methysergide and methylergometrine after intravenous administration of methysergide is in the same range, oral administration of methysergide results in 10 times greater AUC for methylergometrine than for the parent drug. The elimination half-lives of methysergide and methylergometrine are 60 and 220 minutes, respectively (10).
In contrast to methysergide, methylergometrine has dopaminergic activity (8). The metabolism of methysergide to methylergometrine probably explains why methysergide has little dopaminergic activity upon parenteral administration (8), but its oral administration can result in a significant decrease in the plasma prolactin level (32). Thus, when methysergide is used orally in humans, there are serotonergic effects both because of the parent drug and the metabolite methylergometrine, as well as some dopaminergic effects because of the metabolite methylergometrine.
Pharmacologic Background
It is well known that methysergide is a potent 5-HT2 receptor antagonist, but it does not distinguish between the 5-HT2A, 5-HT2B and 5-HT2C subtypes (45,89). Thus, methysergide antagonizes the contractile effects of 5-HT on vascular and nonvascular smooth muscles with a pA2 of more than 8 (67). Indeed, in the human isolated temporal artery, which contains predominantly 5-HT2 receptors (27), both methysergide and its active metabolite methylergometrine are potent antagonists; the latter compound is some 40 times more active than the parent drug (97).
In the early 1970s, Saxena et al. reported that the vasoconstrictor effect of 5-HT within the canine carotid vascular bed was not much modified by methysergide or by two other potent 5-HT2 receptor antagonists, mianserin and cyproheptadine (Fig. 55-2) (78,80,84). Therefore, the receptors for 5-HT in the external carotid vascular bed
appeared to be of a novel type, which were later named 5-HT1-like receptor (86). Interestingly, methysergide proved to be an agonist at these receptors (45) and selectively decreased carotid blood flow by constricting arteriovenous anastomoses (80,82). Although this effect of methysergide is much less marked than that of ergotamine or sumatriptan (23, 24, 25), its mediation by novel 5-HT1-like receptors undeniably provided incentive for the development of sumatriptan, which at the time of its introduction was regarded as a selective 5-HT1-like receptor agonist (48).
appeared to be of a novel type, which were later named 5-HT1-like receptor (86). Interestingly, methysergide proved to be an agonist at these receptors (45) and selectively decreased carotid blood flow by constricting arteriovenous anastomoses (80,82). Although this effect of methysergide is much less marked than that of ergotamine or sumatriptan (23, 24, 25), its mediation by novel 5-HT1-like receptors undeniably provided incentive for the development of sumatriptan, which at the time of its introduction was regarded as a selective 5-HT1-like receptor agonist (48).
As recently argued (85), 5-HT1-like receptor is now redundant because the composition of this heterogeneous group has been delineated. This group comprises the sumatriptan-insensitive 5-HT7 receptor mediating vasorelaxation (22,28), as well as sumatriptan-sensitive 5-HT1B, 5-HT1D, and, in some tissues, even 5-HT1F receptors. Methysergide is a potent antagonist at the 5-HT7 (and 5-HT2) receptor and an agonist at 5-HT1B and, possibly, also 5-HT1D receptors. In vitro functional and radioligand studies confirm that methysergide is an agonist at the 5-HT1B receptor (64, 65, 66).
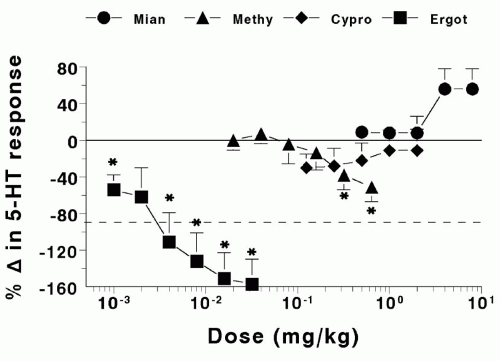 FIGURE 55-2. Dog external carotid vascular bed. Effect of mianserin (Mian), methysergide (Methy), cyproheptadine (Cypro), and ergotamine (Ergot) on the vasoconstrictor response to serotonin. Values below the interrupted line (i.e., change more than −100%) mean that serotonin caused vasodilatation instead of vasoconstriction. *Significant (P < .05) change compared with parallel administration of saline. Data from Saxena et al. (84) and Saxena (78). |
The pharmacology of the metabolite methylergometrine has been investigated less thoroughly. However, it is a more potent vasoconstrictor than methysergide both in vivo (60) and in vitro on, for example, canine saphenous veins and human basilar (66) and coronary (59) arteries (Fig. 55-3). The last two effects may bestow efficacy in migraine (63) and coronary side effect potential (53,68) to methylergometrine.
Last, chronic but not acute treatment with methysergide has been reported to attenuate dural plasma extravasation following electrical stimulation of the trigeminal ganglion in the rat (77). The discrepancy between the effect of acute and chronic treatment with methysergide in this model is most likely because of the presence of methylergometrine during chronic administration of methysergide. Although Saito et al. have implied a presynaptic inhibition of the release of calcitonin gene-related peptide (CGRP) from perivascular sensory nerves, functional antagonism (via vasoconstriction) of the vasodilator effects of CGRP cannot be ruled out. Indeed, such a functional antagonism between methysergide and CGRP has recently been described in the rabbit eye (52) and should be investigated further with the use of methylergometrine.
Possible Mechanism of Antimigraine Action
The mechanism of action of methysergide in migraine is not well understood. The efficacy of methysergide has been ascribed to its 5-HT2 receptor antagonist property, but this is unlikely because potent 5-HT2 receptor antagonists such as mianserin, sergolexole, ketanserin, and ICI 169,369 have little or no prophylactic effect in migraine, and for cyproheptadine the claimed efficacy (67) has never been
confirmed in controlled clinical trials. Therefore, it is highly improbable that 5-HT2 receptor antagonism plays any role in migraine prophylaxis (83,96). It should be noted that, in essence, these same arguments also apply against the advocated role of 5-HT2C receptor antagonism (methysergide is a potent 5-HT2C receptor antagonist [67]) in migraine (35,36).
confirmed in controlled clinical trials. Therefore, it is highly improbable that 5-HT2 receptor antagonism plays any role in migraine prophylaxis (83,96). It should be noted that, in essence, these same arguments also apply against the advocated role of 5-HT2C receptor antagonism (methysergide is a potent 5-HT2C receptor antagonist [67]) in migraine (35,36).
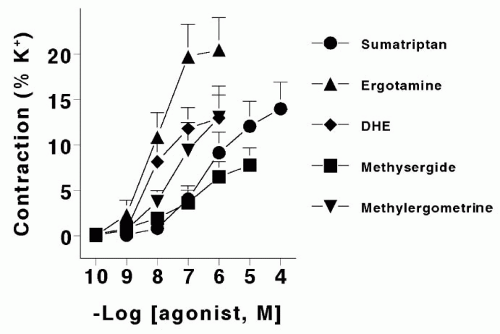 FIGURE 55-3. Concentration response (expressed as a percentage of the response to 100 mmol K+) curves in human isolated coronary arteries (n = 9) obtained with ergotamine, dihydroergotamine (DHE), sumatriptan, methysergide, and its metabolite methylergometrine. Data, displayed as means ± SEM, are from MaassenVanDenBrink et al. (59). |
We believe that the vasoconstrictor action of methysergide within the carotid vascular bed (79,82), which is mediated by the 5-HT1B receptor (98), is most likely involved in the therapeutic efficacy. The carotid vasoconstrictor effect of methysergide is weaker, both in potency and efficacy, than that of ergotamine (24) or sumatriptan (23,25), which have the ability to abort migraine attacks (for more details, see Chapter 52). Thus, it is possible that methysergide owes its therapeutic effect in migraine to its metabolic product methylergometrine. Indeed, methylergometrine has a more potent vasoconstrictor action than methysergide (59,66).
Apart from 5-HT1B receptors, the craniovascular effects of methysergide (and methylergometrine) are likely to involve 5-HT7 receptor blockade (22,28,98). Also worth investigating is the involvement of dopamine receptors because Bell (6,7) has presented evidence that dilatation of arteriovenous anastomoses can be mediated by a neural release of dopamine.
Inhibition of peptide release from perivascular sensory nerve endings as well as neurogenic inflammation by methysergide, as demonstrated in the rat, also has been invoked as a mechanism of action in migraine (77). But, as argued elsewhere (21), there is considerable doubt whether inhibition of neurogenic inflammation in experimental animals is connected with antimigraine efficacy because several such compounds were found clinically ineffective in migraine.
Results of Clinical Trials
Open Trials
In open studies including approximately 1,400 migraine patients (18,37,39,55), methysergide was found to decrease migraine attack frequency in the majority of patients. However, about 10 (18,55) to 20% (37,39) of the patients had to stop the drug because of side effects. One case of ergotism was observed, and in one case angina pectoris was precipitated (37).
During the first years of clinical use of methysergide it became evident that continuous use of the drug for longer periods can induce retroperitoneal fibrosis, as well as pleural and heart valve fibrosis, with an estimated incidence of 1 in 5,000 treated patients (40,41). In most cases the fibrotic process regressed after discontinuation of methysergide (40). The metabolism of methysergide was unchanged in patients who had developed this side effect (9), and its mechanisms remain elusive. This side effect of methysergide limits its clinical use.
Controlled Clinical Trials
Methysergide has been compared with placebo or another drug in nine double-blind randomized clinical trials (2,5,33,43,71,74,75,91,95). The daily dosage of methysergide varied from 3 to 6 mg. In two trials methysergide was superior to placebo for either severe headaches (91) or frequency of attacks (71), but in one trial methysergide was not superior to placebo (75). In four trials (2,33,71,75), methysergide was found comparable with pizotifen (Table 55-1). Methysergide was also found comparable with lisuride (25 μg three times daily) (42), propranolol (40 mg tid) (5), and flunarizine (10 mg daily) (95).
The side effects reported in these trials were dizziness, nausea and vomiting, weight gain, epigastric pain, and psychic reactions. In some studies high drop-out rates of 20% (42) and 26% (91) occurred with a daily dosage of 6 mg methysergide, but apparently this dosage was tolerated in other studies (33,95).
Taken together, the controlled trials with methysergide show that the drug is efficacious in migraine prophylaxis. The problems with side effects also have been demonstrated for this potent drug.
Therapeutic Use
Because of potentially grave side effects, methysergide should be reserved for severe cases for which other attempts of migraine prophylaxis have not produced optimal results. The daily dosage of methysergide in migraine prophylaxis is 3 to 6 mg given in three divided doses. To minimize the acute side effect, the dosage should be increased gradually, starting with 1 mg per day and increasing with 1 mg every third day. Methysergide should not be taken continuously for long periods, which can result in retroperitoneal fibrosis (4,40,41). Instead, the drug should be given for 6 months, with a 2-month interruption before starting the drug again. When methysergide is stopped it should be weaned off gradually over 1 week to avoid rebound headache.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree